Vascular Bleeding Disorders

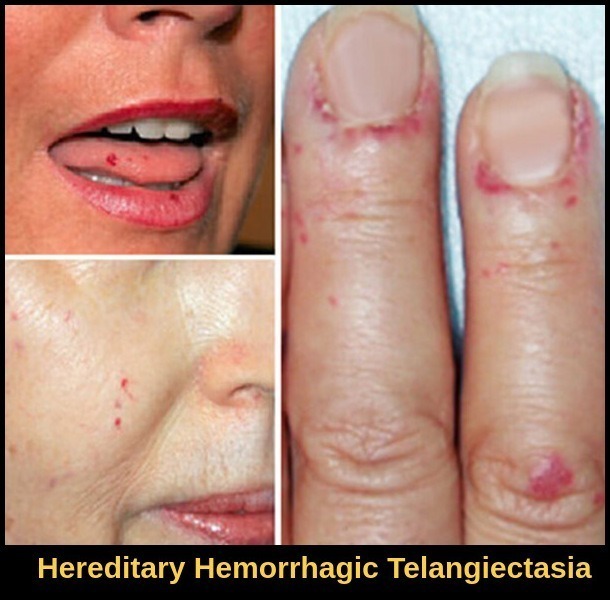
Hereditary hemorrhagic telangiectasia (HHT) showing characteristic mucocutaneous telangiectases involving the lips, oral mucosa, nasal mucosa, and skin, a classic cause of vascular bleeding disorders and recurrent epistaxis.
Vascular bleeding disorders are a group of conditions in which structural abnormalities of the blood vessel wall result in bleeding manifestations such as easy bruising, petechiae, purpura, and mucocutaneous bleeding. In contrast to platelet or coagulation factor disorders, major or life-threatening hemorrhage is uncommon, with the notable exception of hereditary hemorrhagic telangiectasia (HHT). These disorders arise from inherited or acquired defects affecting vascular integrity or perivascular connective tissue, leading to increased vessel fragility and bleeding in response to minimal trauma. Inherited causes include connective tissue disorders such as Ehlers–Danlos syndrome, pseudoxanthoma elasticum, osteogenesis imperfecta, Marfan syndrome, and other rare conditions characterized by abnormal collagen or elastic fiber synthesis. Acquired vascular bleeding disorders may result from vitamin C deficiency (scurvy), immunoglobulin A–associated vasculitis (Henoch–Schönlein purpura), inflammatory small-vessel vasculitis, corticosteroid exposure, or age-related vascular fragility.
From a hematologic perspective, abnormal bleeding can broadly result from defects in platelet number or function, coagulation factors, or blood vessels, with vascular bleeding disorders representing an important but often under-recognized category. A clear understanding of their pathophysiology, clinical presentation, differential diagnosis, and management is essential to avoid unnecessary investigations, distinguish them from primary hemostatic disorders, and ensure optimal patient care.
Hereditary Hemorrhagic Telangiectasia
(Osler-Weber-Rendu Syndrome)
Hereditary hemorrhagic telangiectasia (HHT) is a hereditary disorder of vascular malformation transmitted as an autosomal dominant trait affecting men and women.
HHT is a genetic disorder. Each person with HHT has one gene that is altered (mutated), which causes HHT, as well as one normal gene. It takes only one mutant gene to cause HHT. When someone with HHT has children, each child has a 50% chance to receive the mutant gene from his/her parent, and therefore to have HHT, as well. Each child also has a 50% chance to receive the normal gene and not be affected with HHT. At least five different genes can cause HHT, three of which are known.
More than 80% of patients have mutations in one of the following genes (1):
- Endoglin (ENG) gene, which encodes a receptor for transforming growth factor beta-1 (TGF-β1) and transforming growth factor beta-3
- ACVRL1 gene, which encodes the activin receptor-like kinase (ALK1)
- MADH4 gene, which encodes SMAD4, a protein active in the TGF beta signaling pathway
HHT is a disorder in which some blood vessels do not develop properly. A person with HHT may form blood vessels without the capillaries (tiny blood vessels that pass blood from arteries to veins) that are usually present between arteries and veins. The space between an artery and a vein is often fragile and can burst and bleed much more easily than other blood vessels. Men, women, and children from all racial and ethnic groups can be affected by HHT and experience the problems associated with this disorder, some of which are serious and potentially life-threatening. Fortunately, if HHT is discovered early, effective treatments are available. However, there is no cure for HHT.
Symptoms and Signs of HHT:
The most characteristic lesions of hereditary hemorrhagic telangiectasia are small red-to-violet telangiectatic lesions on the face, lips, oral and nasal mucosa, and tips of the fingers and toes. Similar lesions may be present throughout the mucosa of the gastrointestinal (GI) tract, resulting in recurrent GI bleeding. Patients may experience recurrent, profuse nosebleeds. Some patients have pulmonary arteriovenous malformations (AVMs). These AVMs may cause significant right-to-left shunts, which can result in dyspnea, fatigue, cyanosis, or erythrocytosis. However, the first sign of the presence of AVMs may be a brain abscess, transient ischemic attack, or stroke as a result of infected or noninfected emboli. Cerebral or spinal AVMs occur in some families and may cause subarachnoid hemorrhage, seizures, or paraplegia. Hepatic AVMs may lead to liver failure and high output heart failure. Chronic iron deficiency anemia is commonly present.
