Common Hematology Tests
Common hematology tests are essential diagnostic blood tests used in hematology to evaluate blood, blood-forming organs, and blood disorders. Core hematology investigations such as the full blood count (FBC/CBC), peripheral blood smear/blood film, reticulocyte count, erythrocyte sedimentation rate (ESR), and coagulation studies (PT and aPTT) are indispensable in everyday clinical practice. These tests play a critical role in the detection, monitoring, and treatment planning of a wide range of conditions, including anemia, infections, hemophilia, coagulation disorders, and leukemia. Understanding the interpretation of hematology blood tests provides valuable insight into a patient’s overall health and guides appropriate clinical decision-making.
The components of human blood include:
- Plasma. This is the liquid component of blood in which the following blood cells are suspended.
- Red blood cells (erythrocytes). These carry oxygen from the lungs to the rest of the body.
- White blood cells (leukocytes). These help fight infections and aid in the immune process. Types of white blood cells include the following:
- Lymphocytes
- Monocytes
- Eosinophils
- Basophils
- Neutrophils
- Platelets (thrombocytes). These help to control bleeding.
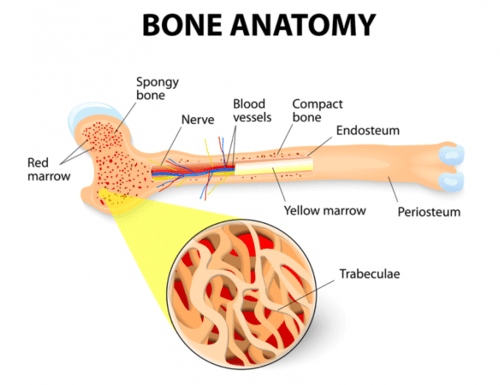
Blood cells are made in the bone marrow. The bone marrow is the soft, spongy material in the center of the bones that produces about 95% of the body’s blood cells. Most of the adult body’s bone marrow is in the pelvic bones, breast bone, and the bones of the spine.
There are other organs and systems in our bodies that help regulate blood cells. The lymph nodes, spleen, and liver help regulate the production, destruction, and differentiation of cells. The production and development of new cells is a process called hematopoiesis.
Blood cells formed in the bone marrow start out as a stem cell. A stem cell (or hematopoietic stem cell) is the initial phase of all blood cells. As the stem cell matures, several distinct cells evolve, such as the red blood cells, white blood cells, and platelets. Immature blood cells are also called blasts. Some blasts stay in the marrow to mature and others travel to other parts of the body to develop into mature, functioning blood cells.
Full blood count (FBC) testing:
Full blood count or FBC testing, also known as a complete blood cell count (CBC) is a routine test that evaluates three major components found in the blood: white blood cells (WBCs), red blood cells (RBCs) and platelets.
A complete blood cell count is a measurement of size, number, and maturity of the different blood cells in a specific volume of blood. A complete blood cell count can be used to determine many abnormalities with either the production or destruction of blood cells. Variations from the normal number, size, or maturity of the blood cells can be used to indicate an infection or disease process. Often with an infection, the number of white blood cells will be elevated. Many forms of cancer can affect the bone marrow production of blood cells. An increase in the immature white blood cells in a complete blood cell count can be associated with leukemia. Anemia and sickle cell disease will have abnormally low hemoglobin.
There are many reasons for FBC testing, but common reasons include infection, anemia, and certain cancers of the blood.
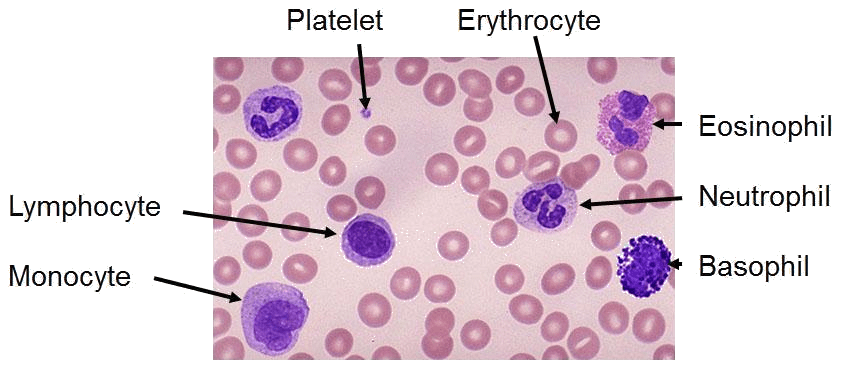
A high-magnification micrograph of a blood film showing the morphology and distribution of erythrocytes, leukocytes, and thrombocytes.
FBC testing is pretty simple and takes just a few minutes. A nurse or lab tech will take a sample of blood by inserting a needle into a vein in your arm.
FBC testing measures several components and features of blood, including:
Red blood cells (Erythrocytes – RBCs): the primary function of red blood cells, or erythrocytes, is to carry oxygen from the lungs to the body tissues, and carbon dioxide as a waste product away from the tissues and back to the lungs. If your RBC count is too low, you may have anemia or another condition. The normal range for men is 5 million to 6 million cells/mcL; for women, it’s 4 million to 5 million cells/mcL.
Hemoglobin (Hb): hemoglobin is an important protein in the red blood cells that carries oxygen from the lungs to all parts of our body. The normal range for men is 14 to 17 grams per deciliter (gm/dL); for women it’s 12 to 15 gm/dL.
Hematocrit: the proportion of red blood cells to the fluid component, or plasma, in the blood. In other words, how much of your blood is red blood cells? A low score on the range scale may be a sign that you have too little iron, the mineral that helps produce red blood cells. A high score could mean you’re dehydrated or have another condition. The normal range for men is between 41% and 50%. For women, the range is between 36% and 44%.
