Pediatric Hematology

Compassionate consultation for pediatric blood disorders and oncology: A specialist checking a smiling child patient’s heart and lungs in a medical setting.
Pediatric hematology and general hematology center on the study of blood cells, beginning with embryonic hematopoiesis and progressing to mature cell functions. These blood components—red blood cells (erythrocytes), white blood cells (leukocytes), and platelets (thrombocytes)—are essential for oxygen transport, immune defense, and hemostasis.

Diagram illustrating major blood cell types in pediatric hematology, including erythrocytes, platelets, and white blood cell subtypes relevant to childhood blood disorders.
Pediatric hematology has emerged as a specialized science with age-specific reference ranges that correlate with the hematopoietic, immunologic and chemical changes in a developing child. A newborn represents the culmination of developmental events from conception and implantation through organogenesis. The embryo requires red cells for the transport of maternal oxygen to permit this growth and development. Birth brings dramatic changes in circulation and oxygenation, which affects hematopoiesis, as the newborn makes the transition to a separate biological existence.
During embryogenesis, hematopoiesis occurs in distinct sites, including the extraembryonic yolk sac, the fetal liver, and the preterm bone marrow. Erythropoiesis is established soon after implantation of the blastocyst, with primitive erythroid cells appearing in yolk sac blood islands by day 18 of gestation.
Hematopoietically active bone marrow is referred to as red marrow, as opposed to inactive yellow (fatty) marrow. At the time of birth, the bone marrow is fully active and extremely cellular, with all hematopoietic cell lineages undergoing cellular differentiation and amplification. In addition to the mature cells in fetal blood, there are significant numbers of circulating progenitor cells in cord blood.
In a full-term infant, hepatic hematopoiesis has ceased except in widely scattered small foci that become inactive soon after birth. Postembryonic extramedullary hematopoiesis is abnormal in a full-term infant. In a premature infant, foci of hematopoiesis are frequently seen in the liver and occasionally observed in the spleen, lymph nodes, or thymus.
Dramatic changes occur in the blood and bone marrow of the newborn infant during the first hours and days after birth and there are rapid fluctuations in the quantities of all hematologic elements. The values of most of the hematological parameters studied were highest especially hemoglobin concentration, packed cell volume, reticulocyte count and red cell indices on the first day of life and thereafter declined. Factors contributing to the decline in hematological parameters in the newborn are due to decrease in blood erythropoietin concentration soon after birth, reducing the erythropoietic rate. Also, transient hemolysis is high during the first days or week after birth. Significant hematologic differences are seen between term and preterm infants and among newborns, infants, young children, and older children.
Neonatal hematologic values are affected by the gestational age of the infant, the age in hours after delivery, the presence of illness, and the level of support required. Other important variables to be considered when evaluating laboratory data include the site of sampling and technique (capillary versus venous puncture, warm or unwarmed extremity), the timing of sampling, and conditions such as the course of labor and the treatment of the umbilical vessels. The presence of fetal hemoglobin (Hb F), bilirubin, and lipids in newborns can also interfere with hematology laboratory testing. As with all laboratory testing, each laboratory should establish reference intervals based on its instrumentation, methods, and patient population.
The concentration of hemoglobin fluctuates dramatically in the weeks and months after birth as a result of physiologic changes, and various factors must be considered when analyzing pediatric hematologic values. The site of sampling, gestational age, and the time interval between delivery and clamping of the umbilical cord can influence the hemoglobin level in newborn infants. In addition, there are significant differences between capillary and venous blood hemoglobin levels. Capillary samples in newborns generally have a higher hemoglobin concentration than venous samples, which can be attributed to circulatory factors. Racial differences must also be considered when evaluating hemoglobin levels in children. African American children have hemoglobin levels averaging 0.5 g/dL lower than those in white children.

Understanding the ‘Physiological Anemia of Infancy’: Normal Hemoglobin (Hb) levels change significantly during the first six months of life. Use this table as a quick reference for expected values in full-term vs. premature infants.
Hemoglobin synthesis results from an orderly evolution of a series of embryonic, fetal, and adult hemoglobins. At birth, Hb F constitutes 70% to 80% of the total hemoglobin. Hb F declines from 90% to 95% at 30 weeks’ gestation to approximately 7% at 12 weeks after birth and stabilizes at 3.2 ± 2.1% at 16 to 20 weeks after birth. The switch from Hb F to Hb A is genetically controlled and determined by gestational age; it does not appear to be influenced by the age at which birth occurs.
Early normoblasts are megaloblastic, hypochromic, and irregularly shaped. During hepatic hematopoiesis, normoblasts are smaller than the megaloblasts of the yolk sac but are still macrocytic. Erythrocytes remain macrocytic from the first 11 weeks of gestation until day 5 of postnatal life.