Nosebleeds are the most common sign of HHT, resulting from small abnormal blood vessels within the inside layer of the nose. Abnormal blood vessels in the skin can appear on the hands, fingertips, face, lips, lining of the mouth, and nose as delicate red or purplish spots that lighten briefly when touched. Bleeding within the stomach or intestines is another possible indicator of HHT that occurs because of abnormal blood vessels lining the digestive tract. Additional signs of HHT include abnormal artery-vein connections within the brain, lungs, and liver, which often do not display any warning signs before rupturing.
Hereditary hemorrhagic telangiectasia (HHT) demonstrating characteristic telangiectases involving the lips, perioral skin, nailfolds, and fingers, a hallmark of vascular bleeding disorders and recurrent mucocutaneous bleeding.
Diagnosis of HHT:
- Clinical evaluation
- Sometimes endoscopy or angiography
- Sometimes genetic testing
Diagnosis of hereditary hemorrhagic telangiectasia is based on the finding of characteristic arteriovenous malformations on the face, mouth, nose, digits, and/or internal organs in the context of epistaxis and family history. The Curaçao criteria include the following:
- Spontaneous recurrent epistaxis
- Multiple telangiectasias in typical locations
- Documented visceral arteriovenous malformations (eg, in the lung, liver, brain, and spine)
- First-degree family member with hereditary hemorrhagic telangiectasia
Hereditary hemorrhagic telangiectasia is definite if 3 of these criteria are met and possible if 2 are met.
Endoscopy or angiography is sometimes needed. Laboratory findings are usually normal except for iron deficiency anemia in many patients.
HHT can be diagnosed by performing genetic testing. Genetic testing can detect a gene mutation in about ¾ of families with signs of HHT, which if found can establish the diagnosis of HHT in individuals and families who are unsure about whether they have HHT. HHT can also be diagnosed by using clinical criteria (presence of signs and a history of signs in a parent, sibling, or child). Testing for the ENG, ACVRL1, and MADH4 mutations may be helpful in some patients with atypical features or for screening asymptomatic family members.
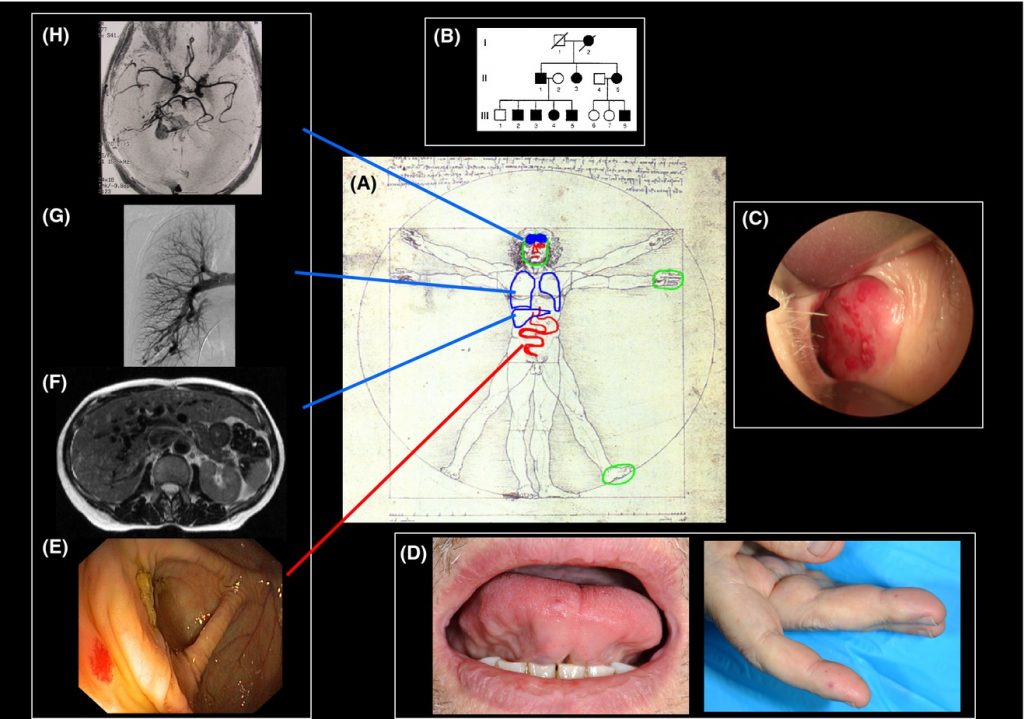
Clinical spectrum of hereditary hemorrhagic telangiectasia (HHT) illustrating its systemic nature and the Curaçao diagnostic criteria. (A) Central body schematic highlighting multisystem involvement. (B) Autosomal dominant inheritance pattern. (C) Nasal mucosal telangiectases causing recurrent spontaneous epistaxis, the most common clinical manifestation of HHT. (D) Mucocutaneous telangiectases involving the lips, tongue, and fingers, which bleed less frequently but are diagnostically important. (E) Gastrointestinal telangiectases that may cause chronic bleeding and iron-deficiency anemia. (F) Hepatic vascular malformations, present in up to 70–75% of patients, typically asymptomatic, shown on magnetic resonance angiography as corkscrew-like vessels. (G) Pulmonary arteriovenous malformations (PAVMs) visible on angiography as peripheral aneurysmal sacs, requiring early detection due to the risk of paradoxical emboli, cerebral abscesses, and infarction; screening is recommended even in asymptomatic patients. (H) Cerebral vascular malformations, which are less common but clinically significant. Together, mucocutaneous findings (B–D) and visceral involvement (E–H) define the four Curaçao criteria used to establish the diagnosis of HHT.
