New Oral Anticoagulants
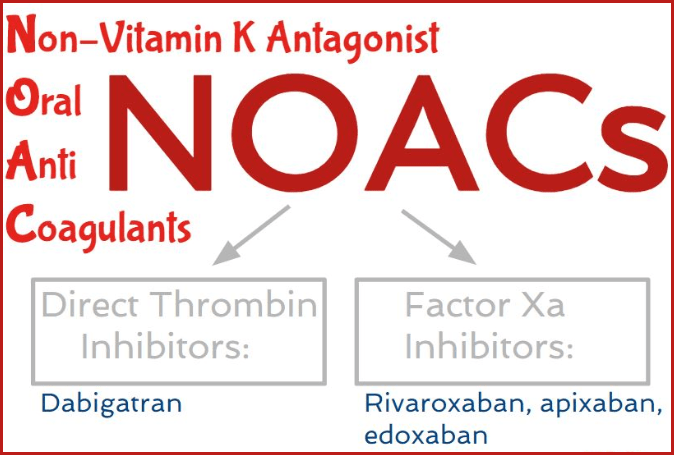
Classification of non-vitamin K antagonist oral anticoagulants (NOACs) according to their mechanism of action: dabigatran is a direct thrombin inhibitor, while rivaroxaban, apixaban, and edoxaban inhibit factor Xa.
The NOACs are new oral anticoagulants, or sometimes called non-vitamin K antagonist (VKA) oral anticoagulants (since they are no longer that new).
Oral anticoagulants are widely used for long-term prevention and treatment of venous and arterial thromboembolism. Until recently, vitamin K antagonists, such as warfarin, were the only available oral anticoagulants. This situation changed with the recent introduction of the non-vitamin K antagonist oral anticoagulants (NOACs), which include dabigatran, rivaroxaban, apixaban, and edoxaban.
Designed to overcome the limitations of warfarin, the NOACs have revolutionized oral anticoagulation because they are at least as effective as warfarin, but are more convenient to administer because the NOACs can be given in fixed doses without routine coagulation monitoring. Moreover, as a class, the NOACs are associated with significantly less intracranial bleeding than warfarin. This is an important advantage because bleeding into the brain is the most feared complication of anticoagulation therapy.
In the United States, rivaroxaban and apixaban are licensed for prevention of venous thromboembolism (VTE) after elective hip or knee replacement surgery and dabigatran, rivaroxaban, apixaban, and edoxaban are approved for treatment of VTE and for stroke prevention in patients with atrial fibrillation (AF). Although not approved in the United States for this indication, rivaroxaban is licensed in Europe for the prevention of recurrent ischemia in stabilized patients with acute coronary syndrome (ACS).
Comparison between Warfarin and NOACs
Warfarin inhibits vitamin K epoxide reductase, thereby attenuating the reduction of oxidized vitamin K in the liver. Without reduced vitamin K as a cofactor for hepatic γ-carboxylase, functional levels of the vitamin K–dependent clotting proteins, factors II, VII, IX, and X decrease. This results in attenuated thrombin generation regardless of whether clotting is triggered via the extrinsic, intrinsic, or common pathway of coagulation. Because of its indirect mechanism of action, the onset and offset of action of warfarin are delayed for several days, a phenomenon that often necessitates bridging with a rapidly acting parenteral anticoagulant when initiating warfarin therapy, and complicates periprocedural management.

Mechanism of action of major oral anticoagulants in the coagulation cascade. Warfarin interferes with vitamin K–dependent clotting factors, factor Xa inhibitors (rivaroxaban, apixaban, edoxaban) block factor Xa, and dabigatran directly inhibits thrombin. TF refers to tissue factor, a key initiator of the extrinsic coagulation pathway.
In contrast to warfarin, the NOACs directly inhibit a single clotting enzyme; dabigatran inhibits thrombin, whereas rivaroxaban, apixaban, and edoxaban inhibit factor Xa. As direct inhibitors, these agents have a rapid onset of action such that peak plasma levels are achieved 1 to 4 hours after oral administration. With half-lives of ≈12 hours, the NOACs also have a rapid offset of action.

Dabigatran etexilate (Pradaxa) capsules in three common strengths (75 mg, 110 mg, and 150 mg). Dabigatran is a direct thrombin inhibitor and one of the direct oral anticoagulants (DOACs) used for stroke prevention in atrial fibrillation and for the treatment and prevention of venous thromboembolism.