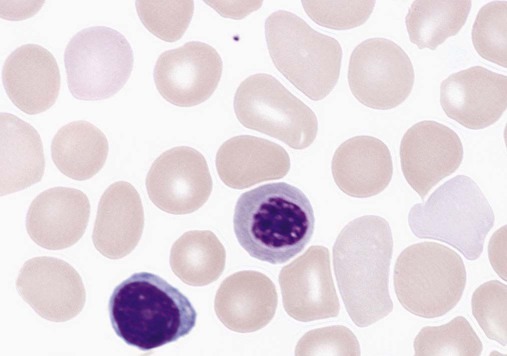
Normal findings in a newborn blood film: The presence of a few Nucleated Red Blood Cells (NRBCs) is a common and usually benign finding in term infants due to extramedullary hematopoiesis.
The macrocytic RBC morphology gradually changes to the characteristic normocytic, normochromic morphology. Orthochromic normoblasts frequently are observed in the full-term infant on the first day of life but disappear within postnatal days 3 to 5. These nucleated RBCs (NRBCs) may persist longer than a week in immature infants. The average number of NRBCs ranges from 3 to 10 per 100 white blood cells (WBCs) in a normal full-term infant to 25 NRBCs per 100 WBCs in a premature infant. The presence of NRBCs for more than 5 days suggests hemolysis, hypoxic stress, or acute infection.

Blood Film Findings in Prematurity: Premature infants often demonstrate a higher count of Nucleated Red Blood Cells (NRBCs) and greater anisocytosis (variation in red cell size) compared to full-term neonates.
The erythrocytes of newborns show additional morphologic differences. The number of biconcave discs relative to stomatocytes is reduced in neonates (43% discs, 40% stomatocytes) compared with adults (78% discs, 18% stomatocytes). In addition, increased numbers of pitted cells, echinocytes, spherocytes, and other abnormally shaped erythrocytes are seen in neonates. The number of these “dysmorphic” cells is even higher in premature infants. Zipursky et al found 40% discs, 30% stomatocytes, and 27% additional poikilocytes in premature infants.
Apparent reticulocytosis exists during gestation, decreasing from 90% reticulocytes at 12 weeks’ gestation, to 15% at 6 months’ gestation, and ultimately to 4% to 6% at birth. Reticulocytosis persists for about 3 days after birth, then declines abruptly to 0.8% reticulocytes on postnatal day 4 to 7. At 2 months, the number of reticulocytes increases slightly, followed by a slight decline from 3 months to 2 years, when adult levels of 0.5% to 1.5% are attained. The reticulocyte count of premature infants is typically higher than that of term infants; however, the count can vary dramatically depending upon how ill the newborn is. Significant polychromasia seen on a Wright-stained blood film is indicative of postnatal reticulocytosis
In summary, hemopoiesis mainly occurs in the liver of the fetus. After birth and throughout life, hemopoiesis occurs in the bone marrow. In infants and the growing child, this occurs in all bones of the skeleton. An understanding of the normal cytology is essential in order to allow for the identification of abnormalities on the blood film. Red cells are the most numerous cell type encountered in the blood film. In the pediatric film, normal red cells are the size of the lymphocyte nucleus with a diameter of 7–9µm and a mean corpuscular volume (MCV) of 75–90 fL. They should be round in shape with a smooth contour appearing as a biconcave disc. Approximately, one-third of the cell should have a central pallor. The neonatal blood film differs from the pediatric blood film. It is not uncommon to see burr cells (echinocytes), occasional nucleated red blood cells (RBC), target cells, fragmented red cells, and some spherocytes. Neonates typically have an elevated MCV and red cells are therefore macrocytic. Nucleated red cells or normoblasts are immature red cells with a retained nucleus. They are usually found only in the circulating blood of the fetus and the newborn infant. Post infancy, their presence in the peripheral blood is indicative of disorder in blood production. Nucleated red blood cells are immature cells, they do not enter the peripheral blood under normal circumstance. They are often seen in the peripheral blood in leukoerythroblastic anemia, hemolysis, hypoxia, and marrow infiltration.
White blood cells can be divided into the myeloid/monocytic cells (neutrophils, eosinophils, basophils, and monocytes) and lymphocytes. Segmented neutrophils are the predominant white cells in the peripheral blood. The total white cell count and the neutrophil, monocyte and lymphocyte counts are often much higher in the neonate than the older child. In addition, it is important to remember that the automated lymphocyte count may be
falsely elevated due to the presence of nucleated red blood cells.
Healthy newborn infants may have a WBC count from 13,000 to 38,000 per mm3 (13.0 to 38.0 × 109 per L) at 12 hours of life. By two weeks of age, this decreases to approximately 5,000 to 20,000 per mm3 (5.0 to 20.0 × 109 per L), and gradually declines throughout childhood to reach adult levels of 4,500 to 11,000 per mm3 (4.5 to 11.0 × 109 per L) by about 21 years of age. There is also a shift from relative lymphocyte to neutrophil predominance from early childhood to the teenage years and adulthood.

