Chronic Myeloid Leukemia

Philadelphia chromosome in chronic myeloid leukemia (CML) illustrating the t(9;22)(q34;q11) BCR-ABL1 fusion.
Chronic myeloid leukaemia (CML), also known as chronic myelogenous leukaemia, is a clonal myeloproliferative neoplasm driven by the BCR-ABL1 fusion (Philadelphia chromosome), resulting in uncontrolled proliferation of the granulocytic lineage while preserving normal maturation. It typically presents in the chronic phase but may progress to accelerated or blast phase if untreated. CML is classified under ICD-10 code C91.10.

Philadelphia chromosome demonstrated on karyotype with shortened chromosome 22 characteristic of chronic myeloid leukemia (CML).
Aetiology:
In chronic myeloid leukemia (CML) there is a malignant transformation of the multipotential hematopoietic stem cells (pluripotent stem cells). There are excessive cells in the granulocytic series, primarily in the bone marrow but also in extramedullary sites (e.g. spleen, liver), although these malignant cells do differentiate nearly normally.
Although granulocyte production predominates, the neoplastic clone includes RBCs, megakaryocytes, monocytes, and even some T and B cells.
The term “chronic” in chronic myelogenous leukemia indicates that this cancer tends to progress more slowly than in acute forms of leukemia.
CML accounts for about 15% of all adult leukemias. CML can strike at any age, although it is uncommon before age 10, and the median age at diagnosis is 45 to 55. CML may occur in either sex.
Pathophysiology:
Most cases of CML appear to be induced by a translocation known as the Philadelphia (Ph) chromosome, which is demonstrable in 95% of patients. It is a reciprocal translocation t(9;22) in which a piece of chromosome 9 containing the oncogene c-abl is translocated to chromosome 22 and fused to the gene BCR. The fusion gene BCR-ABL is important in the pathogenesis and expression of CML and results in the production of a specific tyrosine kinase.
CML has 3 phases:
- Chronic phase: An initial indolent period that may last months to years.
- Accelerated phase: Treatment failure with increasing malaise, infections and fevers, bone pain, worsening anemia, progressive thrombocytopenia or thrombocytosis, persistent or worsening splenomegaly, clonal evolution, increasing blood basophils, and increasing marrow or blood blasts.
- Blast phase: Accumulation of blasts in extramedullary sites (eg, bone, CNS, lymph nodes, skin), blasts in blood or marrow increased to > 20%. These are usually myeloblasts but lymphoid blast crisis occurs in about 20% of cases. A myelofibrotic crisis occurs in some. The blast phase leads to fulminant complications resembling those of acute leukemia, including sepsis and bleeding. Some patients progress directly from the chronic to the blast phase.
Clinical features of chronic phase CML:
There is an insidious onset of:
- Anorexia and weight loss.
- Symptoms of anemia.
- Fever.
- Night sweats.
- Easy bleeding.
- Splenomegaly, which may be massive ± spleen pain.
- Hepatomegaly.
Investigations:
- FBC reveals raised white cell count with immature forms in the blood and often increased eosinophils and basophils. Total WBC count: 20,000-60,000 cells/μL.
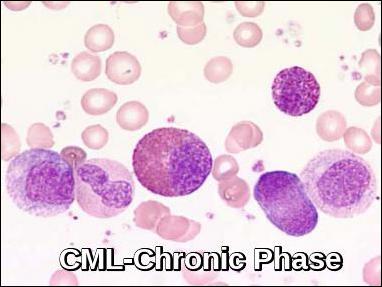
Chronic phase chronic myeloid leukemia (CML) blood smear demonstrating mature granulocytes with a left shift and preserved differentiation.
- The Hb is often initially normal and then falls. Anemia is usually normochromic and normocytic.
- Platelet count up or down.
- The neutrophil alkaline phosphatase score is very low to absent in most cells.
- Marrow is hypercellular with gross myeloid hyperplasia. Megakaryocytes are prominent and may be increased. Mild fibrosis in the reticulin stain.
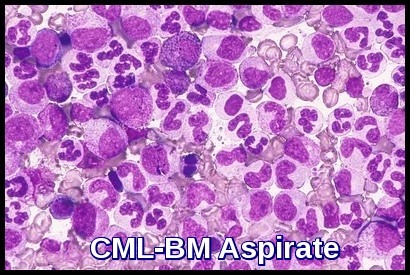
Bone marrow aspirate in chronic myeloid leukemia (CML) demonstrating granulocytic hyperplasia with abundant myeloid precursors.