Screening for HHT:
If a family history of pulmonary, hepatic, or cerebral arteriovenous malformations exists, screening at puberty and at the end of adolescence with pulmonary CT, hepatic CT, and cerebral MRI is recommended.
Complications and Treatments of HHT:
The complications of HHT can vary widely, even among people affected by HHT in the same family. Complications and treatment of HHT depend on the parts of the body that are affected by this disorder. Treatment may include controlling bleeding and anemia and preventing complications from abnormal artery-vein connections in the lungs and brain.
- Sometimes laser ablation, surgical resection, or embolization of symptomatic arteriovenous malformations
- Supplemental iron therapy
- Possibly blood transfusions
- Sometimes antifibrinolytic drugs (eg, aminocaproic acid, tranexamic acid)
- Sometimes angiogenesis inhibitors (eg, bevacizumab, pomalidomide, thalidomide)
Treatment for most patients is supportive, but accessible telangiectasias (eg, in the nose or gastrointestinal tract via endoscopy) may be treated with laser ablation. Arteriovenous malformations may be treated by surgical resection or coil embolization.
Repeated blood transfusions may be needed; therefore, immunization with hepatitis B vaccine is important.
Many patients require continuous iron therapy to replace iron lost in repeated mucosal bleeding; many patients require parenteral iron and sometimes erythropoietin.
Treatment with drugs that inhibit fibrinolysis, such as aminocaproic acid or tranexamic acid, may be beneficial.
Treatment with drugs that inhibit angiogenesis such as bevacizumab, pomalidomide, or thalidomide can reduce the number and density of abnormal vessel growth. In addition, bevacizumab has recently been shown to reduce the incidence of nasal and gastrointestinal bleeding.
To avoid paradoxical embolization of particulate matter to the brain through pulmonary arteriovenous malformations, all IV fluids need to be given through a filter.
Ehlers-Danlos syndrome
The Ehlers-Danlos syndromes (EDS) are a group of hereditary disorders of connective tissue that are varied in the ways they affect the body and in their genetic causes. The underlying concern is the abnormal structure or function of collagen and certain allied connective tissue proteins.
They are generally characterized by joint hypermobility (joints that move further than normal range), joint instability (subluxation (partial separation of the articulating surfaces of a joint)) and dislocations (full separation of the surfaces of a joint), scoliosis, and other joint deformities, skin hyperextensibility (skin that can be stretched further than normal) and abnormal scarring, and other structural weakness such as hernias and organ prolapse through the pelvic floor. In the rarer types of EDS, there is also weakness of specific tissues that can lead, for example, to major gum and dental disease, eye disease, cardiac valve and aortic root disorders, and life-threatening abdominal organ, uterine, or blood vessel rupture.
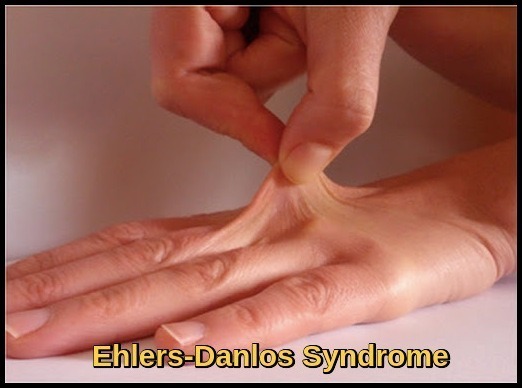
Hyperelastic skin in Ehlers-Danlos syndrome, reflecting abnormal collagen synthesis and connective tissue weakness, a classic inherited cause of vascular fragility and easy bruising within vascular bleeding disorders.

Joint hypermobility in Ehlers-Danlos syndrome, a hereditary connective tissue disorder associated with abnormal collagen structure, vascular fragility, and easy bruising, contributing to vascular bleeding disorders.
Inheritance is usually autosomal dominant, but Ehlers-Danlos syndrome is heterogeneous. Different gene mutations affect the amount, structure, or assembly of different collagens. Mutations can exist in the genes that encode collagens (eg, type I, III, or V) or collagen-modifying enzymes (eg, lysyl hydroxylase, a collagen-cleaving protease).
There are 6 major types:
- Classic
- Hypermobility (hEDS)
- Vascular
- Kyphoscoliosis
- Arthrochalasis
- Dermatosparaxis
There are also several rare or hard-to-classify types.
Easy bruising, at sites of trauma, accompanies most forms of EDS including hEDS. This occurs due to increased fragility of dermal blood capillaries and poor structural integrity of the skin rather than a clotting abnormality.
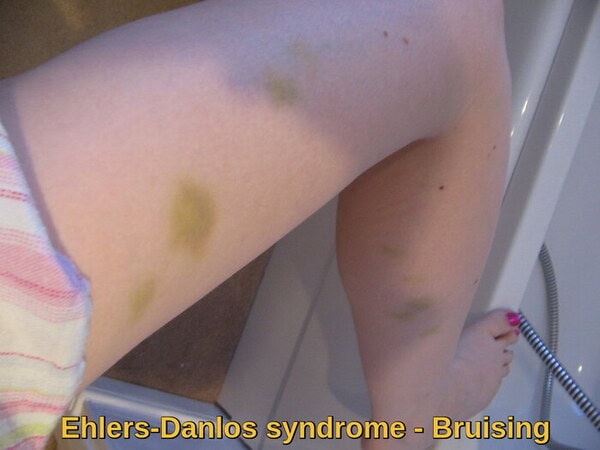
Extensive ecchymoses in Ehlers-Danlos syndrome, illustrating easy bruising caused by connective tissue weakness and increased vascular fragility, a typical manifestation of inherited vascular bleeding disorders.
