Hemophilia
Hemophilia is an inherited X-linked bleeding disorder caused by deficiency or dysfunction of clotting factor VIII (hemophilia A) or factor IX (hemophilia B). The impaired coagulation results in prolonged or spontaneous bleeding, particularly into joints and muscles. Hemophilia primarily affects males, while females are typically carriers. The term hemophilia is the standard international spelling (also rendered as haemophilia in the UK and hémophilie, hemofilie, hemofilia, hämophilie, or emofilia in other languages); for consistency, we use hemophilia throughout this section.
There are 13 known clotting factors that work with platelets, small blood cells formed in the bone marrow, to form a stable clot. According to the World Federation of Hemophilia (WFH), approximately one in 10,000 individuals is born with this rare condition. Hemophilia results from genetic mutations, deletions, or inversions involving the factor VIII gene (Hemophilia A) or factor IX gene (Hemophilia B), both located on the X chromosome. Consequently, the disorder predominantly affects males, while daughters of affected men are obligate carriers. Each son of a carrier mother has a 50% chance of having hemophilia, and each daughter a 50% chance of being a carrier.
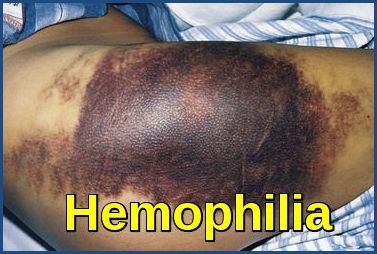
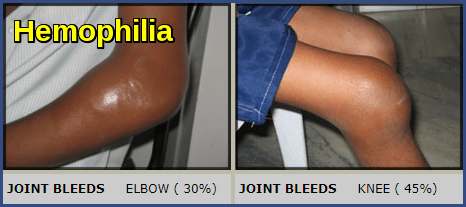
Clinically, patients experience easy bruising, delayed clotting, spontaneous bleeding, and hemarthrosis—bleeding into joints that can cause pain, swelling, and long-term damage. Without appropriate management, hemophilia can lead to severe or even life-threatening complications.
The three forms of hemophilia are hemophilia A, B, and C.
Hemophilia A is the most common type of hemophilia, and it’s caused by a deficiency in factor VIII. According to the National Heart, Lung, and Blood Institute (NHLBI), eight out of 10 people with hemophilia have hemophilia A.
Hemophilia B, which is also called Christmas disease, is caused by a deficiency of factor IX.
Hemophilia C is a mild form of the disease that’s caused by a deficiency of factor XI. People with this rare type of hemophilia often don’t experience spontaneous bleeding. Hemorrhaging typically occurs after trauma or surgery. Hemophilia C is primarily an inherited disorder, but unlike hemophilia A and B, the inheritance of hemophilia C follows an autosomal recessive pattern. The gene that causes factor XI deficiency is not present on a sex chromosome and the condition, therefore, affects both genders equally.
Hemophilia is not curable, but it can be treated to minimize symptoms and prevent future health complications.
In extremely rare cases, hemophilia can develop after birth. This is called “acquired hemophilia”. This is the case in people whose immune system forms antibodies that attack factors VIII or IX. The patient develops the condition during his/her lifetime and it does not have a genetic or heritable cause. It occurs when the body forms antibodies that attack one or more blood clotting factors, (usually factor VIII), thus preventing the blood clotting mechanism from working properly. Patients may be male or female and the pattern of bleeding is rather different from that of classical hemophilia, the joints being rarely affected. The disorder is particularly associated with old age and occasionally complicates pregnancy.
Hemophilia A (factor VIII deficiency), which affects about 80% of patients with hemophilia, and hemophilia B (factor IX deficiency) have identical clinical manifestations and screening test abnormalities. Both are X-linked genetic disorders. Specific factor assays are required to distinguish the two.
Pathophysiology:
Normal hemostasis requires > 30% of normal factor VIII and IX levels. Most patients with hemophilia have levels < 5%; some have extremely low levels (< 1%). The functional level (activity) of factor VIII or IX in hemophilia A and B, and thus bleeding severity, varies depending on the specific mutation in the factor VIII or IX gene.
Carriers usually have levels of about 50%; rarely, random inactivation of their normal X chromosome in early embryonic life results in a carrier having factor VIII or IX levels of < 30%.
In the 1960s, drug companies manufactured factor concentrates derived from human blood, and the world changed for people living with hemophilia. They could inject themselves at home, and avoid horrible pain and hospitalizations. Modern medicine transformed hemophilia from a fatal disease to a chronic condition and patients began to lead nearly normal lives.
Sadly, this “miracle” product had unseen consequences. Each dose of factor concentrate was made by pooling 60,000 individual blood donations. Because the process needed many donors to manufacture, people were paid to donate clotting factors. Collection centers were set up in poor neighborhoods, places more likely to attract potential donors who might need extra cash. People needing cash to support their kids or their drug habit were frequent donors. Blood was also used from prisoners.
Patients needing clotting factors were at enormous risk of receiving contaminated blood products, and viral hepatitis infection was nearly universal. Many experts thought that hepatitis an “acceptable risk” for these patients, and patients were rarely warned.
Hepatitis may have been considered an “acceptable risk,” but HIV and AIDS were not. In 1982, the Centers for Disease Control and Prevention (CDC) announced three cases of apparent AIDS in hemophiliacs who had received clotting factors. It was a year after the first case of AIDS had been reported in anyone, and by this point, there were already 471 cases of immune suppression, including 184 deaths.
Occasional patients developed immune thrombocytopenia secondary to HIV infection, which exacerbated bleeding.