WCC Reference Range: Normal total leukocyte (white cell) counts vary significantly by age and physiological state. Note the substantially higher normal range for newborn infants.
Platelets are small, non-nucleated cells. They normally measure 1.5–3 µm in diameter. They are derived from the cytoplasmic fragments of megakaryocytes. The platelet count usually ranges from 150 to 400 × 109/L for full-term and preterm infants, comparable to adult values. Platelet counts generally increase in both term and preterm infants in the first few months of life, as evidenced by increased mean platelet volume in the first month of life. Thrombocytopenia of fewer than 100 × 109 platelets/L may be seen in high-risk infants with sepsis or respiratory distress and neonates with trisomy syndromes, and investigation should be undertaken for underlying pathology. Platelets of a newborn infant show great variation in size and shape.
The physiology of the hemostatic system in infants and children is different from that in adults. The vitamin K–dependent coagulation factors (factors II, VII, IX, and X) are at about 30% of adult values at birth; they reach adult values after 2 to 6 months, although the mean values remain lower in children than in adults. Levels of factor XI, factor XII, prekallikrein, and high-molecular-weight kininogen are between 35% and 55% of adult values at birth, reaching adult values after 4 to 6 months. In contrast, the levels of fibrinogen, factor VIII, and von Willebrand factor are similar to adult values throughout childhood. Factor V decreases during childhood, with lower levels during the teen years as compared with adults. The physiologic anticoagulants and inhibitors of coagulation—protein C, protein S, antithrombin, and a disintegrin-like and metalloprotease domain with thrombospondin type 1 motifs 13 (ADAMTS 13)—are reduced to about 30% to 40% at birth. Antithrombin reaches adult values by 3 months, whereas protein C does not normalize until after 6 months. In the fibrinolytic system, levels of plasminogen and α2-antiplasmin are similar to adult levels at birth, whereas levels of tissue plasminogen activator are low and levels of plasminogen activator inhibitor (PAI) are increased. The hemostatic components are not only changing in concentration over the first few weeks to months of life, but their values are also dependent upon the gestational age of the child, and premature infants have different values at birth than term infants.
Blood disorders in children can be broadly classified into three categories: anemia, bleeding disorders, and blood cancers.
Anemia:
Anemia is a condition in which there is a decrease in the number of red blood cells (RBCs) or the amount of hemoglobin (Hb) in the blood. This can lead to fatigue, weakness, shortness of breath, and other symptoms. The most common types of anemia in children are iron deficiency anemia, thalassemia, and sickle cell anemia.
Iron deficiency anemia is caused by a lack of iron in the body, which is needed to make hemoglobin. Thalassemia is an inherited condition in which the body produces abnormal hemoglobin. Sickle cell anemia is also an inherited condition in which the body produces abnormal hemoglobin, leading to the formation of sickle-shaped RBCs that can cause pain and other complications.

Identifying Iron Deficiency Anemia: This blood film demonstrates red cells that are microcytic (smaller than normal) and hypochromic (pale, with an increased central pallor), the hallmarks of iron deficiency.

Classic Signs of Thalassemia: The persistent, severe anemia in Thalassemia Major leads to excessive bone marrow expansion, causing characteristic maxillary hypertrophy (left) and the pathognomonic ‘hair-on-end’ appearance on the skull X-ray (right).

The Sickle Cell Blood Film: The key diagnostic finding in Sickle Cell Disease is the presence of drepanocytes (sickle-shaped red cells), along with features of hemolysis like target cells and howell-jolly bodies (often visible due to splenic dysfunction).
Bleeding Disorders:
Bleeding disorders are conditions in which the blood does not clot properly, leading to excessive bleeding or bruising. The most common bleeding disorder in children is von Willebrand disease, which is caused by a deficiency or dysfunction of von Willebrand factor, a protein that helps the blood clot.

Recognizing VWD: Frequent, prolonged nosebleeds (epistaxis) and easy bruising are hallmark symptoms of Von Willebrand Disease (VWD), the most common inherited bleeding disorder.
Other bleeding disorders in children include hemophilia A and B, which are caused by a deficiency of clotting factors VIII and IX, respectively. These disorders can cause spontaneous bleeding into joints and muscles, as well as prolonged bleeding after injury or surgery.
Blood Cancers:
Blood cancers are a group of disorders that affect the production and function of blood cells. The most common types of blood cancers in children are leukemia and lymphoma.
Leukemia is a cancer of the white blood cells (WBCs) that can affect both children and adults. The two main types of leukemia in children are acute lymphoblastic leukemia (ALL) and acute myeloid leukemia (AML).