Apixaban (Eliquis) tablets in two common strengths (5 mg and 2.5 mg). Apixaban is a direct factor Xa inhibitor belonging to the class of direct oral anticoagulants (DOACs) used for stroke prevention in atrial fibrillation and for the treatment and prevention of venous thromboembolism.

Rivaroxaban (Xarelto) 20 mg film-coated tablets. Rivaroxaban is a direct factor Xa inhibitor and a widely used direct oral anticoagulant (DOAC) for stroke prevention in atrial fibrillation and for the treatment and prevention of venous thromboembolism.

Edoxaban (Lixiana) 60 mg film-coated tablets. Edoxaban is a direct factor Xa inhibitor and one of the direct oral anticoagulants (DOACs) used for stroke prevention in atrial fibrillation and for the treatment of venous thromboembolism.
Although warfarin is predominantly cleared through nonrenal mechanisms, the NOACs are excreted, at least in part, via the kidneys. The extent of renal clearance varies; ≈80% of absorbed dabigatran is cleared unchanged by the kidneys, whereas 50%, 33%, and 27% of absorbed edoxaban, rivaroxaban, and apixaban, respectively, are cleared unchanged via the renal route. Consequently, the drugs can accumulate in patients with renal impairment, thereby potentially placing them at risk for bleeding. To avoid this complication, NOACs should be used with caution in patients with a creatinine clearance <30 mL/min, and they should not be used if the creatinine clearance is <15 mL/min.
The dose of warfarin varies between patients reflecting differences in dietary vitamin K intake, multiple drug-drug interactions, and common polymorphisms that affect warfarin metabolism or pharmacodynamics. Warfarin has a narrow therapeutic window; thus, under anticoagulation can lead to recurrent thrombosis, whereas excessive anticoagulation can cause bleeding. Consequently, frequent coagulation monitoring and dose adjustments are necessary to ensure that the international normalized ratio (INR) remains within the therapeutic range. In contrast, because the NOACs produce a more predictable anticoagulant response, they can be given in fixed doses without routine monitoring, thereby simplifying therapy.
Although there are few clinically important drug-drug interactions with the NOACs, potent inhibitors or inducers of CYP 3A4 and p-glycoprotein can be problematic with rivaroxaban and apixaban, whereas potent inhibitors of p-glycoprotein may increase exposure with dabigatran and edoxaban. Dietary vitamin K intake does not influence the NOACs and there are no dietary restrictions except that therapeutic doses of rivaroxaban should be administered with a meal to maximize its absorption.
Indications and Doses for the NOACs:
The recommended doses for the NOACs for each approved indication are provided in the table. In general, the doses used for thromboprophylaxis are half those used for VTE treatment or for stroke prevention in AF. When used for stroke prevention, the doses of the NOACs are reduced based on important patient characteristics to maximize the benefit-to-risk profile.

Comparison of recommended dosing regimens for the major direct oral anticoagulants (DOACs)—dabigatran, rivaroxaban, apixaban, and edoxaban—in atrial fibrillation, treatment of venous thromboembolism, and thromboprophylaxis after hip or knee arthroplasty.
Reversal of Anticoagulation:
Vitamin K is the antidote for warfarin. When given orally or by slow intravenous infusion, vitamin K restores the INR to baseline levels, but this can take 12 to 24 hours. Rapid warfarin reversal can be achieved with 4-factor prothrombin complex concentrate (PCC). Fresh frozen plasma is an alternative to PCC, but it produces incomplete restoration of the INR to baseline levels, its infusion takes longer than administration of PCC and large volumes of plasma are often needed, which can be problematic for patients with compromised cardiopulmonary function. For these reasons, guidelines recommend PCC over fresh frozen plasma for patients who require urgent warfarin reversal.
There are no specific antidotes for the NOACs, but as outlined by Crowther et al, these are under development. Although nonactivated or activated PCC may be effective for reversal of the anticoagulant effects of the NOACs, clinical data in patients with serious bleeding are limited.
Andexxa (andexanet alfa) works by acting as a decoy for Factor Xa inhibitors in the blood, thereby preventing them from inhibiting the activity of native Factor Xa. As a result, the native Factor Xa is available to participate in the coagulation process and restore hemostasis (normal clotting).