Mean corpuscular volume (MCV): this is the average size of your red blood cells. If they’re bigger than normal, your MCV score goes up. That could indicate low vitamin B12 or folate levels. If your red blood cells are smaller, you could have a type of anemia e.g. iron deficiency anemia or thalassemia. A normal-range MCV score is 80 to 95 femtoliters.
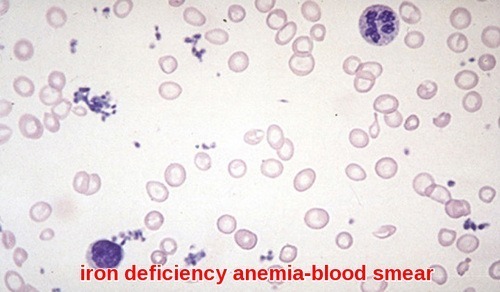
A blood film showing the characteristic microcytic and hypochromic red blood cells typically observed in patients with iron deficiency anemia.
Mean corpuscular hemoglobin (MCH): which is the amount of hemoglobin per red blood cell. The MCH should be 27 to 33 picograms per cell. A low MCH value typically indicates the presence of iron deficiency anemia. In more rare cases, low MCH can be caused by a genetic condition called thalassemia. High MCH value can often be caused by anemia due to a deficiency of B vitamins, particularly B-12 and folate.
Mean corpuscular hemoglobin concentration (MCHC): which is the amount of hemoglobin relative to the size of the cell or hemoglobin concentration per red blood cell. The MCHC should be 33.4 to 35.5 grams per deciliter. The most common cause of low MCHC is anemia. Hypochromic microcytic anemia commonly results in low MCHC. This condition means your red blood cells are smaller than usual and have a decreased level of hemoglobin. When the MCHC is high, the red cells are referred to as being hyperchromic. Possible causes of a high MCHC (which is uncommon) include autoimmune hemolytic anemia (AIHA), a condition in which the bodies immune system attacks its own red blood cells and hereditary spherocytosis, a genetic condition characterized by anemia and gallstones.
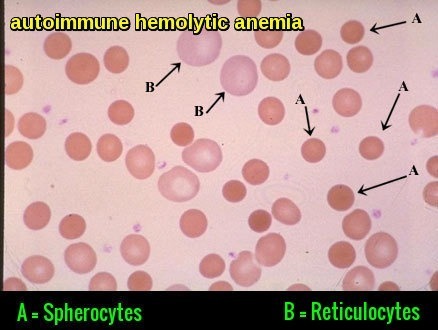
Peripheral blood film in autoimmune hemolytic anemia demonstrating spherocytes (A) and reticulocytes (B), reflecting immune-mediated red cell destruction and marrow response.
Reticulocyte Count: The reticulocyte count is the percentage of immature red blood cells (reticulocytes) in the total red blood cell count (reticulocytes / red blood cells). Reticulocyte Count Normal Range is 1-2% of the total RBC count.

Reticulocytosis demonstrated by increased reticulocytes on peripheral blood smear using Wright stain and supravital new methylene blue stain highlighting ribosomal RNA.
Red blood cell distribution width (RDW): is a measure of the range of variation of red blood cell (RBC) volume that is reported as part of a standard complete blood count. Usually, red blood cells are a standard size of about 6-8 μm in diameter. Certain disorders, however, cause a significant variation in cell size. Higher RDW values indicate greater variation in size. The normal reference range of RDW in human red blood cells is 11.5-14.5%. If anemia is observed, RDW test results are often used together with mean corpuscular volume (MCV) results to determine the possible causes of the anemia. It is mainly used to differentiate an anemia of mixed causes from an anemia of a single cause.
Deficiencies of Vitamin B12 or folate produce a macrocytic anemia (large cell anemia) in which the RDW is elevated in roughly two-thirds of all cases. However, a varied size distribution of red blood cells is a hallmark of iron deficiency anemia, and as such shows an increased RDW in virtually all cases. In the case of both iron and B12 deficiencies, there will normally be a mix of both large cells and small cells, causing the RDW to be elevated. An elevated RDW (red blood cells of unequal sizes) is known as anisocytosis.

Peripheral blood film in megaloblastic anemia demonstrating marked macrocytosis with a characteristic hypersegmented neutrophil, a classic morphological hallmark of vitamin B12 or folate deficiency.
An elevation in the RDW is not characteristic of all anemias. Anemia of chronic disease, hereditary spherocytosis, acute blood loss, aplastic anemia (anemia resulting from an inability of the bone marrow to produce red blood cells), and certain hereditary hemoglobinopathies (including some cases of thalassemia minor) may all present with a normal RDW.
White blood cells (Leukocytes – WBCs):
The primary function of white blood cells, or leukocytes, is to fight infection. They are part of the immune system. They release special enzymes to help protect your body against foreign invaders. There are several types of white blood cells and each has its own role in fighting bacterial, viral, fungal, and parasitic infections.
If you have high WBC levels, it tells your doctor you have inflammation or infection somewhere in your body. If it’s low, you could be at risk for infection. The normal range is 4,500 to 10,000 cells per microliter (cells/mcL). Leukocytosis refers to an increase in the total number of white blood cells (WBCs) due to any cause. From a practical standpoint, leukocytosis is traditionally classified according to the component of white cells that contribute to an increase in the total number of WBCs. Therefore, leukocytosis may be caused by an increase in:
(1) Neutrophil count:
Neutrophils are the most plentiful white blood cells, making up 55 to 70 percent of white blood cell count. The absolute neutrophil count (ANC) ranges between 1.5-8.0 109/l in normal conditions.
