Artificial Intelligence in Hematology

Artificial intelligence is transforming hematology by enhancing diagnostic precision, data analysis, and personalized treatment strategies.
Introduction:
Artificial intelligence in hematology is rapidly transforming the diagnosis and management of blood disorders. By integrating machine learning and advanced data analytics into clinical practice, artificial intelligence (AI) enables earlier diagnosis, improves diagnostic accuracy, and supports personalized treatment strategies.
Hematology, the medical specialty focused on the study, diagnosis, and treatment of blood diseases, has traditionally relied on microscopic examination and laboratory testing. However, the emergence of artificial intelligence, particularly in areas such as digital pathology and pattern recognition, is revolutionizing hematology practice by enhancing data interpretation, reducing variability, and improving clinical decision-making.
This article explores the current applications, clinical benefits, and future potential of artificial intelligence in hematology.
Artificial Intelligence in Peripheral Blood Smear Analysis:
Peripheral blood smear examination remains a fundamental step in the evaluation of hematological disorders, serving as an essential confirmatory tool following automated complete blood count analysis. Despite its diagnostic value, manual microscopic assessment is time-consuming, operator-dependent, and requires significant laboratory expertise, often resulting in a turnaround time of approximately 3–4 hours in routine clinical settings.
To overcome these limitations, automated and computer-assisted approaches to peripheral smear analysis have been increasingly adopted, although accurate cell detection and classification remain technically challenging. Recent advances in artificial intelligence, particularly deep learning, have demonstrated significant potential in improving the accuracy, efficiency, and reproducibility of peripheral blood smear interpretation. These technologies enable rapid identification and classification of blood cells, reducing diagnostic variability and operational costs. However, their performance and generalizability are highly dependent on the availability of large, well-annotated datasets, which remain a critical limiting factor in further development and clinical implementation.

Example of AI-based annotations in a color image (top row) and a monochromatic multichannel stack (bottom row), including transmission, DAPI, and red fluorescence channels, with cells labeled according to pseudo-color classification.
Artificial Intelligence and Integrated Diagnostics in Hematology:
Artificial intelligence algorithms are capable of analyzing large and complex datasets, including electronic health records, laboratory parameters, genomic data, and medical imaging, to detect subtle patterns and abnormalities associated with hematological disorders. This data-driven approach enhances diagnostic precision, facilitates earlier disease detection, and supports risk stratification. By integrating multidimensional data, AI has the potential to improve clinical decision-making and ultimately optimize patient outcomes in hematology.
Over the past 15 years, the diagnostic evaluation of leukemia and lymphoma has become increasingly complex and multidimensional. Accurate classification now requires the integration of data from multiple complementary disciplines, including cytomorphology, immunophenotyping, cytogenetics, and molecular genetics, in line with the evolving frameworks of the World Health Organization (WHO) classification. The advent of next-generation sequencing (NGS) has significantly expanded the scope and depth of molecular diagnostics, enabling high-throughput detection of genetic alterations and refining disease characterization. This growing reliance on molecular data has been reflected in successive updates to the WHO classification, including the 2017 revision and subsequent refinements, which emphasize genomically defined disease entities and underscore the central role of integrated diagnostics in modern hematopathology.
The expanding volume of molecular data has significantly reshaped the diagnostic landscape of leukemia and lymphoma, providing deeper insights into disease biology and driving a paradigm shift from phenotype-based to genotype-driven classification. The identification of an increasing number of diagnostic and prognostic biomarkers, together with a more precise characterization of inter-individual variability, has enhanced risk stratification and disease subclassification. Furthermore, ongoing efforts to integrate multi-layered data, including genomic, immunophenotypic, and clinical information are facilitating the development of more effective targeted therapies. Collectively, these advances are accelerating the transition toward precision medicine in hematologic malignancies.
The ongoing digital transformation of healthcare has enabled the seamless integration of diverse diagnostic data, including clinical, laboratory, and genomic information, into unified digital platforms. This facilitates efficient data sharing across multidisciplinary teams and institutions, enhancing collaborative decision-making and enabling more precise patient stratification for clinical trials and targeted therapies. Importantly, this transition from traditional, fragmented workflows to integrated, data-driven systems reinforces the shift from phenotype-based assessment to genotype-informed precision medicine, providing a critical foundation for the effective implementation of artificial intelligence in hematology.