Andexxa is specifically indicated for patients treated with rivaroxaban and apixaban when reversal of anticoagulation is needed due to life-threatening or uncontrolled bleeding.

Andexxa (andexanet alfa), a recombinant modified factor Xa protein used as a specific reversal agent for factor Xa inhibitors, including rivaroxaban and apixaban, in patients with life-threatening or uncontrolled bleeding.
Andexxa is supplied as a lyophilized powder for solution for intravenous injection. Dose Andexxa based on the specific FXa inhibitor, the dose of FXa inhibitor, and time since the patient’s last dose of FXa inhibitor. Administer as an intravenous (IV) bolus, with a target rate of 30 mg/min, followed by continuous infusion for up to 120 minutes. There are two dosing regimens: Low Dose: Initial IV Bolus: 400 mg at a target rate of 30 mg/min. Follow-On IV Infusion: 4mg/min for up to 120 minutes. High Dose: Initial IV Bolus: 800 mg at a target rate of 30 mg/min. Follow-On IV Infusion: 8mg/min for up to 120 minutes.
Idarucizumab is a humanized monoclonal antibody fragment developed as a specific reversal agent for dabigatran, a direct thrombin inhibitor. It binds dabigatran with very high affinity, neutralizing both free and thrombin-bound drug and rapidly reversing its anticoagulant effect. Idarucizumab is administered intravenously and is primarily used in emergency situations such as life-threatening bleeding or when urgent surgery or invasive procedures are required in patients receiving dabigatran. Clinical studies have demonstrated that idarucizumab can produce rapid and complete reversal of dabigatran anticoagulation within minutes, restoring normal hemostasis in most patients.
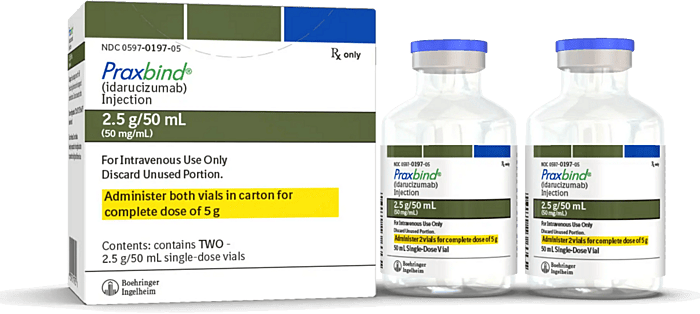
Praxbind (idarucizumab) injection, a specific reversal agent for dabigatran, used in emergency situations such as life-threatening bleeding or when urgent surgery is required in patients receiving dabigatran therapy.
Summary:
Direct oral anticoagulants (DOACs), previously known as non-vitamin K antagonist oral anticoagulants (NOACs), are modern oral medications used to prevent and treat thromboembolic disorders. Unlike warfarin, which interferes with vitamin K–dependent clotting factors (II, VII, IX, and X), DOACs act by directly inhibiting specific components of the coagulation cascade. Dabigatran is a direct thrombin inhibitor, while rivaroxaban, apixaban, and edoxaban are direct factor Xa inhibitors. These drugs are widely used for stroke prevention in non-valvular atrial fibrillation, treatment and prevention of venous thromboembolism (deep vein thrombosis and pulmonary embolism), and thromboprophylaxis after major orthopedic surgery. Compared with warfarin, DOACs have a rapid onset of action, predictable pharmacokinetics, fixed dosing, and generally do not require routine laboratory monitoring. They are also associated with a lower risk of intracranial hemorrhage. However, dose adjustments may be required in renal impairment, and clinicians must consider drug interactions and bleeding risks. Specific reversal agents are now available, including idarucizumab for dabigatran and andexanet alfa for factor Xa inhibitors. These advances have significantly improved the safety and clinical use of oral anticoagulation therapy.
Questions and Answers:
What are direct oral anticoagulants (DOACs)?
Direct oral anticoagulants (DOACs) are a group of oral blood-thinning medications that directly inhibit specific clotting factors in the coagulation cascade. They include dabigatran, rivaroxaban, apixaban, and edoxaban and are commonly used to prevent and treat thromboembolic diseases.
What is the difference between DOACs and warfarin?