Neutrophilia is an increase in circulating neutrophils above that expected in a healthy individual of the same age, sex, race, and physiological status. This represents an increase in the neutrophil count above 8.0×109/l and is one of the most frequently observed changes in the FBC. Neutrophils are the primary white blood cells that respond to a bacterial infection, so the most common cause of neutrophilia is a bacterial infection, especially pyogenic infections. Neutrophils are also increased in any acute inflammation, so will be raised after a heart attack, other infarct or burns.

Peripheral blood film demonstrating neutrophilia with an increased number of mature segmented neutrophils, commonly seen in infection, inflammation, or stress responses.
On the other hand, Neutropenia is when a person has a low level of neutrophils. Some level of neutropenia takes place in about half of people with cancer who are receiving chemotherapy. It is a common side effect in people with leukemia or other conditions that affect the bone marrow directly, such as lymphoma, multiple myeloma, and myelodysplasia. Radiation therapy to several parts of the body or to bones in the pelvis, legs, chest, or abdomen can also cause neutropenia. Autoimmune neutropenia occurs when an individual’s immune system attacks and destroys neutrophils. People who have neutropenia should adhere to good personal hygiene to lower their risk of infection. This includes washing their hands regularly. People who have neutropenia have a higher risk of getting serious infections. This is because they do not have enough neutrophils to kill organisms that cause infection.
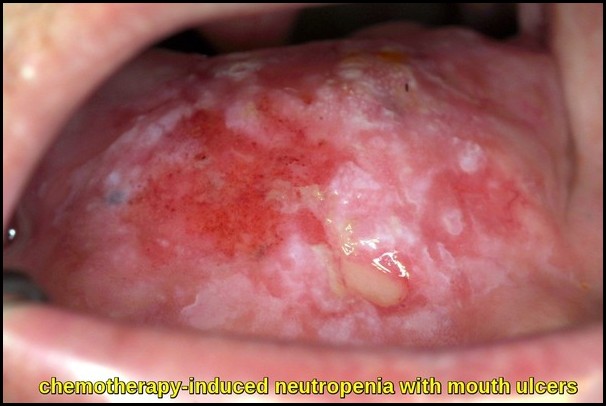
Chemotherapy-induced neutropenia presenting with painful oral ulcers due to severe reduction in neutrophil-mediated mucosal immunity.
For patients with neutropenia, even a minor infection can quickly become serious. They should talk with their health care team right away if they have any of these signs of infection:
- A fever, which is a temperature of 38°C or higher.
- Chills or sweating.
- A sore throat, sores in the mouth, or a toothache.
- Abdominal pain.
- Pain near the anus.
- Pain or burning when urinating, or urinating often.
- Diarrhea or sores around the anus.
- A cough or shortness of breath.
- Any redness, swelling, or pain (especially around a cut, wound, or catheter).
- Unusual vaginal discharge or itching.
(2) Lymphocyte count:
Lymphocytosis is an increase in the number of lymphocytes in the blood. In adults, lymphocytosis is present when the lymphocyte count is greater than 4000 per microliter (4.0×109/l), in older children greater than 7000 per microliter and in infants greater than 9000 per microliter. The three types of lymphocytes are B lymphocytes, T lymphocytes, and natural killer (NK) cells. All of these cells help protect the body from infection. Lymphocytes normally represent 20 to 40% of circulating white blood cells. Lymphocytosis is a feature of infection, particularly in children. In the elderly, lymphoproliferative disorders, including chronic lymphocytic leukemia and lymphomas, often present with lymphadenopathy and a lymphocytosis.
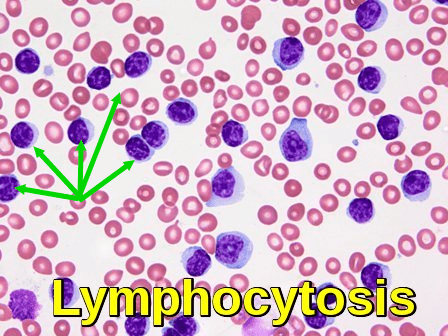
Peripheral blood film demonstrating lymphocytosis with a predominance of circulating lymphocytes, as indicated by arrows.
On the other hand, lymphocytopenia refers to a count of fewer than 1,000 lymphocytes per microliter of blood in adults or less than 3,000 lymphocytes per microliter of blood in children. Most people who have lymphocytopenia have low numbers of T lymphocytes. Sometimes they also have low numbers of the other types of lymphocytes. Many diseases, conditions, and factors can lead to a low lymphocyte count. These conditions can be acquired or inherited.
One of the most common acquired causes of lymphocytopenia is AIDS. Inherited causes include DiGeorge anomaly, Wiskott-Aldrich syndrome, severe combined immunodeficiency syndrome, and ataxia-telangiectasia. These inherited conditions are rare.
Lymphocytopenia can range from mild to severe. The condition alone may not cause any signs, symptoms, or serious problems.
How long lymphocytopenia lasts depends on its cause. The treatment for this condition depends on its cause and severity. Mild lymphocytopenia may not require treatment. If an underlying condition is successfully treated, lymphocytopenia will likely improve.
If lymphocytopenia causes serious infections, medicines or other treatments are required.
(3) Monocyte count:
Monocytes migrate into the tissues where they become macrophages, with specific characteristics depending on their tissue localization. Monocyte cell count in normal adults is 0.2–1.0×109/l (2–10%). Monocytosis is defined as an increase in peripheral blood monocytes greater than 1.0×109/l.
The differential diagnosis is broad, as monocytosis is not representative of a specific condition. It is often a marker of chronic inflammation, either as a result of infection, autoimmune disease or blood born malignancy.