- Diagnosis is confirmed by finding the Philadelphia chromosome in samples examined with cytogenetic or molecular studies, although it is absent in 5% of patients. Philadelphia chromosome (translocation of the long arm of 22 to the long arm of 9) is present in 95% of cases. This results in the activation of a hybrid bcr-abl proto-oncogene, which can be detected by reverse transcriptase polymerase chain reaction.
Treatment:
The treatment goals of chronic myelogenous leukemia (CML) are threefold and have changed markedly in the past 10 years. They are as follows:
Hematologic remission (normal FBC and physical examination ie, no organomegaly).
Cytogenetic remission (normal chromosome returns with 0% Philadelphia chromosome–positive (Ph+) cells).
Molecular remission (negative polymerase chain reaction [PCR] result for the mutational BCR/ABL mRNA), which represents an attempt for cure and prolongation of patient survival.
Treatment of CML includes the following measures:
- Supportive treatment: transfusions, treatment of infections, Allopurinol for gout.
- A Tyrosine Kinase Inhibitor (TKI), sometimes with chemotherapy.
- Sometimes, stem cell transplantation.

Tyrosine kinase inhibitor (TKI) mechanism of action in chronic myeloid leukemia, blocking abnormal BCR-ABL–driven signalling pathways.
Except when stem cell transplantation is successful, treatment is not known to be curative.
However, since the advent of tyrosine kinase inhibitors (TKIs), chronic myeloid leukemia (CML) has been transformed from a disease that was usually fatal into one in which patients have a close-to-normal life expectancy. When TKIs are used, survival is prolonged, and maximum overall survival is not reached. Some patients may be able to discontinue tyrosine kinase inhibitors and remain in remission. The durability of these remissions is not yet known.
Imatinib (Gleevec) and several newer drugs e.g. Dasatinib (Sprycel), Nilotinib (Tasigna), Bosutinib (Bosulif) and Ponatinib (Iclusig) inhibit the specific tyrosine kinase that results from the BCR-ABL gene product.
Imatinib (Gleevec) was the first drug to specifically target the BCR-ABL tyrosine kinase protein, and it quickly became the standard treatment for CML patients. Because it was the first TKI, imatinib is known as a first-generation tyrosine kinase inhibitor.

Imatinib mesylate, a first-line tyrosine kinase inhibitor (TKI) used in the treatment of chronic myeloid leukemia (CML).
Almost all CML patients respond to treatment with imatinib, and most of these responses seem to last for many years. This drug doesn’t seem to make leukemia go away and stay away, so patients need to take it indefinitely (or until it stops working). Imatinib is taken by mouth as a pill with food, usually once a day. Common side effects can include diarrhoea, nausea, muscle pain, and fatigue. These are generally mild. About 30% of people taking the drug have itchy skin rashes. Most of these symptoms can be treated effectively if needed. Another common side effect is fluid buildup around the eyes, feet, or abdomen. In rare cases, the fluid may collect in the lungs or around the heart, which can cause trouble breathing.
Imatinib also is superior to other treatments in the accelerated and blast phases. In the blast phase, chemotherapy combinations with imatinib have a higher response rate than therapy with either approach alone.
Tyrosine kinase inhibitors (TKIs) are dramatically effective in achieving complete clinical and cytogenetic remissions of Ph chromosome–positive CML and are clearly superior to other regimens (e.g., interferon with or without cytarabine).
Treatment tolerance is excellent. The high level of durable complete remissions associated with TKI therapy has led to the prospect of a cure for the disease. However, the gene products of some BCR-ABL mutations are resistant to current TKIs and remain very difficult to control.
Recently, increasing numbers of patients with CML have begun to pursue a dose optimization strategy. Dose optimization may be considered at all stages of the entire treatment, which includes dose reduction and discontinuation of TKIs therapy. In general, reduction of the TKI dose is considered to be an important measure to reduce adverse events (AEs) and improve quality of life on the premise of maintaining molecular responses. Furthermore, discontinuing TKIs therapy has been demonstrated to be feasible and safe for about half of patients with a stable optimal response and a longer duration of TKI treatment.