Identifying Lymphoblasts: These large, immature white cells (lymphoblasts) with scant cytoplasm and a high nuclear-to-cytoplasmic ratio are the hallmark finding in the peripheral blood of patients with Acute Lymphoblastic Leukemia (ALL).
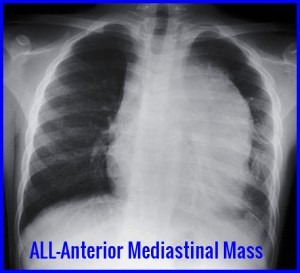
Mediastinal Mass in ALL: The presence of a prominent anterior mediastinal mass is a classic presentation for the T-cell subtype of Acute Lymphoblastic Leukemia (T-ALL), sometimes causing symptoms like superior vena cava (SVC) syndrome.
Lymphoma is a cancer of the lymphatic system, which is part of the immune system. The two main types of lymphoma in children are Hodgkin lymphoma and non-Hodgkin lymphoma.

Hodgkin Lymphoma Presentation: In children and adolescents, the most common presentation of Hodgkin Lymphoma is a painless, firm enlargement of lymph nodes in the neck (cervical lymphadenopathy).
Diagnosis and Treatment:
The diagnosis of blood disorders in children typically involves a combination of physical examination, laboratory tests, and imaging studies. Treatment depends on the type and severity of the disorder.
Anemia may be treated with iron supplements, blood transfusions, or bone marrow transplantation in severe cases. Bleeding disorders may be treated with clotting factor replacement therapy, desmopressin, or other medications. Blood cancers may be treated with chemotherapy, radiation therapy, bone marrow transplantation, or a combination of these treatments.
Summary:
Pediatric hematology is a sub-specialty of medicine that deals with the study and treatment of blood disorders in children. These disorders can range from benign conditions, like certain types of pediatric anemia, to life-threatening conditions, such as childhood leukemia, affecting the production and function of blood cells. Pediatric hematologists are physicians who specialize in the diagnosis and management of these disorders, providing the specialized knowledge and expertise required to address the unique hematopoietic challenges of a developing child. With ongoing advances in research and technology, there is increasing hope for improved outcomes and quality of life for children with these conditions.
Questions and Answers:
What is pediatric hematology and how is it different from adult hematology?
Pediatric hematology is the medical specialty focused on diagnosing and managing blood and bone marrow disorders in infants, children, and adolescents, with age-specific reference ranges and developmental considerations that differ from adults.
What are the most common childhood anemias and how are they diagnosed?
The most common pediatric anemias include iron deficiency anemia, thalassemia, and sickle cell anemia; diagnosis is based on clinical signs and laboratory tests such as complete blood count and peripheral smear.
What symptoms suggest a blood disorder in a child?
Symptoms of pediatric blood disorders may include fatigue, pallor, frequent infections, unexplained bruising, prolonged bleeding, or unusual bleeding episodes.
When should I suspect a bleeding disorder in a child?
Consider evaluation for a bleeding disorder when a child experiences excessive bruising, frequent nosebleeds, or prolonged bleeding after minor injuries; common conditions include von Willebrand disease and hemophilia.
What are the main types of childhood leukemia and how are they treated?
Acute lymphoblastic leukemia (ALL) is the most common childhood leukemia, accounting for the majority of cases, while acute myeloid leukemia (AML) is less common. Treatment is risk-adapted and primarily based on multi-agent chemotherapy; selected high-risk cases may require hematopoietic stem cell transplantation or targeted therapies depending on leukemia subtype and molecular features.
How is sickle cell disease managed in children?
Management of sickle cell disease in pediatric patients includes routine monitoring, pain crisis management, hydration, infection prevention, and potentially disease-modifying therapies such as hydroxyurea.
What tests are used to diagnose pediatric blood cancers?
Diagnosis of pediatric blood cancers involves careful review of the peripheral blood smear, complete blood count with differential, and confirmatory bone marrow aspiration or biopsy. Immunophenotyping by flow cytometry, cytogenetic analysis, and molecular genetic studies are essential to accurately classify the leukemia, assess prognosis, and guide risk-adapted therapy. Imaging studies are used when needed to evaluate disease extent or extramedullary involvement.
Can normal hematologic values in children differ from adults?
Yes — normal hematologic values vary with age, with neonates and young infants having distinct hemoglobin and white blood cell ranges compared to older children and adults.
What role do pediatric hematologists play in care?
Pediatric hematologists specialize in accurately diagnosing and managing blood disorders in children, coordinating multidisciplinary care and individualized treatment plans.
Are inherited blood disorders common in children?
Inherited blood disorders such as sickle cell disease and thalassemias are important causes of pediatric anemia and require lifelong specialist care.