Common infections causing monocytosis include tuberculosis, subacute bacterial endocarditis, syphilis, protozoal or rickettsial disease. Common autoimmune diseases in the differential include SLE, rheumatoid arthritis, sarcoidosis, and inflammatory bowel disease. Malignancy, especially monocytic leukemia, should always be investigated in a patient with monocytosis and appropriate symptom features. Monocytosis can also develop during the recovery phase of an acute infection.

Peripheral blood film demonstrating monocytosis with prominent circulating monocytes highlighted by arrows.
Monocytopenia is a reduction in blood monocyte count to < 0.4×109/l. Monocytopenia can increase the risk of infection, and it can indicate poor prognosis in patients with acetaminophen-induced hepatic damage and thermal injuries. Peripheral blood monocytopenia does not usually indicate a decrease in tissue macrophages; in some cases, it can be associated with impaired granuloma formation in response to infections.
Monocytopenia can result from:
- Chemotherapy-induced myelosuppression (along with other cytopenias).
- Hematopoietic cell mutation involving GATA2.
- Neoplastic disorders (eg, hairy cell leukemia, acute lymphoblastic leukemia, Hodgkin lymphoma).
- Infections (eg, HIV infection, Epstein-Barr virus infection, adenovirus infection, miliary tuberculosis).
- Corticosteroid or immunoglobulin therapy.
- Gastric or intestinal resection.
Transient monocytopenia can occur with endotoxemia, hemodialysis, or cyclic neutropenia.
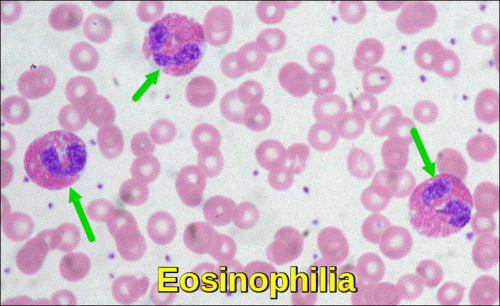
(4) Eosinophilic granulocyte count:
Eosinophils are a type of disease-fighting white blood cell. Eosinophilia is defined as a peripheral blood eosinophil count > 500/μL. Peripheral blood eosinophilia can be caused by numerous allergic, infectious, and neoplastic disorders, which require a variety of different treatments. A major goal of the initial evaluation is to identify disorders that require specific treatments (eg, parasitic infection, drug hypersensitivity, leukemia, non-hematologic cancer). Parasitic diseases and allergic reactions to medication are among the more common causes of eosinophilia. Eosinophilia that causes organ damage is called Hypereosinophilic Syndrome. This syndrome tends to have an unknown cause or results from certain types of cancer, such as bone marrow or lymph node cancer. Treatment is directed at the cause.
Important considerations in approaching patients with eosinophilia are preexisting diagnoses and medications (even if started years ago). Mild eosinophilia can commonly be caused by atopic dermatitis and asthma. However, in patients with eosinophilia over 1500/μL, an alternative diagnosis should be sought. Asymptomatic eosinophilia in a patient without a history of travel outside of the US and Europe is unlikely to have an infectious cause.
(5) Basophilic granulocyte count:
Basophils are a type of white blood cell produced in the bone marrow. Normally, basophils make up less than 1% of your circulating white blood cells.
Basophilia is defined as an elevated absolute basophil count greater than 200 cells/uL or relative basophil count greater than 2%. The elevation of these cells can often suggest underlying diseases, such as a myeloproliferative disorder eg, chronic myeloid leukemia or chronic inflammation.
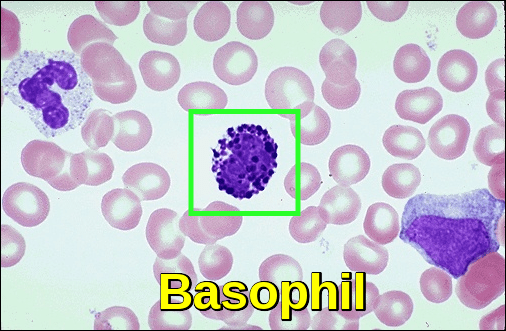
Peripheral blood film demonstrating a basophil with coarse basophilic cytoplasmic granules obscuring the nucleus, a characteristic feature seen on routine blood film examination.
A low basophil level is called Basopenia. It can be caused by infections, severe allergies, or an overactive thyroid gland.
(6) Immature Blood cells: Many people with leukemia have no symptoms at all and are diagnosed during a routine medical examination. The doctor may find an enlarged lymph node or spleen. Likewise, the physical examination may be normal but there is an abnormality on a routine complete blood count (CBC). The CBC may show abnormally high or abnormally low white blood cell counts. In addition, there may be abnormalities of the red blood cells or platelets. In some cases, there may be blasts (immature white blood cells) present in the CBC.
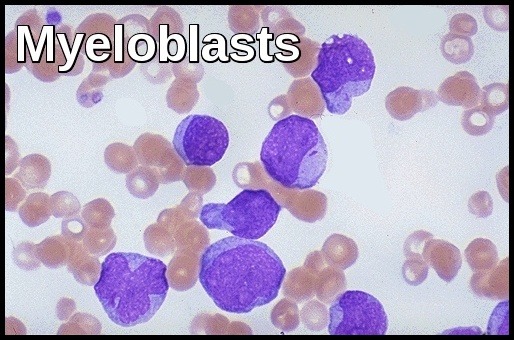
Peripheral blood film demonstrating numerous myeloblasts with high nuclear-to-cytoplasmic ratio, consistent with acute myeloid leukemia.