Warfarin works indirectly by inhibiting vitamin K–dependent clotting factors, requiring regular INR monitoring and dietary restrictions. DOACs directly inhibit either thrombin or factor Xa, have more predictable effects, and usually do not require routine laboratory monitoring.
Which drugs belong to the DOAC or NOAC class?
The main drugs in this class are dabigatran (a direct thrombin inhibitor) and the factor Xa inhibitors rivaroxaban, apixaban, and edoxaban.
What conditions are treated with DOACs?
DOACs are commonly prescribed for stroke prevention in patients with non-valvular atrial fibrillation, treatment and secondary prevention of deep vein thrombosis (DVT) and pulmonary embolism (PE), and thromboprophylaxis after hip or knee replacement surgery.
When should dabigatran be chosen instead of factor Xa inhibitors such as rivaroxaban, apixaban, or edoxaban?
Dabigatran may be preferred in patients where the availability of a specific and rapidly acting reversal agent (idarucizumab) is considered particularly important, such as individuals at higher risk of bleeding or those who may require urgent procedures. Dabigatran is a direct thrombin inhibitor, whereas rivaroxaban, apixaban, and edoxaban inhibit factor Xa in the coagulation cascade. In clinical practice, the choice among these direct oral anticoagulants (DOACs) depends on several factors including renal function, dosing convenience, bleeding risk, potential drug interactions, and the availability of reversal strategies. For example, dabigatran has significant renal clearance and may be less suitable in severe renal impairment, while apixaban is often favored in patients with higher bleeding risk due to its favorable safety profile.
How do factor Xa inhibitors work?
Factor Xa inhibitors such as rivaroxaban, apixaban, and edoxaban block factor Xa in the coagulation cascade, preventing the generation of thrombin and the formation of fibrin clots.
What is factor X and why is factor Xa an important target for anticoagulant drugs?
Factor X (FX) is an inactive clotting protein, or zymogen, produced by the liver that plays a central role in the coagulation cascade. When activated, it becomes factor Xa, an enzyme that converts prothrombin into thrombin, a key step in the formation of fibrin blood clots. Because factor Xa sits at a critical point in the coagulation pathway, inhibiting it effectively reduces thrombin generation and prevents clot formation. For this reason, factor Xa inhibitors such as rivaroxaban, apixaban, and edoxaban are widely used as anticoagulant medications to prevent and treat conditions such as stroke in atrial fibrillation and venous thromboembolism.
How does dabigatran work?
Dabigatran directly inhibits thrombin (factor IIa), preventing the conversion of fibrinogen to fibrin and therefore inhibiting clot formation.
Do DOACs require blood monitoring like warfarin?
Routine coagulation monitoring is generally not required for DOACs because they have predictable pharmacokinetics and fixed dosing.
Are reversal agents available for DOACs?
Yes. Idarucizumab is used to reverse dabigatran, while andexanet alfa can reverse the anticoagulant effects of factor Xa inhibitors such as rivaroxaban and apixaban.
Are DOACs safe in kidney disease?
Renal function must be considered when prescribing DOACs because several of these drugs are partially eliminated through the kidneys. Dose adjustments or avoidance may be necessary in patients with significant renal impairment.
Why are DOACs increasingly preferred over warfarin?
DOACs offer several advantages including rapid onset of action, fewer drug and dietary interactions, predictable dosing, and a lower risk of intracranial bleeding compared with warfarin.