References:
ESAN AJ. Hematological differences in newborn and aging: a review study. Hematology & Transfusion International Journal. 2016;3(3):178–190.
https://medcraveonline.com/HTIJ/HTIJ-03-00067.php
GOOSSEN LH. Pediatric and geriatric hematology. Oncohema Key.
https://oncohemakey.com/pediatric-and-geriatric-hematology/
MCNAUGHTEN B, THOMPSON A, MACARTNEY C, et al. Interpreting the full blood count in children. Archives of Disease in Childhood – Education and Practice Edition. 2018;103:263–266.
https://ep.bmj.com/content/edpract/103/5/263.full.pdf
RILEY LK, RUPERT J. Evaluation of patients with leukocytosis. American Family Physician. 2015;92(11):1004–1011.
https://www.aafp.org/afp/2015/1201/p1004.html
ADEWOYIN AS, NWOGOH B. Peripheral blood film – a review. Annals of Ibadan Postgraduate Medicine. 2014;12:71–79.
GORDON-SMITH T. Haemopoiesis – the formation of blood cells. Medicine. 2009;37:129–132.
HAYS T, JAMIESON B. Atlas of Paediatric Peripheral Blood Smears. 1st ed. Abbott Laboratories; 2008.
BAIN BJ, BATES I, LAFFAN MA. Dacie and Lewis Practical Haematology. 12th ed. Elsevier Health Sciences; 2016.
BAIN BJ. Blood Cells: A Practical Guide. 5th ed. John Wiley & Sons; 2015.
AMBOSS. Hodgkin lymphoma.
https://www.amboss.com/us/knowledge/hodgkin-lymphoma
NATHAN DG, ORKIN SH. Hematology of Infancy and Childhood. 8th ed. Philadelphia: Saunders Elsevier; 2015.
LANZKOWSKY P. Manual of Pediatric Hematology and Oncology. 5th ed. San Diego: Elsevier Academic Press; 2011.
KLIEGMAN RM, STANTON BF, ST GEME JW III, SCHOR NF. Nelson Textbook of Pediatrics. 20th ed. Philadelphia: Elsevier; 2016.
AMERICAN SOCIETY OF HEMATOLOGY. Pediatric hematology: a resource for healthcare providers. Accessed August 8, 2023.
https://www.hematology.org/education/pediatric-hematology
Keywords:
pediatric hematology, pediatric hematology oncology, pediatric blood disorders, childhood blood disorders, pediatric hematologic diseases, pediatric anemia, anemia in children, childhood leukemia, pediatric leukemia diagnosis, pediatric AML and ALL, pediatric lymphoma, hematologic malignancies in children, pediatric thrombocytopenia, pediatric bleeding disorders, pediatric coagulation disorders, pediatric hemophilia, pediatric sickle cell disease, thalassemia in children, inherited blood disorders in children, pediatric blood smear interpretation, pediatric bone marrow examination, pediatric flow cytometry, pediatric cytogenetic studies, pediatric molecular hematology, pediatric hematologist, pediatric hematology specialist, pediatric hematology clinic, pediatric hematology diagnosis and treatment, pediatric hematology services

Request Online Consultation With Dr M Abdou
Fee: US$100
Secure payment via PayPal (credit and debit cards accepted)
Pay Now



My son has been having some health issues, and he might need blood work. It makes sense that I would want to find a professional who has experience doing that kind of work with children! I can imagine it would be pretty hard to do without practice.
Hi Braden,
Thank you for your message.
I hope your son will get well soon.
Happy to advise but need additional information on the medical problem your son suffers from.
Best wishes,
Hi! My son is a 3yo boy with allergic rhinitis but otherwise healthy with no symptoms or signs of any disease recently. In his control labs they found inmature granulocytes elevated (110/uL, normal percentage of 1%), no anemia, no leukopenia, normal platelets and eosinophils slightly elevated (1000/uL). In the red formula there is a slightly elevated MCHC (35.6)
Is there any reason why I should worry about?
Hi,
Thank you for your comment.
No, there is nothing here to worry about. These changes are likely reactive to infection or to his allergy.
BW,
My almost 4 month old son had bloodwork after a nosebleed while he was sleeping and results indicate teardrop cells 1+ and burr cells 1+. Is this something to be worried about? We have a followup with a pediatric hematologist but it is over a month away. Should I try to request an earlier appointment?
Hi,
Thanks for your comment.
I would suggest checking his platelet count and coagulation screen.
BW,
My 5 year old daughter has chronic allergic rhinitis. She had a CBC and blood smear where rare immunoblasts and numerous ovalocytes were noted as well as occasional atypical lymphocytes and large granular lymphocytes. Also increased RBC, Platelet count of 454 and a lymphocyte percentage of 57%. Could all of this be allergy related or should a hematologist follow up?