Platelets (Thrombocytes): platelets, also called thrombocytes, are special cell fragments that play an important role in normal blood clotting. A person who does not have enough platelets may be at an increased risk of excessive bleeding and bruising. An excess of platelets can cause excessive clotting or if the platelets are not functioning properly excessive bleeding. The CBC measures the number and size of platelets present. The normal range is 140,000 to 450,000 cells/mcL.
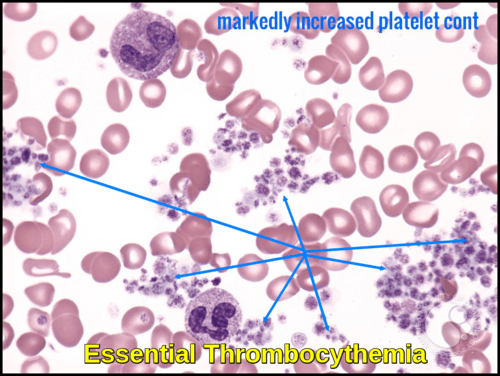
Peripheral blood film demonstrating essential thrombocythemia with marked thrombocytosis and platelet aggregates, as highlighted by arrows.
Erythrocyte Sedimentation Rate (ESR)
Erythrocyte sedimentation rate (ESR or sed rate) is a test that indirectly measures the degree of inflammation present in the body. The test actually measures the rate of fall (sedimentation) of erythrocytes (red blood cells) in a sample of blood that has been placed in a tall, thin, vertical tube. Results are reported as the millimeters of clear fluid (plasma) that are present at the top portion of the tube after one hour.

Erythrocyte sedimentation rate (ESR) measurement using the Westergren method, showing varying degrees of red cell sedimentation.
When a sample of blood is placed in a tube, the red blood cells normally settle out relatively slowly, leaving little clear plasma. The red cells settle at a faster rate in the presence of an increased level of proteins, particularly proteins called acute phase reactants. The level of acute phase reactants such as C-reactive protein (CRP) and fibrinogen increases in the blood in response to inflammation.
Inflammation is part of the body’s immune response. It can be acute, developing rapidly after trauma, injury or infection, for example, or can occur over an extended time (chronic) with conditions such as autoimmune diseases or cancer e.g. multiple myeloma.
The ESR is not diagnostic; it is a non-specific test that may be elevated in a number of these different conditions. It provides general information about the presence or absence of an inflammatory condition.
There have been questions about the usefulness of the ESR in light of newer tests that have come into use that are more specific. However, ESR test is typically indicated for the diagnosis and monitoring of temporal arteritis, systemic vasculitis, and polymyalgia rheumatica. Extremely elevated ESR is useful in developing a rheumatic disease differential diagnosis. In addition, ESR may still be a good option in some situations, when, for example, the newer tests are not available in areas with limited resources or when monitoring the course of a disease.
Clotting Screen:
When a body tissue is injured and begins to bleed, it initiates a sequence of clotting factor activities – the coagulation cascade – leading to the formation of a blood clot. This cascade is comprised of three pathways: extrinsic, intrinsic, and common.

Diagram of the blood coagulation cascade showing the intrinsic and extrinsic pathways converging on factor Xa and the common pathway.
Routine clotting tests comprise of the prothrombin time (PT), fibrinogen, activated partial thromboplastin time (APTT), thrombin time and D-Dimer. Abnormal clotting results that cannot be explained in the clinical context should be investigated further by a Specialist Thrombosis and Haemostasis laboratory.
The clotting screen is a bundled group of tests used pre-operatively to assess bleeding risk and used to monitor bleeding conditions and some therapies. It tests the PT (and INR), APTT and fibrinogen.
Prothrombin Time (PT):
The PT measures the vitamin K dependent clotting pathways (extrinsic pathway) and is therefore of particular use in measuring the effect of warfarin therapy (warfarin is a vitamin K antagonist). The vitamin K-dependent coagulation factors are factors II, VII, IX, X (and proteins C, S, and Z). Normal ranges for the PT vary up and down worldwide depending on the sensitivity of the reagent and the technology used to determine the clotting times. The INR (international normalized ratio) is a calculation which is used that takes into account the normal clotting times and the sensitivity of the reagent used to provide a ratio which is the same worldwide. For example, a PT of 16 seconds in Leeds and a PT of 33 seconds in London can produce the same INR. The prothrombin time is also used to determine if there are any deficiency in extrinsic clotting factors and is a useful liver function test. Raised PTs without cause should be investigated further.
Activated Partial Thromboplastin Time (APTT):
Measures the intrinsic clotting pathway. The intrinsic pathway requires the clotting factors VIII, IX, X, XI, and XII. Also required are the proteins prekallikrein (PK) and high-molecular-weight kininogen (HK or HMWK), as well as calcium ions and phospholipids secreted from platelets. APTT is particularly useful in monitoring heparin therapy. The APTT ratio provides the ratio of APPT : Normal Clotting time and is the primary calculation used to monitor heparin therapy. The APTT is also useful in detecting clotting factor deficiencies of the intrinsic pathway and can be raised in the presence of factor deficiencies and lupus anticoagulants. Raised APTTs without cause should be investigated further.
* The INR and APTT ratio do not have normal ranges but therapeutic ranges. These are based on the condition being treated and are decided by the clinical teams.
Thrombin Time (TT):
Is primarily requested by the Liver Disease Units and measures the time it takes for fibrinogen to form fibrin (one of the later stages of the common clotting pathway). It is also requested by the laboratories to confirm the presence of heparin contamination of a sample in the event of an unexplained raised APTT.