References:
Yeh CH, Hogg K, Weitz JI. Overview of the new oral anticoagulants. Arterioscler Thromb Vasc Biol. 2015;35(5):1055–1065. doi:10.1161/ATVBAHA.115.303397. https://www.ahajournals.org/doi/10.1161/ATVBAHA.115.303397
On K, Hunter LA, Hunter RJ. Ten top tips – NOACs. Pulse Today. https://www.pulsetoday.co.uk/clinical/clinical-specialties/cardiovascular/ten-top-tips-noacs/20034760.article
Messerschmidt C, Friedman RJ. Clinical experience with novel oral anticoagulants for thromboprophylaxis after elective hip and knee arthroplasty. Arterioscler Thromb Vasc Biol. 2015;35(3):771–778. doi:10.1161/ATVBAHA.115.305081
Bacchus F, Schulman S. Clinical experience with the new oral anticoagulants for treatment of venous thromboembolism. Arterioscler Thromb Vasc Biol. 2015;35(3):513–519. doi:10.1161/ATVBAHA.115.305084
Carreras ET, Mega JL. Role of oral anticoagulants in patients after an acute coronary syndrome. Arterioscler Thromb Vasc Biol. 2015;35(3):520–524. doi:10.1161/ATVBAHA.115.305087
Patel MR, Mahaffey KW, Garg J, et al. Rivaroxaban versus warfarin in nonvalvular atrial fibrillation (ROCKET-AF trial). N Engl J Med. 2011;365:883–891. doi:10.1056/NEJMoa1009638
Connolly SJ, Ezekowitz MD, Yusuf S, et al. Dabigatran versus warfarin in patients with atrial fibrillation (RE-LY trial). N Engl J Med. 2009;361(12):1139–1151. doi:10.1056/NEJMoa0905561
Granger CB, Alexander JH, McMurray JJV, et al. Apixaban versus warfarin in patients with atrial fibrillation (ARISTOTLE trial). N Engl J Med. 2011;365:981–992. doi:10.1056/NEJMoa1107039
Giugliano RP, Ruff CT, Braunwald E, et al. Edoxaban versus warfarin in patients with atrial fibrillation (ENGAGE AF-TIMI 48 trial). N Engl J Med. 2013;369:2093–2104. doi:10.1056/NEJMoa1310907
Cuker A, Burnett A, Triller D, et al. Reversal of direct oral anticoagulants: Guidance from the Anticoagulation Forum. Am J Hematol. 2019;94(6):697–709. doi:10.1002/ajh.25475
Ruff CT, Giugliano RP, Braunwald E, et al. Comparison of the efficacy and safety of new oral anticoagulants with warfarin in patients with atrial fibrillation: A meta-analysis of randomized trials. Lancet. 2014;383(9921):955–962. doi:10.1016/S0140-6736(13)62343-0
Weitz JI, Pollack CV Jr. Practical management of bleeding in patients receiving direct oral anticoagulants. Circulation. 2017;135(22):2076–2099. doi:10.1161/CIRCULATIONAHA.116.026080
StatPearls Publishing. Direct oral anticoagulants. StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK470168/
U.S. Food and Drug Administration (FDA). Direct oral anticoagulants (DOACs) safety communications. https://www.fda.gov/drugs/drug-safety-and-availability/direct-oral-anticoagulants-doacs-safety-communication
European Medicines Agency (EMA). Direct oral anticoagulants (NOACs) – summary of product characteristics. https://www.ema.europa.eu/en/medicines/human
Weitz JI, Fredenburgh JC. Factors influencing the choice of oral anticoagulant therapy. Blood. 2020;135(5):351–359. doi:10.1182/blood.2019000940
Keywords:
direct oral anticoagulants, DOACs, NOACs, new oral anticoagulants, oral anticoagulants, anticoagulation therapy, anticoagulants in hematology, DOAC mechanism of action, factor Xa inhibitors, direct thrombin inhibitor, factor Xa inhibition, dabigatran, rivaroxaban, apixaban, edoxaban, Pradaxa, Xarelto, Eliquis, Lixiana, DOAC vs warfarin, oral anticoagulants vs warfarin, DOAC advantages, DOAC bleeding risk, DOAC reversal agents, idarucizumab, andexanet alfa, anticoagulant reversal, stroke prevention atrial fibrillation, atrial fibrillation anticoagulation, DOAC stroke prevention, anticoagulants for atrial fibrillation, venous thromboembolism treatment, deep vein thrombosis treatment, pulmonary embolism treatment, DOAC venous thromboembolism, VTE treatment anticoagulants, thromboprophylaxis hip knee surgery, orthopedic thromboprophylaxis, coagulation cascade anticoagulants, factor Xa role in coagulation, thrombin inhibition anticoagulants, anticoagulation monitoring DOACs, DOAC renal impairment dosing, DOAC safety profile, anticoagulant drug comparison, modern anticoagulants hematology, oral anticoagulant dosing chart, DOAC clinical trials, RE-LY trial dabigatran, ROCKET AF trial rivaroxaban, ARISTOTLE trial apixaban, ENGAGE AF TIMI 48 trial edoxaban

Request Online Consultation With Dr M Abdou
Fee: US$100
Secure payment via PayPal (credit and debit cards accepted)
Pay Now