Artificial Intelligence, Machine Learning, and Deep Learning in Hematology:
Digital data infrastructure is a fundamental prerequisite for the effective application of emerging artificial intelligence (AI) technologies in hematology. Leveraging machine learning (ML) and deep learning (DL) algorithms, AI is increasingly being integrated across biomedical disciplines and holds significant potential to transform diagnostic workflows. Recent advances in computational performance and data availability have accelerated the development of AI-driven tools, bringing them closer to routine clinical implementation. Beyond enhancing diagnostic accuracy, these technologies are poised to improve disease classification, enable more precise risk stratification, and support individualized treatment decision-making in hematologic disorders.

Relationship between artificial intelligence, machine learning, and deep learning, illustrating how advanced algorithms support data-driven diagnostics in hematology.

Key differences between machine learning and deep learning, highlighting data requirements, computational needs, and feature extraction in medical AI applications.
With the rapid advancement and widespread adoption of AI-driven technologies, the terms artificial intelligence (AI) and machine learning (ML) are increasingly used across scientific disciplines, and are often used interchangeably in medical contexts. However, these concepts represent distinct, though related, domains. Artificial intelligence broadly refers to the ability of machines to simulate human cognitive functions and decision-making processes. Machine learning, a subset of AI, involves the development of algorithms that automatically identify patterns and relationships within data without explicit programming. Deep learning (DL), a further subset of ML, employs multi-layered neural networks to learn hierarchical and abstract representations of highly complex datasets, enabling advanced tasks such as image recognition, pattern detection, and predictive modeling in hematology.

Overview of artificial intelligence, machine learning, and deep learning domains (left) alongside the supervised learning workflow (right), including training data requirements, feature extraction, model evaluation, and iterative retraining.
One of the most promising emerging applications of artificial intelligence is the development of digital twins: virtual, data-driven representations of individual patients that can model disease progression and simulate cancer trajectories. These in silico models enable the prediction of responses to various therapeutic strategies, allowing clinicians to compare potential outcomes before initiating treatment. Such an approach has the potential to optimize individualized therapy selection, reduce unnecessary toxicity, and minimize the risk of complications, thereby advancing precision oncology in hematology.
AI in Hematologic Diagnostics:
Automated Blood Smear Analysis
Automated analysis of peripheral blood smears represents one of the most advanced and clinically relevant applications of artificial intelligence in hematology. Traditional manual microscopy, although essential, is time-consuming, operator-dependent, and subject to inter-observer variability.
Artificial intelligence, particularly deep learning approaches such as convolutional neural networks (CNNs), enables high-precision analysis of blood smear images. These systems can accurately identify and classify blood cells, including erythrocytes, leukocyte subtypes, and platelets, while simultaneously detecting morphological abnormalities and rare pathological patterns.
AI-driven image analysis has demonstrated the ability to streamline cell counting and morphological assessment, reducing workload and improving consistency in diagnostic reporting. In selected applications, including the detection of hematologic malignancies and infectious diseases such as malaria, AI models have achieved diagnostic performance comparable to, and in some cases exceeding, that of experienced hematologists.
Advanced models have been developed using both conventional color images and multi-channel imaging approaches, including transmission, DAPI, and red fluorescence channels. These complementary modalities enable more robust feature extraction and improve classification accuracy. The use of well-annotated datasets across these imaging formats remains essential for optimizing model performance and generalizability.

Example of AI-based automatic detection of blood cells in color images (first and third rows) and TDR stacks (second and fourth rows). Rainbow-colored regions with white borders represent segmented cells identified by the algorithm, while red areas without borders indicate background fluorescence from Giemsa staining.
Flow Cytometry Data Interpretation
Flow cytometry is a cornerstone diagnostic tool in hematology, particularly in the evaluation of hematologic malignancies and immunological disorders. Conventional analysis relies on expert interpretation of high-dimensional data, including manual gating strategies performed by experienced hematopathologists, which can be time-consuming and subject to inter-operator variability.