Fibrinogen:
The fibrinogen reported routinely is derived from the PT reaction as it occurs. It is primarily a screen. Any low fibrinogen detected by this method is substituted for the Clauss Fibrinogen test which measures fibrinogen directly. In DIC (Disseminated Intravascular Coagulation) the derived fibrinogen may be misleading. Supplying relevant clinical details will ensure the biomedical scientists report the most appropriate fibrinogen for the clinical situation. The Clauss Fibrinogen can be measured routinely if asked for on the request card and if DIC is suspected; the Clauss fibrinogen should always be measured.
D-Dimers:
D-dimer is one of the protein fragments produced when a blood clot gets dissolved in the body. It is normally undetectable or detectable at a very low level unless the body is forming and breaking down blood clots. Then, its level in the blood can significantly rise. This test detects D-dimer in the blood.
It is, therefore, a useful predictor of recent clot formation. It is not specific however and can be affected by many other conditions such as rheumatoid arthritis. It should only be used as a negative predictor for VTE (venous thromboembolism) i.e. a raised D-Dimer is not diagnostic of a clot formation, but a normal D-Dimer can be a used as a negative predictor of venous thrombosis. In DIC the D-Dimer levels are often very high.
Questions and Answers:
What are the most common hematology tests?
The most common hematology tests include the full blood count (FBC or CBC), peripheral blood smear, reticulocyte count, erythrocyte sedimentation rate (ESR), and coagulation studies such as prothrombin time (PT) and activated partial thromboplastin time (aPTT), all of which are routinely used to assess blood disorders and overall health.
What does a full blood count (FBC or CBC) show?
A full blood count provides quantitative information about red blood cells, white blood cells, hemoglobin, hematocrit, and platelets, helping to detect anemia, infection, inflammation, bone marrow disorders, and hematological malignancies.
Why is a peripheral blood smear (blood film) important?
A peripheral blood smear allows direct microscopic examination of blood cell morphology, enabling identification of abnormal red cells, white cells, platelets, blasts, and inclusion bodies that may not be reliably detected by automated analyzers alone.
What does an increased reticulocyte count indicate?
An increased reticulocyte count reflects enhanced bone marrow red cell production and is typically seen in hemolysis, acute blood loss, or recovery from anemia, indicating an appropriate marrow response. The common causes of reticulocytosis can be remembered by the 3H mnemonic: Hemolysis, Hemorrhage, and Hematinic response, such as recovery following iron, vitamin B12, or folate replacement in previously deficient patients.
What causes macrocytosis and hypersegmented neutrophils on blood film?
Macrocytosis with hypersegmented neutrophils is characteristic of megaloblastic anemia, most commonly caused by vitamin B12 or folate deficiency due to impaired DNA synthesis.
What is neutrophilia and what does it suggest?
Neutrophilia refers to an increased number of neutrophils in the blood and is commonly associated with bacterial infections, inflammation, corticosteroid use, physiological stress, or myeloproliferative disorders.
What are the causes of lymphocytosis on a blood test?
Lymphocytosis may be reactive, as seen in viral infections and chronic inflammation, or clonal, as in lymphoproliferative disorders such as chronic lymphocytic leukemia, and is identified through white cell differential analysis and blood film examination.
What does monocytosis indicate in hematology tests?
Monocytosis is associated with chronic infections, inflammatory and autoimmune conditions, recovery from neutropenia, or myeloid neoplasms such as chronic myelomonocytic leukemia, and requires clinical correlation.
What conditions are associated with eosinophilia?
Eosinophilia is commonly linked to allergic disorders, parasitic infections, drug reactions, autoimmune diseases, and certain hematologic malignancies, and is assessed through differential white cell counts and blood film review.
What is the clinical significance of basophilia?
Basophilia is uncommon but may be seen in myeloproliferative neoplasms, particularly chronic myeloid leukemia, as well as in allergic and inflammatory conditions, and should prompt further hematologic evaluation.
How does smoking affect blood counts?
Smoking can significantly affect blood counts by increasing hemoglobin, hematocrit, and red blood cell count due to chronic carbon monoxide exposure and resulting tissue hypoxia. It may also cause leukocytosis, particularly neutrophilia, and mild thrombocytosis as part of a chronic inflammatory response. These changes can mask underlying anemia or polycythemia and should be considered when interpreting hematology test results.
What does the erythrocyte sedimentation rate (ESR) measure?
ESR is a non-specific marker of inflammation that reflects the rate at which red blood cells settle in plasma and may be elevated in infections, autoimmune diseases, malignancy, and chronic inflammatory states.
How do PT and aPTT help assess coagulation disorders?
Prothrombin time evaluates the extrinsic and common coagulation pathways, while activated partial thromboplastin time assesses the intrinsic and common pathways, aiding in the diagnosis of bleeding disorders and monitoring anticoagulant therapy.
When should fibrinogen be checked as part of the clotting profile?
Fibrinogen should be checked as part of the clotting profile in patients with unexplained bleeding or thrombosis, suspected disseminated intravascular coagulation (DIC), severe liver disease, massive hemorrhage, sepsis, or obstetric complications. It is also useful when PT and aPTT are prolonged or when monitoring replacement therapy in hypofibrinogenemia or consumptive coagulopathies.
What blood tests are used to detect anemia?
Anemia is primarily detected using a full blood count to assess hemoglobin, red cell indices, and hematocrit, with further evaluation by peripheral blood smear, reticulocyte count, and iron, vitamin B12, or folate studies when indicated.
Can blood tests detect leukemia?
Blood tests may suggest leukemia through abnormal white cell counts, circulating blasts, anemia, or thrombocytopenia, but definitive diagnosis requires blood film review, bone marrow examination, immunophenotyping, and genetic testing.
What does a high platelet count mean?
A high platelet count, known as thrombocytosis, may be reactive due to inflammation, infection, or iron deficiency, or clonal as seen in myeloproliferative neoplasms such as essential thrombocythemia.