Symptoms and Signs of Ehlers-Danlos Syndrome:
Symptoms and signs of Ehlers-Danlos syndrome vary widely.
Predominant symptoms include hypermobile joints, abnormal scar formation, and wound healing, fragile vessels, and velvety, hyperextensible skin. Skin can be stretched several centimeters but returns to normal when released.
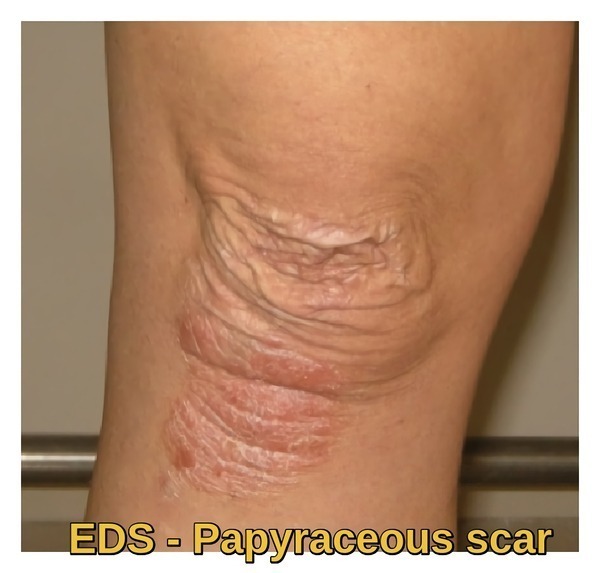
Wide papyraceous scars often overlie bony prominences, particularly elbows, knees, and shins; scarring is less severe in the hypermobility type. Molluscoid pseudotumors (fleshy outgrowths) frequently form on top of scars or at pressure points.
The extent of joint hypermobility varies but may be marked in the arthrochalasis, classic, and hypermobility types.
Bleeding tendency is rare, although the vascular type is characterized by vascular rupture and bruising. Subcutaneous calcified spherules may be palpated or seen on x-rays.
Complications of Ehlers-Danlos syndrome:
Minor trauma may cause wide gaping wounds but little bleeding; surgical wound closure may be difficult because sutures tend to tear out of the fragile tissue. Surgical complications occur because of deep tissue fragility.
Sclera may be fragile, leading to perforation of the globe in the kyphoscoliosis type.
Bland synovial effusions, sprains, and dislocations occur frequently. Spinal kyphoscoliosis occurs in 25% of patients (especially in those with the kyphoscoliosis type), thoracic deformity in 20%, and talipes equinovarus in 5%. About 90% of affected adults have pes planus (flat feet). Developmental dysplasia of the hip (formerly congenital hip dislocation) occurs in 1% (the arthrochalasis type is characterized by bilateral developmental dysplasia of the hip).

Kyphoscoliosis in Ehlers-Danlos syndrome, reflecting severe connective tissue weakness with musculoskeletal deformity, often associated with skin fragility, easy bruising, and vascular involvement in inherited vascular bleeding disorders.
Gastrointestinal (GI) hernias and diverticula are common. Rarely, portions of the GI tract spontaneously hemorrhage and perforate, and dissecting aortic aneurysm and large arteries spontaneously rupture.
Valvular prolapse is a common complication in the most severe type (vascular type).
In pregnant women, tissue extensibility may cause premature birth, cervical incompetence, and possibly uterine rupture; if the fetus is affected, the fetal membrane is fragile, sometimes resulting in early rupture. Maternal tissue fragility may complicate episiotomy or cesarean delivery. Antenatal, perinatal, and postnatal bleeding may occur.
Other potentially serious complications include arteriovenous fistula, ruptured viscus, and pneumothorax or pneumohemothorax.
Diagnosis of Ehlers-Danlos Syndrome:
- Clinical evaluation
- Echocardiography and/or other vascular imaging to screen for cardiovascular complications
The initial diagnosis of Ehlers-Danlos syndrome is largely clinical but should be confirmed by genetic testing, which is now available for most subtypes.
Ultrastructural examination of skin biopsy can help in diagnosing the classic, hypermobility, and vascular types.
Echocardiography and other vascular imaging are done to check for heart disorders (eg, valvular prolapse, arterial aneurysm) that are associated with some of the types.
When to consider the diagnosis of EDS?
Ehlers–Danlos syndrome needs to be considered when, in the absence of another explanation, one or more of the following occur:
- late walking with joint hypermobility
- abnormal bruising and bleeding
- unexplained vessel rupture or dissection
- tissue fragility, atrophic scarring or skin hyperextensibility
- symptomatic joint hypermobility± dislocations
- Hollow organ rupture
Prognosis for Ehlers-Danlos Syndrome:
Life span is usually normal with most types.
Potentially lethal complications occur in certain types (eg, arterial rupture in the vascular type).
Treatment of Ehlers-Danlos Syndrome:
Early recognition and treatment of complications.
There is no specific treatment for Ehlers-Danlos syndrome.
Trauma should be minimized. Protective clothing and padding may help.
If surgery is done, hemostasis must be meticulous. Wounds are carefully sutured, and tissue tension is avoided.
Obstetric supervision during pregnancy and delivery is mandatory.
Genetic counseling should be provided.