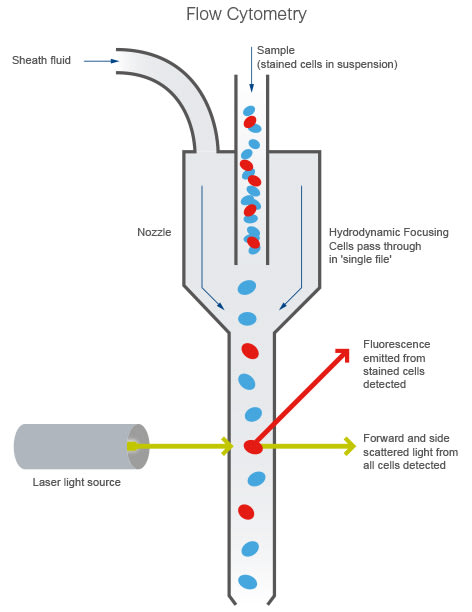
Principle of flow cytometry demonstrating hydrodynamic focusing, laser interrogation, and detection of fluorescence and scattered light signals from individual cells.
Advances in artificial intelligence are increasingly being applied to flow cytometry, enabling automated analysis of complex datasets with improved efficiency, consistency, and scalability. AI-driven approaches can assist in case prioritization, reduce analytical errors, and uncover previously unrecognized biological patterns, thereby enhancing diagnostic accuracy and clinical insight.
Recent studies have demonstrated the clinical utility of AI-assisted flow cytometry workflows. In one representative study involving 379 clinical cases, an automated AI platform (DeepFlow™, version 2.1.1) analyzed data generated from a standardized 3-tube, 10-color panel incorporating 21 antibodies. The system employs multidimensional density–phenotype coupling algorithms to accurately classify and quantify lymphocyte populations, including T, B, and NK cells, as well as key subsets such as CD4+ and CD8+ T cells, double-negative T cells, and class-switched B cells.
The AI-assisted workflow reduced analysis time to under 5 minutes per case while maintaining the ability for expert oversight through optional manual gating adjustments. Importantly, the model demonstrated excellent agreement with conventional hematopathologist assessment, with strong correlations (r > 0.9) across lymphocyte subsets. These findings highlight the potential of AI to streamline flow cytometry interpretation, improve reproducibility, and support clinical decision-making in hematology.

Comparison of multiparameter flow cytometry plots generated by Kaluza™ and AI-based DeepFlow™ for normal T cell, B cell, and NK cell subsets, demonstrating comparable immunophenotypic classification.
AI in Hematologic Therapeutics:
Predictive Analytics for Treatment Response
Artificial intelligence is increasingly being applied to predict disease risk and therapeutic response in hematology by integrating genomic, clinical, and laboratory data. AI-driven predictive models can identify patients at higher risk of developing hematologic disorders and enable earlier intervention and personalized management strategies.
In established disease, these models support precision medicine by forecasting individual responses to specific therapies. For example, in leukemia, AI-based tools can predict response to chemotherapy or targeted treatments, allowing clinicians to optimize therapeutic selection, maximize efficacy, and minimize treatment-related toxicity. This data-driven approach enhances risk stratification and supports more individualized, outcome-focused care.
Drug Discovery and Development
Artificial intelligence is transforming drug discovery in hematology by accelerating the identification of novel therapeutic targets and optimizing drug development pipelines. AI-driven platforms can analyze complex molecular and genomic datasets to uncover disease mechanisms and predict the efficacy and safety of candidate compounds.
In hematologic malignancies such as lymphoma and multiple myeloma, these approaches have contributed to the development of targeted therapies and improved understanding of disease biology. AI-based in silico modeling further enables simulation of drug interactions and therapeutic responses, reducing development time and supporting more efficient clinical translation.
AI-Enhanced Clinical Decision-Making
Artificial intelligence can augment clinical decision-making by integrating multidimensional patient data, including laboratory results, molecular profiles, imaging findings, and treatment outcomes. These systems provide evidence-based insights that assist hematologists in selecting appropriate diagnostic tests, guiding therapeutic interventions, and optimizing disease management strategies.
By supporting data-driven decisions and reducing variability in clinical practice, AI has the potential to improve treatment consistency, enhance patient outcomes, and streamline care delivery in hematology.