Why is a blood film requested after an abnormal CBC?
A blood film is requested to visually assess cell morphology and confirm or clarify abnormalities detected on automated blood counts, including abnormal white cells, red cell shapes, or platelet disorders.
Do hematology blood tests require fasting?
Most routine hematology tests, including full blood count, blood film, reticulocyte count, and coagulation studies, do not require fasting unless they are combined with other biochemical investigations.
References:
Evans TC., Jehle D. “The red blood cell distribution width.” Journal of Emergency Medicine. 1991; 9(Suppl 1):71–74.
Inoue S., Willert J R. “Leukocytosis: Background, Pathophysiology, Epidemiology.” Medscape. Available at: https://emedicine.medscape.com/article/956278-overview
Miale JB. Laboratory Medicine: Hematology. 5th ed. St. Louis, MO: C.V. Mosby; 1977.
O’Connell EM., Nutman TB. “Eosinophilia in Infectious Diseases.” Immunology and Allergy Clinics of North America. 2015; 35(3):493-522. DOI:10.1016/j.iac.2015.04.005.
Johns Hopkins Medicine. Hematology | Health Library. Available at: https://www.hopkinsmedicine.org/healthlibrary/conditions/pathology/hematology_85,P00958
Johns Hopkins Medicine. Facts About Blood | Health Library. Available at: https://www.hopkinsmedicine.org/healthlibrary/conditions/hematology_and_blood_disorders/facts_about_blood_85,P00083
National Heart, Lung, and Blood Institute (NHLBI). Lymphocytopenia. Available at: https://www.nhlbi.nih.gov/health-topics/lymphocytopenia
Princess Margaret Cancer Centre, University of Toronto. Chemotherapy-Induced Neutropenia with Mouth Ulcers. Available at: http://pmhdentaloncology.ca/chemotherapy.php
Cancer.Net (ASCO). Neutropenia. Available at: https://www.cancer.net/navigating-cancer-care/side-effects/neutropenia
WebMD. What Is a Complete Blood Count (CBC)? Available at: https://wb.md/2xMbi3W
Doyle B. “Monocytosis – Hospital Medicine.” Cancer Therapy Advisor. Available at: https://www.cancertherapyadvisor.com/hospital-medicine/monocytosis/article/601608/
Kellner C. “Erythrocyte Sedimentation Rate (ESR).” Medscape. Available at: https://emedicine.medscape.com/article/2085201-overview
Territo M., Geffen D. “Monocytopenia.” MSD Manual Professional Edition. Available at: https://www.msdmanuals.com/professional/hematology-and-oncology/leukopenias/monocytopenia
McGill Physiology Virtual Lab. “Hemostasis – Blood Clot Formation (PT and aPTT).” Available at: https://www.medicine.mcgill.ca/physio/vlab/bloodlab/PT_PTT.htm
O’Sullivan M. “Clotting Screen.” The Leeds Teaching Hospitals NHS Trust. Updated February 21, 2017. Available at: http://www.pathology.leedsth.nhs.uk/pathology/ClinicalInfo/Haematology/ClottingScreen.aspx
Hoffbrand AV., Higgs DR., Keeling DM., Mehta AB. Postgraduate Haematology. 7th ed. Wiley-Blackwell; 2016.
Rodak BF., Fritsma GA., Keohane EM. Hematology: Clinical Principles and Applications. 6th ed. Elsevier; 2020.
Merck Manual Professional Edition. “Overview of Hematologic Tests.” Available at: https://www.merckmanuals.com/professional/hematology-and-oncology/approach-to-the-patient-with-hematologic-disease/overview-of-hematologic-tests
StatPearls Publishing. “Complete Blood Count – StatPearls [Internet].” Treasure Island (FL): StatPearls. Available at: https://www.ncbi.nlm.nih.gov/books/NBK431097/
Mayo Clinic. “Complete Blood Count (CBC) – Purpose and Results.” Available at: https://www.mayoclinic.org/tests-procedures/complete-blood-count/about/pac-20384919
Keywords:
common hematology tests, hematology investigations, hematology lab tests, hematology laboratory procedures, complete blood count, full blood count, CBC test, CBC interpretation, peripheral blood smear, blood film examination, reticulocyte count, ESR test, erythrocyte sedimentation rate, platelet count, white blood cell count, red blood cell count, hemoglobin test, hematocrit test, mean corpuscular volume, RDW, red blood cell distribution width, coagulation profile, prothrombin time, PT test, activated partial thromboplastin time, aPTT test, INR test, fibrinogen level, bleeding time, clotting screen, bone marrow examination, anemia workup, thrombocytopenia tests, leukocytosis causes, eosinophilia causes, neutropenia management, lymphocytopenia causes, hematology reference ranges, blood disorders diagnosis, hematology tests explained, hematology results interpretation, hematology parameters, diagnostic blood tests, Dr Moustafa Abdou hematology articles, Ask Hematologist blood investigations, purpose of CBC test, when to order aPTT, risks of bone marrow examination, blood smear differential diagnosis, coagulation profile algorithm

Request Online Consultation With Dr M Abdou
Fee: US$100
Secure payment via PayPal (credit and debit cards accepted)
Pay Now

Leukaemia is a type of cancer that affects blood and its components. Once the marrow cell undergoes a leukaemia change, the leukaemia cells may grow and survive better than normal cells. Over time, the leukaemia cells crowd out or suppress the development of normal cells.
The rate at which leukaemia progresses is different from each type of leukaemia. Leukaemia can broadly be divided into rapidly growing or acute leukaemia and more indolent, chronic leukaemia. A piece of proper knowledge about this disease is essential as it helps patients and his caregivers to cope better with this disease. After diagnosis and treatment, a large number of people with leukaemia live many good, quality years.