Hi Amanda,
Thanks for your comment.
Likely reactive changes.
I would suggest checking her CRP and treating any underlying infection.
Also, maintain adequate hydration and repeat her CBC after 4 weeks.
BW,
When my daughter was only 17 days old, we had a trip to the ER and they ran a CBC w/diff auto diff, differential morphology and LFTs with a couple other tests. She had poikilocytosis 2+, tear cells 1+, burr cells 1+. Her auto diff showed “low” absolute neutrophils of 2.06 x10(3)/mcL but higher than normal absolute monocytes, eosinophils and basophils. Her absolute imm gran automated was 0.08 x10(3)/mcL. Percentage breakdown of these were neutrophils 18.3%, lymphs 66.1, monocytes 9, eosinophils 5.1, basophils 0.8 and imm gran automated 0.7. Platelet count was 611 x10(3)/mcL with a mean platelet volume of 9.9 fL. Nucleated was 0. Bilirubin direct was 0.5 and bilirubin total was 1.7. Alanine Aminotransferase was 38 and the aspartate was 71. No one ever mentioned any of this to me, is this normal in a 17 day old or should I have a follow up appointment and additional blood work? We do not ever remember her being sick at this time, we were in the ER for a broken bone.
Hi Kathryn,
Thanks for your comment.
The changes you have mentioned in your daughter’s CBC are non-specific and they don’t mean any particular pathology in a 17 days old girl.
If she feels well in herself you just need to repeat her CBC and to check her hematinics (iron/B12/Folate).
BW,
After Intrauterine death of my son in 28+4 weeks pregnancy ,they did a lot of blood work every thing was fine but my plasma protein C was slightly elevated normal value is 70-140 but mine is 204 is there anything to worry about??
Hi Shaina,
Thanks for your comment.
Raised protein C could be a reactive finding, I would suggest repeating the test after 3 months and checking your Antiphospholipid screen.
BW,
My 7 year old was recently hospitalized for elevated ESR and chronic abdominal pain + diarrhea. Ultimately, she was diagnosed with Crohn’s after many scans, scopes, and labs. She is currently on entera nutrition via NG tube. After a 10 day inpatient stay at Children’s Hospital of Philadelphia, we received lab results that seem concerning to me but do not have her follow up appt for iron infusion and med treatment until April 11th. Here is what I received:
IgG g4 427
GGT 107
ANA positive
ANA pattern homogeneous
ANA Ab titer 1.64
ALT 45
AST 85
RBC 3.53
HGB 8.7
HCT 28.7
RDW 18.5
Platelets 560
MPV 8.8
ESR 109
Atypical Lymphocyte % 0.0
CD3 +/CD8 + % 16.3
CD20+ % 31.9
CD20+ abs 565
Eos % 7.2
Abs Eos 1210
Abs Neut 14,810
Metamyelocytes % .9
Anisocytosis +1
Poikilocytosis +1
Morphology present
Giant platelets present
There are other results that were not within the standard range as well, but did not concern me AS much…
She is anemic (microcytic) so those labs are also out of range – hence the iron infusions every three days.
My concern is that there is something else wrong that hasn’t been brought up yet and I would like to ask the right questions or make plausible suggestions at her next appointment. Any advice would be much appreciated! Thank you.
Hi Heather,
Thank you for your comment and wishing your daughter gets well soon.
In patients with inflammatory bowel disease, the etiology of anemia is often multifactorial.
Various causes include iron deficiency, anemia of inflammation/chronic disease, vitamin deficiencies (e.g. B12 and Folate), hemolysis, or myelosuppressive effect of drugs.
Her thrombocytosis is likely reactive to blood loss and inflammation.
The rise in her white cell count is likely reactive to infection/inflammation.
I hope the treatment of her Crohn’s disease and the replacement of iron and other vitamin deficiencies would improve her ESR and abnormal blood indices.
BW,
Hello Dr. Abdou,
My 4 yo son has been having some health issues and recently had lab work that seems concerning. His ESR is 20, ANC is 0.6 and his RBC was noted to be abnormal due to the presence of ‘burr cells, microcytes and anisocytosis.’ This is following high fever 104-105 of unknown origin for 8+ days. Could this be due to viral illness? Would you suggest additional studies to look for certain diagnoses? This is the third FUO this year for him.
Hi,
Yes, neutropenia could complicate viral infections, but it should be transient.
Microcytosis may be secondary to iron deficiency or a hemoglobinopathy.
I would suggest initially a repeat FBC, a blood film, serum iron/B12/folate, and CRP.
BW,
I have a 5 month old girl who had low platelets at birth because of my ITP. Her platelet count finally went to normal at 2 months of age. However, her ANC was at 880 at her 4 month appointment. We just had a recheck, and her ANC is now at 900 and platelets are back down to 94. Should we be concerned?