Marfan Syndrome
Marfan syndrome (also called Marfan’s syndrome or Marfans syndrome) is a condition that affects the connective tissue. Connective tissue holds the body together and provides support to many structures throughout the body. In Marfan syndrome, the connective tissue isn’t normal. As a result, many-body systems are affected, including the heart, blood vessels, bones, tendons, cartilage, eyes, nervous system, skin, and lungs.

Typical body habitus in an adolescent with Marfan syndrome, demonstrating characteristic skeletal features including kyphoscoliosis, pectus excavatum, and genu recurvatum, reflecting underlying connective tissue abnormalities with potential vascular involvement.
Although some patients with Marfan syndrome complain of easy bruising there is no increased vascular fragility of capillaries or of small and middle-size arteries or veins. This can be explained by the fact that patients with Marfan syndrome may have thinner skin, and less subcutaneous fat, and hence less protective cushioning for minor traumata. Also, joint laxity and poor visual acuity may contribute to a predisposition to contusions in some patients. Some patients with Marfan syndrome present mild hyperextensibility of the skin, reminiscent of EDS. However, the most common skin manifestations of Marfan syndrome are the striae atrophicae or stretch marks, which usually present on the back, the shoulders, and the thighs.
Inheritance of Marfan syndrome is autosomal dominant. About 75% of the time, the condition is inherited from a parent with the condition, while 25% of the time it is a new mutation. The basic molecular defect results from mutations in the gene encoding the glycoprotein fibrillin-1 (FBN1), which is the main component of microfibrils and helps anchor cells to the extracellular matrix. The principal structural defect involves the cardiovascular, musculoskeletal, and ocular systems. The pulmonary system and central nervous system are also affected. There are many different manifestations of the genetic mutation that causes Marfan syndrome; however, it is typically recognized by the constellation of long limbs, aortic root dilation, and dislocated lenses.
Marfan syndrome is fairly common, affecting 1 in 10,000 to 20,000 people. It has been found in people of all races and ethnic backgrounds.
Symptoms and signs of Marfan syndrome:
Sometimes Marfan syndrome is so mild, few if any, symptoms occur. In most cases, the disease progresses with age, and symptoms of Marfan syndrome become noticeable as changes in connective tissue occur.
Cardiovascular system
Major findings include:
Aortic aneurysm
Valvular prolapse
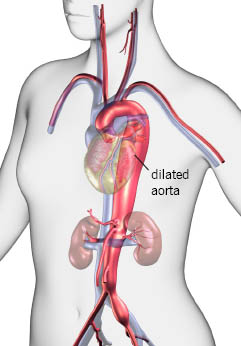
Cardiovascular manifestations of Marfan syndrome illustrating aortic root dilatation and ascending aortic aneurysm, the most clinically significant vascular complication due to connective tissue weakness and abnormal fibrillin-1, with risk of aortic dissection and rupture.
Most severe complications result from pathologic changes in the aortic root and ascending aorta. The aortic media is affected preferentially in areas subject to the greatest hemodynamic stress. The aorta progressively dilates or acutely dissects, beginning in the coronary sinuses, sometimes before 10 years of age. The aortic root dilates in 50% of children and in 60 to 80% of adults and can cause aortic regurgitation, in which case a diastolic murmur may be heard over the aortic valve.
Redundant cusps and chordae tendineae may lead to mitral valve prolapse or regurgitation; mitral valve prolapse may cause a systolic click and a late systolic murmur or, in severe cases, a holosystolic murmur. Bacterial endocarditis may develop in affected valves.
Musculoskeletal system
Severity varies greatly. Patients are taller than average for age and family; arm span exceeds height. Arachnodactyly (disproportionately long, thin digits) is noticeable, often by the thumb sign (the distal phalanx of the thumb protrudes beyond the edge of the clenched fist). Sternum deformity—pectus carinatum (outward displacement) or pectus excavatum (inward displacement)—is common, as are joint hyperextensibility (but usually small flexion contractures to the elbows), genu recurvatum (backward curvature of the legs at the knees), pes planus (flat feet), kyphoscoliosis, and diaphragmatic and inguinal hernias. Subcutaneous fat usually is sparse. The palate is often high-arched.

Positive thumb sign (Steinberg sign) in Marfan syndrome, where the thumb protrudes beyond the ulnar border of the clenched fist, reflecting arachnodactyly and underlying connective tissue abnormality.

Marfan syndrome is characterized by arachnodactyly, with abnormally long, slender fingers. In this image, the wrist sign (Walker–Murdoch sign) is demonstrated, where the thumb and index finger overlap when encircling the opposite wrist, reflecting underlying connective tissue abnormality with potential vascular involvement.
People with Marfan syndrome are often very tall and thin. Their arms, legs, fingers, and toes may seem out of proportion, too long for the rest of their body. Their spine may be curved and their breastbone (sternum) may either stick out or be indented. Their joints may be weak and easily become dislocated. Often, people with Marfan syndrome have a long, narrow face and the roof of the mouth may be higher than normal, causing the teeth to be crowded.
Ocular system
Findings include ectopia lentis (subluxation or upward dislocation of the lens) and iridodonesis (tremulousness of the iris). The margin of the dislocated lens can often be seen through the undilated pupil. High-grade myopia may be present, and spontaneous retinal detachment may occur.
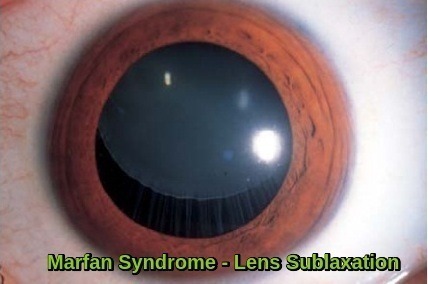
Lens subluxation (ectopia lentis) in Marfan syndrome, a characteristic ocular manifestation caused by weakened zonular fibers due to connective tissue abnormality, and an important diagnostic feature of the condition.