I’m wondering my blood results always seem normal to doctors but have low levels of Mean cell hemoglobin, mean cell volume, and Meamaocrit.
But the red blood cell count is always high.
Hemoglobin level is always between (12 and 13.5 g/dL)- Normal as to the report.
And tested negative for thalassemia.
Should I worry about a serious health issue?
Hi Amy,
Thank you for your comment.
Have you checked your fasting serum iron and transferrin saturation? You might have some degree of iron deficiency!
https://askhematologist.com/iron-deficiency-anemia/
Also, there are many other forms of abnormal hemoglobins not only thalassemia which you need to exclude if your serum iron is normal.
https://askhematologist.com/abnormal-hemoglobins/
Best wishes,
Hi!
Wondering what the following means:
XT RET % and XT RET#
Thank you so much! (Testing for clotting disorder, am on eliquis for small DVT).
Hi Gina,
Sorry, I don’t know what the abbreviations XT RET % and XT RET# stand for!
BW,
hi,
would appreciate your advise if there is a concern of haematology esoinophils result shown 7.3% abs 0.5? reference range shown on test is (0.04 – 0.40) – What should i be prepared for ?
Also urine micorsocopy shown white blood cells result at 31 vs ref. range (<7) – result seems alarming.
thank you
Hi Alice,
Thank you for your comment.
This mild eosinophilia is likely reactive to an underlying infection, allergy, hypersensitivity reaction to a medicine, etc.
It shouldn’t cause concern but I would suggest repeating your CBC after 4 weeks to see if it has resolved or not.
BW,
Sir,
Can i share my Hematology Test report tomorrow in comments for your review & Advise.
Thanks & Regards,
Amirzada Mumtaz
Hi Amirzada,
Thank you for your comment.
It would be better to send your personal blood results to our contact us email address
Thank you,
Sir,
Would you please Share Email ID, So i can share Reports.
Thanks & Regards,
Amirzada
in**@*************st.com
What is the purpose of ASO TITRE test? If it is more than required limits, how it will be effected to human health? Could you please kindly advise me.
Hi Rao,
The ASO titer test measures antibodies produced by your body in response to a toxin known as streptolysin O.
Streptolysin O is a toxin produced by group A Streptococcus (GAS) bacteria.
Your body makes the antistreptolysin O antibodies when you have a strep infection caused by GAS bacteria.
I would suggest reading this useful article:
Antistreptolysin-o-titer
BW,
Hi! I have dysautonomia (POTS) and have had a positive ANA of 1:320 (speckled) for the last two years. Since I have had periodic leg pain, GI issues, lethargy in the morning, and cannot exercise much due to fatigue, I saw a rheumatologist last year. My blood work for lupus was negative so no treatment was recommended. I had annual blood work done recently and my neutrophils are low (23%) and my lymphocytes are high (64%). Total WBC is 4.5. I have not been ill, just tired. Is this cause for revisiting a rheumatologist?
Hi Li,
Thank you for your comment.
The absolute neutrophil count gives a better idea if you have neutropenia or not. What you actually have is called “relative neutropenia” and “relative lymphocytosis” because your total white cell count is within the normal range. Neutropenia is commonly seen in autoimmune disorders in the context of your positive ANA.
BW,
Evening my wife of 41years always have her HB results of 7.6 and anamic with numbness since 2012 advice
Hi Mike,
Thank you for your comment.
You can send me her CBC to have a look through (contact us).
Initially, you need to check her hematinics (iron, folate, and B12), reticulocyte count, bilirubin, LDH, blood smear, and Hb electrophoresis.
BW,
My Dr. ran a blood clotting panel on me to see if HRT was an option for me. It came back as positive for factor 5 leiden and anti thrombin 3 deficiency. Do I need to do anything about this other than inform doctors prior to a surgery? I am in my 40s so pregnancy is not an issue. Thank you for your help.
Hi Amy,
Thanks for your comment.
In view of your FVL and AT3 deficiency, I discourage the use of HRT in your case.
However, the risk of vascular complications depends on the route of administration (oral versus transdermal), age, duration of administration, and type of hormones.
The use of HRT in people with acquired and/or hereditary thrombophilias (eg, factor V Leiden, prothrombin mutation G20210A, and others) lead to a further increase of risk for venous thromboembolism, stroke, or myocardial infarction.
HRT via the transdermal route seems to be the safest opportunity for hormone replacement therapy if its use is unavoidable.
BW,
Hi I’m a family practitioner trying to work my way back into primary care after a few decades working rural ER’s. I find myself encountering a number of clinic patients in whom I’m clumsily trying to r/o a plasma cell dyscrasia as a cause of their findings, be that elevated serum protein, bone pain, fatigue, etc. The lab testing now available nowadays is dizzying compared to what I remember back in the day. My question is: In a general sense, is SPEP and maybe a spot UPCR a reasonable place to start? If this is not a reasonably simple yes-or-no question then I’ll dive into Up-to-Date and try to ferret out the answer. Thanks!
Hi Len,
Thanks for your comment.
If you suspect a plasma cell dyscrasia I would suggest checking CBC, U&Es, serum calcium, serum immunoglobulins, and SPEP.
BW,
What workup would you order for a new patient with Lymphocytosis?
Hi Eileen,
Thank you for your comment.
Initially if isolated lymphocytosis I repeat the CBC after 4 weeks with a blood film and CRP.
If lymphocytosis is persistent >>> blood flow cytometry to out rule a lymphoproliferative disorder.
BW,
very good info.
Hello Gary,
I’m glad to hear that you found the article helpful.
Best wishes,
Dr. M. Abdou