Hi KJ,
It sounds that your daughter’s neutropenia and thrombocytopenia are immune.
I would suggest a blood film and the autoantibody screen including ANCA.
I would also suggest checking her thyroid functions.
BW,
Recently hospitalized for dehydration due to CMPA. Baby had some vomiting and diaherrea. Neutrophils came back very low. platelets and monocytes slightly elevated. Smudge cells were seen. Is this something to worry about? Specifically do smudge cells strongly suggest Leukemia?
Hi Faith,
Neutropenia (low neutrophil count) could complicate viral infections, but it should be transient.
Although smudge cells are classically a feature of chronic lymphocytic leukaemia (CLL), several other conditions may result in smudge cells on a blood film like infectious mononucleosis or incorrect storage of the blood sample. I would suggest a repeat CBC after complete recovery from the current infection.
BW,
Hi we have a 15 month old who went in for her routine 12 month check up and 3 weeks later they call us saying her atypical cells was slightly elevated per the nurses words and her mch I think it was slightly high per the nurse well the dr finally called said very few atypical lymphocytes was there. Recheck was done this past friday she said still very few there nothing to worry about that everything else looked great but i cant help but worry she says its normal for a few to be there is that correct and any ideas why they are there as she hasnt ever been sick in her life. Thank you so so much also iron was good and anemia she said.
Hi Alisha,
Thank you for your comment.
Atypical lymphocytes are quite common in this age group and could represent a reaction to a recent infection, particularly a viral infection.
However, you can send me the full blood count result to have a look.
BW,
My 7-month-old ex-36-week son is being worked up for growth issues. He was born around the 30%ile for weight and height, and ~50%ile for head circumference. He is now <1%ile for weight and height, and his head circumference remains ~50%ile. He is otherwise developing normally. Thus far the work-up has been normal, including CMP, amylase/lipase, CK, and IGF-1.
He has had 2 CBCs with manual differentials about 3 weeks apart; he was not particularly sick at either time. He does go to daycare, though, so it's possible something was brewing. Both CBCs showed thrombocytosis (608K and 618K) as well as leukocytosis (16.8K and 13.7K). Hgb/Hct were normal, as was RBC morphology. On manual differential, he has giant platelets and 1+ metamyelocytes. His WBC are notable for lymphocytic predominance (70%), improving on the second test (58%), and also smudge cells. Could any of this be related to his poor growth? Does he need to see hematology?
Hi LR,
Thank you for your comment.
Thrombocytosis and leukocytosis could be reactive to an underlying infection or inflammation and they should revert back to normal after treating the underlying cause.
However, if they persist after a couple of weeks you should seek advice from a pediatric hematologist.
BW,
ESR and CRP were also normal.
We went in to my daughters 1 year old check up with a fall off of the growth scale (pt was not eating solids yet very well so still heavy breast milk intake) so labs were drawn resulted abnormals were 10/21/22 (2 weeks after an ear infection) WBC 17.6, platelets 593, anion gap 17.8, creatinine 0.15, calcium 10.6, albumin 4.9, A/G ratio 2.6, alkaline phos 295, AST 47, Anisocytosis 1+, Tear Drop Cells 1+, platelet estimate- Increased. We were referred to a pediatric hematologist who said an appt wasn’t needed and to start on iron – repeat labs were ordered after a month but we had to wait until pt was healthy again.
repeat labs on 1/20/23 – after starting on Mary Ruths multivitamin with iron- CBC and CRP were drawn with abnormals being Hgb 10.6 when previously 12.8, MCH 23.5, Platelets 480, lymphocytes 37%, metamyelocytes 1%, ANC 7.8
PA stated she was unconcerned about the drop in the Hgb even after starting a multivitamin. Of course i happen to work in a cancer center so my mind wonders terribly! Of course googling tear drop cells is terrible. Any insight is appreciated as to whether I need to maybe request another redraw this summer. Pt has a history of multiple ear infections (2nd set of tubes done 1/20 after labs were drawn)and fevers. Labs drawn on 1/20 were specifically drawn during a period when she was healthy.
Hi Megan,
Thank you for your comment.
I suggest checking her CRP and the hemolysis screen (reticulocytes, total and conjugated bilirubin, LDH, and DCT). Check her MCV, MCH, and MCHC as well.
BW,
My 2 year old had blood work done and a few came back not normal, we did the blood test because when they were checking her for celiac(which they don’t think she has) they couldn’t do the correct test because her immune system test said her immune system was too low. The allergist went for this test to look at her immune system. She has a few other issues including congenital hypothyroidism as well as perioral cyanosis which they have never been able to figure out why. The levels that came back flagged were platelets 417, hemoglobin 11.4 , basophilis absolute manual .11 burr cell said few, total cells said 118, seg neutrophils absolute was borderline at 4.99. Any thy thoughts on this?