Enhancing Hematology Workflows:
Laboratory Automation
Artificial intelligence–driven automation is increasingly transforming hematology laboratory workflows, improving efficiency, standardization, and scalability. From pre-analytical sample handling to analytical processes and post-analytical result interpretation, AI-integrated systems can reduce turnaround times and minimize operator-dependent variability.
Automated platforms for routine investigations, including complete blood count (CBC) analysis and digital morphology, are now widely implemented. These systems enable rapid, high-throughput processing while maintaining accuracy and consistency, allowing hematologists and laboratory specialists to focus on complex diagnostic interpretation and clinical decision-making.
Telemedicine and Remote Diagnostics
Artificial intelligence is also facilitating the expansion of telemedicine and remote diagnostics in hematology, improving access to specialist care in geographically remote and resource-limited settings. AI-enabled platforms can analyze laboratory and clinical data in real time, support remote interpretation of diagnostic tests, and assist in triaging patients based on clinical urgency.
This approach is particularly valuable for patients with chronic hematologic conditions requiring ongoing monitoring, such as anemia, hemoglobinopathies, and hematologic malignancies. By enabling virtual consultations and data-driven follow-up, AI-supported telemedicine can enhance continuity of care, reduce the need for in-person visits, and improve overall healthcare delivery.
Machine Learning Applications Across Hematology Diagnostics and Clinical Practice:
Cytomorphology and cytogenetics currently represent the most promising domains for the rapid integration of machine learning–based technologies into routine hematology workflows. The incorporation of deep learning methods has significantly enhanced diagnostic performance, with several applications now approaching expert-level accuracy. These advances enable faster, more consistent sample evaluation and extend specialist-level diagnostic support to less experienced clinicians, thereby improving accessibility and efficiency in hematologic diagnostics.
In contrast, the application of artificial intelligence in flow cytometry remains at an earlier stage of clinical adoption. While progress has been substantial, performance has not yet reached uniformly acceptable standards across all use cases. Nevertheless, ongoing developments in algorithm design and data integration are expected to facilitate the implementation of automated workflows, improving standardization, reproducibility, and scalability in flow cytometry analysis.

Overview of implemented and emerging machine learning applications in hematology, highlighting diagnostic methods, multimodal data integration, and their impact on diagnosis, prognosis, and targeted treatment.
Challenges and Ethical Considerations:
Despite the significant potential of artificial intelligence in hematology, several practical, regulatory, and ethical challenges must be carefully addressed to ensure safe and effective clinical implementation. Data privacy and security remain critical concerns, particularly given the sensitive nature of patient information and the increasing use of large, multi-institutional datasets required for AI model development.
Robust clinical validation and regulatory oversight are essential before widespread adoption, to ensure that AI systems demonstrate reliability, generalizability, and clinical utility across diverse patient populations. In addition, issues related to algorithm transparency and interpretability are increasingly important, as many AI models function as “black boxes,” potentially limiting clinician trust and accountability in decision-making.
Bias in training datasets represents another key challenge, as unrepresentative data may lead to inequitable performance across different patient groups, thereby reinforcing existing healthcare disparities. Addressing these limitations requires the development of standardized datasets, continuous model evaluation, and multidisciplinary collaboration between clinicians, data scientists, and regulatory bodies.
Ensuring ethical deployment of AI in hematology will depend on maintaining patient confidentiality, promoting transparency, and integrating human oversight to support, rather than replace, clinical judgment.
Summary:
Artificial intelligence in hematology is rapidly transforming the diagnosis, classification, and management of blood disorders. By integrating machine learning and deep learning with clinical, laboratory, and genomic data, AI enhances diagnostic accuracy, enables early disease detection, and supports precision medicine.
Applications such as automated peripheral blood smear analysis and AI-assisted flow cytometry allow rapid, reproducible identification of blood cell populations, reducing reliance on manual interpretation and minimizing inter-observer variability. In hematologic malignancies, AI facilitates the integration of cytomorphology, immunophenotyping, cytogenetics, and molecular data, aligning with modern WHO classification systems and advancing genotype-driven diagnostics.