Pulmonary system
Cystic lung disease and recurrent spontaneous pneumothorax may occur. These disorders can cause pain and shortness of breath.
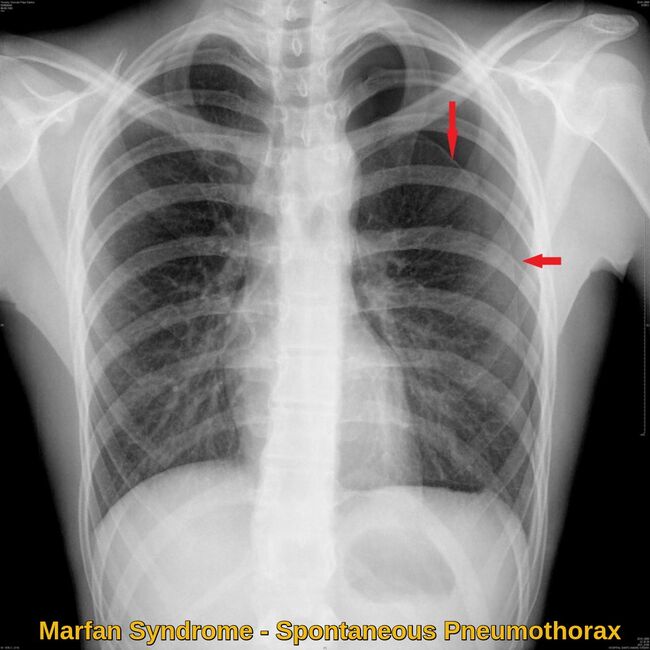
Chest X-ray demonstrating spontaneous pneumothorax in Marfan syndrome, a pulmonary complication related to apical blebs and connective tissue weakness, highlighted by pleural air and partial lung collapse.
Central nervous system
Dural ectasia (widening of the dural sac surrounding the spinal cord) is a common finding and most frequently occurs in the lumbosacral spine. It may cause headache, lower back pain, or neurologic deficits manifested by bowel or bladder weakness.
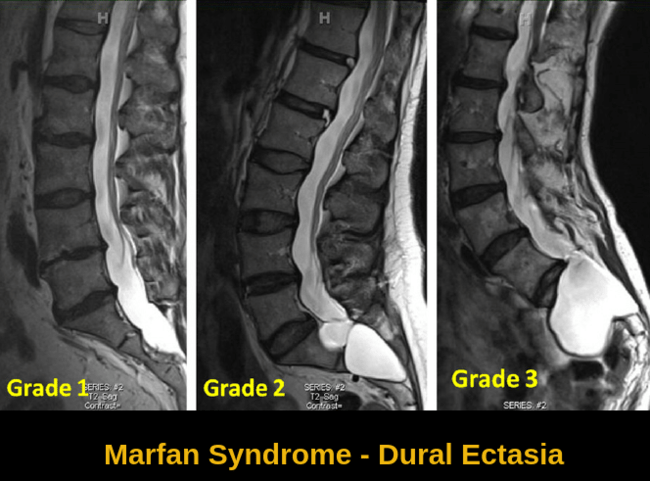
Lumbar spine MRI demonstrating dural ectasia in Marfan syndrome, shown across increasing grades of severity, a common skeletal–neurological manifestation resulting from connective tissue weakness and an important diagnostic feature of the condition.
Diagnosis of Marfan syndrome:
- Genetic testing
- Echocardiography/MRI (measurement of the aortic root, detection of valve prolapse)
- Slit-lamp examination (lens abnormalities)
- X-rays of the skeletal system (hand, spine, pelvis, chest, foot, and skull for characteristic abnormalities)
- MRI of the lumbosacral spine (dural ectasia)
Diagnosis of Marfan syndrome can be difficult because many patients have only a few typical symptoms and signs and no specific histologic or biochemical changes. Considering this variability, diagnostic criteria are based on constellations of clinical findings and family and genetic history.
Nonetheless, diagnosis is uncertain in many partial cases of Marfan syndrome.
Homocystinuria can partially mimic Marfan syndrome but can be differentiated by detecting homocystine in the urine. Genetic testing for FBN1 mutations can help establish the diagnosis in people who do not meet all clinical criteria, but FBN1 mutation-negative cases exist. Prenatal diagnosis by analysis of the FBN1 gene is hampered by poor genotype/phenotype correlation (> 1700 different mutations have been described).
Standard imaging of the skeletal, cardiovascular, and ocular systems is done to detect any clinically relevant structural abnormalities and to provide information contributing to the diagnostic criteria (eg, echocardiography to identify aortic root enlargement).
In addition to the criteria established within organ systems, family history (1st-degree relative with Marfan syndrome) and genetic history (presence of the FBN1 mutation known to cause Marfan syndrome) are considered major criteria.
Prognosis for Marfan syndrome:
Advancements in therapy and regular monitoring have improved quality of life and reduced mortality. Median life expectancy increased from 48 years in 1972 to near normal in people receiving appropriate medical care. However, life expectancy is still reduced for the average patient, primarily because of the cardiac and vascular complications. This decreased life expectancy can take an emotional toll on an adolescent and the family.