A dose-reduction regimen could allow for broader clinical use of TKIs (even in patients with comorbidities). For example, if the elderly patient has multiple comorbidities or is previously intolerant to other TKIs, we may suggest a half-dose of dasatinib treatment (50 mg/day). If conditions permit, the dose can also be adjusted according to blood concentration monitoring. For patients with sustained optimal clinical response with a major molecular response (MMR, BCR::ABL1IS ≤ 0.1%) or deep molecular response (DMR, BCR::ABL1IS ≤ 0.01%), reducing TKIs dose can reduce adverse events and improve treatment compliance. The proposal of treatment-free remission (TFR) as a possible final treatment endpoint should be discussed with patients (especially younger patients) at the diagnosis to achieve a DMR rapidly and improve long-term compliance. For patients with stable DMR and long duration of TKIs treatment, it is feasible and safe to stop TKI treatment. Patients who discontinued TKIs should follow the discontinuation standards recommended in ELN or NCCN guidelines. For patients with stable DMR who want to stop TKIs treatment but are afraid of relapse, we recommend to reduce TKIs dose before discontinuation of TKIs therapy. For patients who cannot achieve TFR, the TKIs dose must be reduced without affecting the clinical response. Importantly, patients who underwent dose optimization should be advised for more intensive molecular monitoring, especially during the first 6 months. Once the patients lose the optimal response, physicians should take measures immediately, such as resuming to standard-dose therapy, reintroducing TKIs treatment, or switching to other TKIs, etc. However, evidence for dose optimization in pediatrics CML is limited. Hence, evidence from novel, prospective clinical trials and real-life clinical practice are required to explore dose-optimization strategies, which may provide more promising options for CML treatment.
Older chemotherapy regimens are reserved for BCR-ABL negative patients, patients who relapse after receiving a TKI, and patients in the blast phase. The main agents are busulfan, hydroxyurea, and interferon. Hydroxyurea is the easiest to manage and has the fewest adverse effects.
Allogeneic stem cell transplantation can be useful for patients refractory to therapy.
Although splenic radiation is rarely used, it may be helpful in refractory cases of CML or in patients with terminal disease and marked splenomegaly.
Splenectomy may alleviate abdominal discomfort, lessen thrombocytopenia, and relieve transfusion requirements when splenomegaly cannot be controlled with chemotherapy or irradiation. Splenectomy does not play a significant role during the chronic phase of CML.
CML in childhood:
This may resemble adult disease, but classical “Juvenile CML” is Philadelphia chromosome negative, and is characterized by a prominent monocytosis and a high HbF level.
Summary:
Chronic myeloid leukaemia (CML) is a clonal myeloproliferative neoplasm characterised by the BCR-ABL1 fusion gene (Philadelphia chromosome), leading to uncontrolled granulocytic proliferation with preserved maturation. CML typically presents in the chronic phase but may progress to accelerated or blast phase without effective treatment. Diagnosis relies on peripheral blood findings, bone marrow examination, cytogenetic analysis, and confirmation of the BCR-ABL1 transcript using PCR or FISH. Tyrosine kinase inhibitors (TKIs) such as imatinib, dasatinib, and nilotinib remain the cornerstone of therapy, producing deep molecular responses and significantly improving long-term survival. Regular molecular monitoring, assessment of treatment milestones, and management of resistance or intolerance are essential components of modern CML care. With appropriate therapy and follow-up, most patients achieve excellent prognoses and sustained remission.
Questions and Answers:
What causes Chronic Myeloid Leukemia (CML)?
CML is caused by the BCR-ABL1 fusion gene created by the Philadelphia chromosome t(9;22)(q34;q11), which produces a constitutively active tyrosine kinase that drives uncontrolled myeloid proliferation.
What are the typical blood findings in chronic phase CML?
Chronic phase CML shows marked leukocytosis with granulocytic left shift, basophilia, eosinophilia, and preserved maturation, often accompanied by mild anaemia and thrombocytosis.
How is the Philadelphia chromosome detected in CML?
The Philadelphia chromosome is identified through cytogenetic karyotyping, FISH for BCR-ABL1, or quantitative RT-PCR, which is essential for diagnosis, baseline assessment, and monitoring response.
What does the bone marrow aspirate show in CML?
The bone marrow aspirate typically demonstrates marked granulocytic hyperplasia with increased myeloid precursors, reduced fat spaces, and a myeloid-to-erythroid ratio significantly shifted toward myelopoiesis.
What is the first-line treatment for CML?
First-line treatment is tyrosine kinase inhibitor (TKI) therapy, most commonly Imatinib, Dasatinib, or Nilotinib, which target the abnormal BCR-ABL1 tyrosine kinase and induce deep molecular remission.
What are the most common side effects of TKIs used in CML?
TKIs frequently cause fatigue, nausea, diarrhoea, periorbital oedema, muscle cramps, cytopenias, and fluid retention. Some TKIs also carry cardiovascular, metabolic, or pleural effusion-related risks depending on the agent.
Can Imatinib cause skin rash or itching?
Yes. Imatinib commonly causes skin rash, pruritus, or mild erythematous eruptions, which are usually manageable but may require dose interruption or antihistamines in persistent or severe cases.