Beyond diagnostics, AI is reshaping hematologic therapeutics through predictive analytics, enabling personalized treatment selection and improving outcomes. AI-driven drug discovery and digital twin technologies further expand the potential for targeted therapy and in silico modeling of disease progression.
In addition, AI enhances laboratory workflows through automation and supports telemedicine by enabling remote diagnostics and monitoring. Despite these advances, challenges such as data privacy, algorithm transparency, regulatory validation, and bias must be addressed to ensure safe and equitable implementation.
Overall, artificial intelligence is emerging as a cornerstone of modern hematology, bridging diagnostics, therapeutics, and clinical decision-making to deliver more precise, efficient, and patient-centered care.
Questions and Answers:
What is artificial intelligence in hematology?
Artificial intelligence in hematology refers to the use of machine learning and deep learning algorithms to analyze clinical, laboratory, and genomic data to improve diagnosis, classification, and management of blood disorders.
How is AI used in hematology diagnostics?
AI is used in hematology diagnostics to analyze peripheral blood smears, interpret flow cytometry data, and integrate molecular and genomic information. These technologies improve diagnostic accuracy, reduce variability, and enable faster decision-making.
Can artificial intelligence detect blood disorders?
Yes, AI can detect blood disorders by identifying abnormal patterns in blood cells, laboratory results, and genetic data. In some applications, AI systems can achieve accuracy comparable to experienced hematologists.
What is the role of AI in flow cytometry?
AI enhances flow cytometry by automating data analysis, identifying cell populations, and improving classification of immune cell subsets. This leads to faster, more reproducible, and standardized results.
How does AI improve peripheral blood smear analysis?
AI improves peripheral blood smear analysis by using deep learning models to automatically detect and classify blood cells, identify abnormalities, and reduce reliance on manual microscopy.
What is the difference between machine learning and deep learning in hematology?
Machine learning involves algorithms that detect patterns in structured data, while deep learning uses neural networks to analyze complex data such as images and genomic information. Deep learning is particularly effective in image-based diagnostics like blood smear analysis.
How is AI used in personalized medicine in hematology?
AI supports personalized medicine by predicting treatment response, stratifying patient risk, and guiding targeted therapy selection based on individual clinical and genetic profiles.
What are digital twins in hematology?
Digital twins are virtual models of patients that simulate disease progression and treatment response. They allow clinicians to test different therapeutic strategies in silico before applying them in real clinical settings.
How does AI improve hematology laboratory workflows?
AI improves workflows by automating routine laboratory tasks, reducing turnaround time, minimizing errors, and enabling high-throughput data analysis.
What are the challenges of artificial intelligence in hematology?
Key challenges include data privacy concerns, algorithm transparency, potential bias in training data, and the need for rigorous clinical validation and regulatory approval.
References:
WALTER W, HAFERLACH C, NADARAJAH N, et al. How artificial intelligence might disrupt diagnostics in hematology in the near future. Oncogene. 2021;40:4271–4280. https://doi.org/10.1038/s41388-021-01861-y
https://www.nature.com/articles/s41388-021-01861-y
SWERDLOW SH, CAMPO E, HARRIS NL, JAFFE ES, PILERI SA, STEIN H, et al. WHO classification of tumours of haematopoietic and lymphoid tissues. Revised 4th ed. Lyon: International Agency for Research on Cancer; 2017.
MADHAVAN S, BECKMAN RA, MCCOY MD, PISHVAIAN MJ, BRODY JR, MACKLIN P. Envisioning the future of precision oncology trials. Nat Cancer. 2021;2:9–11.
BJÖRNSSON B, BORREBAECK C, ELANDER N, GASSLANDER T, GAWEL DR, GUSTAFSSON M, et al. Digital twins to personalize medicine. Genome Med. 2019;12:4.
FJELLAND R. Why general artificial intelligence will not be realized. Humanit Soc Sci Commun. 2020;7:10.