Treatment of Marfan syndrome:
- Induction of precocious puberty in tall girls
- Beta-blockers
- Elective aortic repair and valve repair
- Bracing and surgery for scoliosis
Treatment of Marfan syndrome is focused on prevention and treatment of complications.
For very tall girls, inducing precocious puberty by age 10 with estrogens and progesterone may reduce potential adult height.
All patients should routinely be given beta-blockers (eg, atenolol, propranolol) to help prevent cardiovascular complications. These drugs lower myocardial contractility and pulse pressure and reduce the progression of aortic root dilation and the risk of dissection. In people who are unable to take beta-blockers due to asthma or side effects, a calcium channel blocker, such as verapamil, is recommended. Angiotensin II receptor blockers also may be given.
Prophylactic surgery is offered if the aortic diameter is > 5 cm (less in children). Pregnant women are at especially high risk of aortic complications; elective aortic repair before conception should be discussed. Severe valve regurgitation is also surgically repaired. Bacterial endocarditis prophylaxis before invasive procedures is not indicated except in patients who have prosthetic valves or who previously had infective endocarditis.
Scoliosis is managed with bracing as long as possible, but surgical intervention is encouraged in patients with curves of 40 to 50°.
Cardiovascular, skeletal, and ocular findings (including echocardiography) should be reevaluated annually. Appropriate genetic counseling is indicated.
Questions and Answers:
What are vascular bleeding disorders?
Vascular bleeding disorders are conditions in which abnormalities of the blood vessel wall or surrounding connective tissue lead to bleeding manifestations such as easy bruising, petechiae, purpura, and mucocutaneous bleeding, despite normal platelet counts and coagulation studies.
How do vascular bleeding disorders differ from platelet or coagulation disorders?
Unlike platelet or coagulation factor disorders, vascular bleeding disorders are caused by structural vessel wall weakness rather than defects in hemostasis, and they typically result in superficial bleeding rather than deep tissue or joint hemorrhage.
What are the most common clinical features of vascular bleeding disorders?
Common features include easy bruising, petechiae, purpura, epistaxis, gingival bleeding, and prolonged bleeding after minor trauma, while severe or life-threatening hemorrhage is uncommon except in specific conditions such as hereditary hemorrhagic telangiectasia.
Which inherited conditions cause vascular bleeding disorders?
Inherited causes include Ehlers-Danlos syndrome, Marfan syndrome, hereditary hemorrhagic telangiectasia, pseudoxanthoma elasticum, osteogenesis imperfecta, and other rare connective tissue disorders affecting collagen or elastic fibers.
What is hereditary hemorrhagic telangiectasia (HHT)?
Hereditary hemorrhagic telangiectasia is an autosomal dominant vascular disorder characterized by mucocutaneous telangiectases, recurrent epistaxis, and visceral arteriovenous malformations involving the lungs, liver, brain, or gastrointestinal tract.
Why does HHT cause recurrent nosebleeds?
HHT causes fragile telangiectatic vessels within the nasal mucosa that rupture easily, leading to spontaneous and recurrent epistaxis, which is often the earliest and most common manifestation of the disease.
What are the Curaçao criteria for diagnosing HHT?
The Curaçao criteria include recurrent epistaxis, mucocutaneous telangiectases, visceral arteriovenous malformations, and a first-degree family history of HHT; the diagnosis is definite if three or more criteria are present.
Which visceral organs are commonly affected in HHT?
Visceral involvement in HHT most commonly affects the lungs, liver, gastrointestinal tract, and brain, where arteriovenous malformations may lead to anemia, hypoxemia, stroke, cerebral abscess, or high-output cardiac failure.
What causes easy bruising in Ehlers-Danlos syndrome?
Easy bruising in Ehlers-Danlos syndrome results from defective collagen synthesis, leading to fragile blood vessels and poor structural support of the vascular wall.
What are papyraceous scars in Ehlers-Danlos syndrome?
Papyraceous, or cigarette-paper, scars are thin, widened scars that reflect impaired wound healing and skin fragility, a characteristic feature of Ehlers-Danlos syndrome.
How does Marfan syndrome cause vascular complications?
Marfan syndrome causes vascular complications through abnormal fibrillin-1 and connective tissue weakness, leading to aortic root dilatation, aneurysm formation, and increased risk of aortic dissection.
What skeletal features suggest Marfan syndrome?
Typical skeletal features include tall stature, long limbs, arachnodactyly, pectus deformities, scoliosis, increased arm-span-to-height ratio, and positive wrist or thumb signs.
What is the thumb sign in Marfan syndrome?
The thumb sign, or Steinberg sign, is positive when the thumb protrudes beyond the ulnar border of the clenched fist, reflecting arachnodactyly and elongated phalanges.
What is dural ectasia and why is it important in Marfan syndrome?
Dural ectasia is dilation of the dural sac in the lumbar spine caused by connective tissue weakness, and it is a common but often under-recognized diagnostic feature of Marfan syndrome.
Can vascular bleeding disorders present with normal laboratory tests?
Yes, routine coagulation studies and platelet counts are often normal in vascular bleeding disorders, making clinical examination and history essential for diagnosis.
What acquired conditions can cause vascular bleeding disorders?
Acquired causes include vitamin C deficiency (scurvy), IgA-associated vasculitis, inflammatory small-vessel vasculitis, corticosteroid-induced skin fragility, and age-related vascular fragility.
When should vascular bleeding disorders be suspected?
They should be suspected in patients with lifelong easy bruising, normal coagulation tests, connective tissue features, or mucocutaneous bleeding disproportionate to laboratory findings.