How is treatment response monitored in CML?
Response is monitored using serial BCR-ABL1 PCR testing at 3, 6, and 12 months to confirm achievement of molecular milestones, alongside blood counts, clinical assessment, and periodic specialist review.
What is the prognosis for patients with CML on TKI therapy?
With modern TKI therapy, most patients achieve long-term survival with deep molecular responses, and many can maintain durable remission for decades with excellent quality of life.
References:
Kantarjian H, Cortes J. Chronic myeloid leukemia. In: Abeloff MD, Armitage JO, Lichter AS, Niederhuber JE, Kastan MB, McKenna WG. Clinical Oncology. 4th ed. Philadelphia, PA: Elsevier; 2008:2279–2289.
Druker BJ, Marin D. Chronic myelogenous leukemia. In: DeVita VT, Lawrence TS, Rosenberg SA, eds. DeVita, Hellman, and Rosenberg’s Cancer: Principles and Practice of Oncology. 10th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2015:1644–1653.
National Comprehensive Cancer Network (NCCN). NCCN Clinical Practice Guidelines in Oncology: Chronic Myelogenous Leukemia. V.1.2015. Accessed January 8, 2015.
Cortes JE, Talpaz M, O’Brien S, et al. Staging of chronic myeloid leukemia in the imatinib era: evaluation of the World Health Organization proposal. Cancer. 2006;106:1306–1315.
Cortes J, Hochhaus A, Hughes T, Kantarjian H. Front-line and salvage therapies with tyrosine kinase inhibitors and other treatments in chronic myeloid leukemia. J Clin Oncol. 2011;29(5):524–531.
Druker BJ, Guilhot F, O’Brien SG, et al. Five-year follow-up of patients receiving imatinib for chronic myeloid leukemia. N Engl J Med. 2006;355:2408–2417.
Jabbour E, Cortes JE, Kantarjian HM. Molecular monitoring in chronic myeloid leukemia: response to tyrosine kinase inhibitors and prognostic implications. Cancer. 2008;112:2112–2118.
Jabbour E, Cortes JE, Giles FJ, O’Brien S, Kantarjian HM. Current and emerging treatment options in chronic myeloid leukemia. Cancer. 2007;109:2171–2181.
Khoury HJ, Cortes JE, Kantarjian HM, et al. Bosutinib in chronic phase CML after failure of imatinib, dasatinib, and/or nilotinib therapy. Blood. 2012;119(15):3403–3412.
Cheng F, Li Q, Cui Z, Hong M, Li W, Zhang Y. Dose optimization strategies for imatinib, dasatinib, and nilotinib in chronic myeloid leukemia: from clinical trials to real-world settings. Front Oncol. 2023;13:1146108.
Keywords:
chronic myeloid leukemia, chronic myeloid leukemia cml, cml philadelphia chromosome, bcr-abl leukemia, chronic myeloid leukemia symptoms, chronic myeloid leukemia diagnosis, chronic myeloid leukemia treatment, chronic myeloid leukemia prognosis, chronic myeloid leukemia icd 10, cml chronic phase, cml accelerated phase, cml blast phase, tyrosine kinase inhibitors cml, imatinib therapy cml, dasatinib nilotinib bosutinib ponatinib, cml molecular monitoring, cml pcr monitoring, cml bcr-abl transcript, cml bone marrow findings, cml blood smear, cml staging, chronic granulocytic leukemia, chronic myelogenous leukemia, cml treatment response, cml survival rate, cml life expectancy, cml tki side effects, imatinib skin rash itching, cml management guidelines, cml leukocytosis, cml basophilia, cml cytogenetics, philadelphia chromosome t(9;22)


Request Online Consultation With Dr M Abdou
Fee: US$100
Secure payment via PayPal (credit and debit cards accepted)
Pay Now


Good afternoon,
I’m a 4th year student at the University of West of Scotland, and I’m doing a project on Chronic Myeloid Leukaemia and I wondering if I could use your image above.
Regards
Hi Cecilia,
Thank you for your comment.
Please feel free to use the images you need but I would appreciate citing the reference.
Kind regards,
Please, I want to know the indications for bone marrow aspiration and biopsy in CML.
Hi Dr Chinedu,
Thank you for your comment.
The diagnosis of CML is based on the histopathologic findings in the peripheral blood and the Philadelphia chromosome in bone marrow cells. Findings from the workup—in particular, the percentage of blasts in peripheral blood or bone marrow—are used to determine the phase of CML: chronic, accelerated, or blast.
BW,