Hello Jacquelyne,
I appreciate your comment.
I sincerely hope for your daughter’s swift recovery.
Her blood counts appear within acceptable ranges, possibly reacting to an underlying infection or inflammation.
While there’s no immediate cause for concern, it’s crucial for her immune suppression to be thoroughly investigated by her pediatrician or immunologist.
Sending my best wishes.
Dr. M Abdou
My 16 year old daughter currently having blood rash when she itches her skin ot all time 3 eposodes now she does itch quite bad than could cause friction anyways doctor did full blood count all came back normal apart from her clotting test 2 of these were above avarage levels they said no further action unless more rashes have a appeared which have I’ve requested them to refer her for more test as they wasn’t going to should I be worried ?
Hi Christina,
Thank you for your comment.
It is not clear from your message what the nature of your daughter’s “blood rash” is.
Does she bruise easily?
Is there any spontaneous bleeding?
Which clotting tests were abnormal?
Could you please provide more information via email at in**@*************st.com?
Best wishes,
Dr. M Abdou
I have an 8 month old who presented with a sudden and rapid growing lump on the back of his neck. The lump was small and not visable to the eye and 3 days later had tripled in size and is now visible and has MILD blue, spotted discoloration. An ultrasound of the mass indicated it was actually 2 masses that appeared to be lymph nodes measuring at 15x6x12 mm. CT imaging got partial visualization and indicated it was possibly a vascular anomaly. Rbc and wbc seem normal but he does have elevated atypical lymph (11%) and blood smear showed polychrom 1+, ovalocytes 1+, tear drop cells 1+, poikilocytosis 1+, anisocytosis 1+, and smudge cells at 21.4. His ped seems unconcerned because the rbc and wbc are clear and i am unsure who to seek referrals for and what the concern may be.
Hi Alexandra,
Thank you for reaching out regarding your concern about your 8-month-old son’s rapidly growing neck lump, which could potentially be related to lymph nodes or a vascular anomaly.
While I appreciate your query, my expertise primarily lies in interpreting blood film findings.
The presence of atypical lymphocytes and other findings on the blood film are a common occurrence following recent viral infections.
These atypical cells typically resolve once the body successfully combats the infection and is not usually a cause for concern.
However, it’s crucial to address your son’s neck lump with a pediatric surgeon or pediatrician for a thorough evaluation and appropriate management.
Best wishes,
Dr. M. Abdou
Also, he had a cold end of March that had cleared by April 3rd and the mass was discovered April 13th and has still not receded
What are your thoughts regarding the elevated platelet count in a 2 month old female with history down syndrome and was not sick at the time the labs were drawn?
F WHITE BLOOD CELL COUNT 5.3 5.0-19.5 (Thousand/uL) TP
F RED BLOOD CELL COUNT 3.76 3.10-5.30 (Million/uL) TP
F HEMOGLOBIN 12.3 9.1-14.0 (g/dL) TP
F HEMATOCRIT 35.0 28.0-42.0 (%) TP
F MCV 93.1 91.0-112.0 (fL) TP
F MCH 32.7 27.0-36.0 (pg) TP
F MCHC 35.1 28.0-36.0 (g/dL) TP
For adults, a slight decrease in the calculated MCHC
value (in the range of 30 to 32 g/dL) is most likely
not clinically significant; however, it should be
interpreted with caution in correlation with other
red cell parameters and the patient’s clinical
condition.
F RDW 11.8 11.5-16.0 (%) TP
F PLATELET COUNT 441 H 150-400 (Thousand/uL) TP
F MPV 10.4 7.5-12.5 (fL) TP
F ABSOLUTE NEUTROPHILS 1293 1000-8800 (cells/uL) TP
F ABSOLUTE LYMPHOCYTES 3503 3300-15000 (cells/uL) TP
F ABSOLUTE MONOCYTES 366 200-1400 (cells/uL) TP
F ABSOLUTE EOSINOPHILS 58 15-700 (cells/uL) TP
F ABSOLUTE BASOPHILS 80 0-250 (cells/uL) TP
F NEUTROPHILS 24.4 (%) TP
F LYMPHOCYTES 66.1 (%) TP
F MONOCYTES 6.9 (%) TP
F EOSINOPHILS 1.1 (%) TP
F BASOPHILS 1.5 (%) TP
Hello Danie,
Thank you for reaching out.
An elevated platelet count (thrombocytosis) of 441 × 10³/μL in a 2-month-old with Down syndrome, in the absence of illness, is not uncommon.
Thrombocytosis in infants with Down syndrome can be reactive, often due to inflammation, iron deficiency (even when subtle or subclinical), or bone marrow hyperactivity, which can occur in this population.
Follow-up with serial counts is recommended to monitor for trends.
Best wishes,
Dr M Abdou