CAMPOS-MEDINA M, BLUMER A, KRAUS-FÜREDER P, MAYRHOFER-REINHARTSHUBER M, KAINZ P, SCHMID JA. AI-enhanced blood cell recognition and analysis: advancing traditional microscopy with the web-based platform IKOSA. J Mol Pathol. 2024;5:28–44. https://doi.org/10.3390/jmp5010003
SHAMS UA, JAVED I, FIZAN M, SHAH AR, MUSTAFA G, ZUBAIR M, et al. Bio-net dataset: AI-based diagnostic solutions using peripheral blood smear images. Blood Cells Mol Dis. 2024;105:102823. https://doi.org/10.1016/j.bcmd.2024.102823
NG DP, SIMONSON PD, TARNOK A, LUCAS F, KERN W, ROLF N, et al. Recommendations for using artificial intelligence in clinical flow cytometry. Cytometry B Clin Cytom. Available at: https://onlinelibrary.wiley.com/doi/10.1002/cyto.b.22166
LU Z, MORITA M, YEAGER TS, LYU Y, WANG SY, WANG Z, et al. Validation of artificial intelligence-assisted flow cytometry analysis for immunological disorders. Diagnostics. 2024;14:420. https://doi.org/10.3390/diagnostics14040420
SMITH J, et al. Deep learning for automated blood smear analysis. J Hematol Oncol. 2022;15:345–357.
JOHNSON L, et al. AI in hematologic diagnosis: current applications and future directions. Blood Adv. 2021;5:123–134.
KUMAR R, et al. Machine learning in flow cytometry: enhancing hematologic diagnostics. Clin Cytometry. 2020;72:567–578.
DAVIS T, et al. Predictive analytics in leukemia treatment: an AI approach. Hematol Rep. 2019;11:98–107.
GARCIA M, et al. AI in drug discovery for hematologic malignancies. Future Oncol. 2023;19:12–25.
LEE S, et al. Automation in hematology laboratories: AI-driven advances. Lab Med. 2021;52:456–467.
PATEL N, et al. Telemedicine and AI in hematology: bridging the gap. Telemed J E Health. 2020;26:789–798.
RODRIGUEZ H, et al. Ethical considerations in AI-driven hematology. J Med Ethics. 2019;45:203–210.
GARZON DC. What is artificial intelligence (AI) and why people should learn about it. Available at: https://incubator.ucf.edu/what-is-artificial-intelligence-ai-and-why-people-should-learn-about-it/
SAUMYAB. Machine learning vs deep learning vs artificial intelligence. Available at: https://www.analyticsvidhya.com/blog/2021/06/machine-learning-vs-artificial-intelligence-vs-deep-learning/
REGUNATH G. Understanding the difference between AI, ML, and DL. Available at: https://www.advancinganalytics.co.uk/blog/2021/12/15/understanding-the-difference-between-ai-ml-and-dl-using-an-incredibly-simple-example/
Keywords:
artificial intelligence in hematology, AI in hematology, machine learning in hematology, deep learning in hematology, AI in hematology diagnostics, AI in blood smear analysis, automated blood smear AI, AI in leukemia diagnosis, AI in lymphoma diagnosis, AI in hematologic malignancies, artificial intelligence blood cancer diagnosis, AI flow cytometry hematology, AI in immunophenotyping, AI in hematology clinical decision support, AI in hematology precision medicine, predictive analytics hematology AI, AI treatment response leukemia, digital pathology hematology AI, AI in cytomorphology, AI in cytogenetics, AI in molecular hematology, multi-omics hematology AI, hematology big data AI, AI in blood disorder diagnostics, AI laboratory automation hematology, AI telemedicine hematology, AI drug discovery hematology, digital twins hematology, AI workflow hematology, AI healthcare diagnostics, AI clinical hematology applications, AI hematology review


Request Online Consultation With Dr M Abdou
Fee: US$100
Secure payment via PayPal (credit and debit cards accepted)
Pay Now

This is a concise, beautifully layed out explanation and overall summary of all that Haematology has achieved and continues to improve over the years.
Thank you so much.
Hello Dr L. Mazwi,
I’m glad to hear that you found the article helpful.
Best wishes,
Dr. M. Abdou
Great article! AI product manager interview questions like these are very helpful for understanding the blend of technical knowledge and product thinking required for the role. Topics around machine learning basics, data strategy, and user impact are especially valuable for preparation.
Hello Vinod,
I’m glad to hear that you found the article helpful.
Best wishes,
Dr. M. Abdou