References:
David J. Kuter, MD, DPhil. Overview of Vascular Bleeding Disorders. MSD Manual Professional Edition.
https://www.msdmanuals.com/en-gb/professional/hematology-and-oncology/bleeding-due-to-abnormal-blood-vessels/overview-of-vascular-bleeding-disorders
Shovlin CL, Guttmacher AE, Buscarini E, et al. Diagnostic criteria for hereditary hemorrhagic telangiectasia (Rendu-Osler-Weber syndrome). American Journal of Medical Genetics. 2000;91(1):66–67. doi:10.1002/(SICI)1096-8628(20000306)91:1<66::AID-AJMG12>3.0.CO;2-P
Kritharis A, Al-Samkari H, Kuter DJ. Hereditary hemorrhagic telangiectasia: Diagnosis and management from the hematologist’s perspective. Haematologica. 2018;103:1433–1443. doi:10.3324/haematol.2018.193003
Al-Samkari H, Kritharis A, Rodriguez-Lopez JM, Kuter DJ. Systemic bevacizumab for the treatment of chronic bleeding in hereditary haemorrhagic telangiectasia. Journal of Internal Medicine. 2019;285(2):223–231. doi:10.1111/joim.12832
Geisthoff UW, Nguyen HL, Röth A, Seyfert U. How to manage patients with hereditary haemorrhagic telangiectasia. British Journal of Haematology. 2015;171:443–452. doi:10.1111/bjh.13606
De Paepe A, Malfait F. Bleeding and bruising in patients with Ehlers–Danlos syndrome and other collagen vascular disorders. British Journal of Haematology. 2004;127:491–500. doi:10.1111/j.1365-2141.2004.05220.x
Sobey G. Ehlers–Danlos syndrome: How to diagnose and when to perform genetic tests. Archives of Disease in Childhood. 2015;100:57–61
Frank Pessler, MD, PhD. Ehlers-Danlos Syndrome. MSD Manual Professional Edition.
https://www.msdmanuals.com/en-gb/professional/pediatrics/connective-tissue-disorders-in-children/ehlers-danlos-syndrome
Frank Pessler, MD, PhD. Marfan Syndrome. Merck Manuals Professional Edition.
https://www.merckmanuals.com/professional/pediatrics/connective-tissue-disorders-in-children/marfan-syndrome
Marco Castori. Ehlers-Danlos syndrome, hypermobility type: An underdiagnosed hereditary connective tissue disorder with mucocutaneous, articular, and systemic manifestations. ISRN Rheumatology. 2012;Article ID 751768. doi:10.5402/2012/751768
Mesfin A, Ahn NU, Carrino JA, Sponseller PD. Ten-year clinical and imaging follow-up of dural ectasia in adults with Marfan syndrome. Spine Journal. 2013;13. doi:10.1016/j.spinee.2012.10.021
Viveiro C, Rocha P, Carvalho C, et al. Spontaneous pneumothorax as a manifestation of Marfan syndrome. BMJ Case Reports. 2013;2013:bcr2013201697
Geisthoff UW, et al. Hereditary haemorrhagic telangiectasia (Rendu-Osler-Weber disease) as a rare disease relevant for otorhinolaryngology. Laryngo-Rhino-Otologie. 2011;90:230–242
Centers for Disease Control and Prevention (CDC). Facts About Hereditary Hemorrhagic Telangiectasia (HHT).
https://www.cdc.gov/ncbddd/hht
National Organization for Rare Disorders (NORD). Hereditary Hemorrhagic Telangiectasia Overview.
https://rarediseases.org/rare-diseases/hereditary-hemorrhagic-telangiectasia
StatPearls Publishing. Ehlers-Danlos Syndrome. StatPearls [Internet].
https://www.ncbi.nlm.nih.gov/books/NBK549814/
StatPearls Publishing. Marfan Syndrome. StatPearls [Internet].
https://www.ncbi.nlm.nih.gov/books/NBK537339/
Michael A. Briones. Vascular Bleeding Disorders. ScienceDirect Topics.
https://www.sciencedirect.com/science/article/pii/B9780123744326001044
Cleveland Clinic. Marfan Syndrome Overview.
https://my.clevelandclinic.org/health/diseases/17209-marfan-syndrome
Medscape. Hereditary Hemorrhagic Telangiectasia (HHT) Overview.
https://emedicine.medscape.com/article/1085062-overview
Keywords:
vascular bleeding disorders, blood vessel bleeding disorders, abnormal blood vessels bleeding, hereditary hemorrhagic telangiectasia, HHT, Osler-Weber-Rendu syndrome, HHT Curaçao criteria, HHT diagnosis, HHT treatment, recurrent epistaxis causes, mucocutaneous telangiectasia, pulmonary arteriovenous malformations, visceral AVMs, vascular malformations hematology, Ehlers-Danlos syndrome, EDS, Ehlers-Danlos vascular type, Ehlers-Danlos bruising, connective tissue bleeding disorders, papyraceous scars, hyperelastic skin, Marfan syndrome, Marfan syndrome vascular complications, aortic root dilatation Marfan, dural ectasia Marfan, Marfan spontaneous pneumothorax, inherited connective tissue disorders, petechiae purpura bruising causes, normal coagulation bleeding, acquired vascular bleeding disorders, hematology vascular disorders, Ask Hematologist, Dr Moustafa Abdou


Request Online Consultation With Dr M Abdou
Fee: US$100
Secure payment via PayPal (credit and debit cards accepted)
Pay Now