Clinical picture:
Patients with hemophilia bleed into tissues (eg, hemarthroses, muscle hematomas, retroperitoneal hemorrhage). The bleeding may be immediate or occur slowly, depending on the extent of trauma and plasma level of factor VIII or IX.
Pain often occurs as bleeding commences, sometimes before other signs of bleeding develop. Chronic or recurrent hemarthroses can lead to synovitis and arthropathy.
Even a trivial blow to the head can cause intracranial bleeding. Bleeding into the base of the tongue can cause life-threatening airway compression.
Severe hemophilia (factor VIII or IX level < 1% of normal) causes severe bleeding throughout life, usually beginning soon after birth (eg, scalp hematoma after delivery or excessive bleeding after circumcision). Moderate hemophilia (factor levels 1 to 5% of normal) usually causes bleeding after minimal trauma. In mild hemophilia (factor levels 5 to 25% of normal), excessive bleeding may occur after surgery or dental extraction.
Diagnosis:
Many people who have or have had family members with hemophilia will ask that their baby boys get tested soon after birth.
About one-third of babies who are diagnosed with hemophilia have no other family members with the disorder. A doctor might check for hemophilia if a newborn is showing certain signs of hemophilia.
Diagnosis includes screening tests and clotting factor tests. Screening tests are blood tests that show if the blood is clotting properly. Clotting factor tests, also called factor assays, are required to diagnose a bleeding disorder. This blood test shows the type of hemophilia and the severity.
Families With a History of Hemophilia
Any family history of bleeding, such as following surgery or injury, or unexplained deaths among brothers, sisters, or other male relatives such as maternal uncles, grandfathers, or cousins should be discussed with a doctor to see if hemophilia was a cause. A doctor often will get a thorough family history to find out if a bleeding disorder exists in the family.
Many people who have or have had family members with hemophilia will ask that their baby boys get tested soon after birth. In the best of cases, testing for hemophilia is planned before the baby’s delivery so that a sample of blood can be drawn from the umbilical cord immediately after birth and tested to determine the level of the clotting factors. Umbilical cord blood testing is better at finding low levels of factor VIII (8) than it is at finding low levels of factor IX (9). This is because factor IX levels take more time to develop and are not at a normal level until a baby is at least 6 months of age. Therefore, a mildly low level of factor IX at birth does not necessarily mean that the baby has hemophilia B. A repeat test when the baby is older might be needed in some cases.
Families With No Previous History of Hemophilia
About one-third of babies who are diagnosed with hemophilia have no other family members with the disorder. A doctor might check for hemophilia in a newborn if:
- Bleeding after circumcision of the penis goes on for a long time.
- Bleeding goes on for a long time after drawing blood and heel sticks (pricking the infant’s heel to draw blood for newborn screening tests).
- Bleeding in the head (scalp or brain) after a difficult delivery or after using special devices or instruments to help deliver the baby (e.g., vacuum or forceps).
- Unusual raised bruises or large numbers of bruises. If a child is not diagnosed with hemophilia during the newborn period, the family might notice unusual bruising once the child begins standing or crawling.
Those with severe hemophilia can have serious bleeding problems right away. Thus, they often are diagnosed during the first year of life. People with milder forms of hemophilia might not be diagnosed until later in life.
Screening Tests
Screening tests are blood tests that show if the blood is clotting properly. Types of screening tests:
Full Blood Count (FBC)
This common test measures the amount of hemoglobin, the size and number of red blood cells and numbers of different types of white blood cells and platelets found in blood. The FBC is normal in people with hemophilia. However, if a person with hemophilia has unusually heavy bleeding or bleeds for a long time, the hemoglobin and the red blood cell count can be low.
Activated Partial Thromboplastin Time (APTT) Test
This test measures how long it takes for blood to clot. It measures the clotting ability of factors VIII (8), IX (9), XI (11), and XII (12). If any of these clotting factors are too low, it takes longer than normal for the blood to clot. The results of this test will show a longer clotting time among people with hemophilia A, B or C.
Prothrombin Time (PT) Test
This test also measures the time it takes for blood to clot. It measures primarily the clotting ability of factors I (1), II (2), V (5), VII (7), and X (10). If any of these factors are too low, it takes longer than normal for the blood to clot. The results of this test will be normal among most people with hemophilia A, B, and C.
Thrombin Time (TT) Test
TT is normal in hemophilia.
Fibrinogen Test
This test also helps doctors assess a patient’s ability to form a blood clot. This test is ordered either along with other blood clotting tests or when a patient has an abnormal PT or APTT test result, or both. Fibrinogen is another name for clotting factor I (1).
Clotting Factor Tests
Clotting factor tests XIII, IX, and XI, also called factor assays, are required to diagnose a bleeding disorder. This blood test shows the type of hemophilia and the severity. It is important to know the type and severity in order to create the best treatment plan. Because factor VIII levels may also be reduced in von Willebrand disease (VWD), von Willebrand factor (VWF) activity, antigen, and multimer composition are measured in patients with newly diagnosed hemophilia A, particularly if the disorder is mild and a family history indicates that both male and female family members are affected.
Determining if a female is a true carrier of hemophilia A is sometimes possible by measuring the factor VIII level. Similarly, measuring the factor IX level often identifies a carrier of hemophilia B. PCR analysis of DNA that comprises the factor VIII gene, available at specialized centers, can be used for diagnosis of the hemophilia A carrier state and for prenatal diagnosis of hemophilia A by chorionic villus sampling at 12 wk or amniocentesis at 16 wk. These procedures carry a 0.5 to 1% risk of miscarriage.
After repeated exposure to factor VIII replacement, about 15 to 35% of patients with hemophilia A develop factor VIII isoantibodies (alloantibodies) that inhibit the coagulant activity of any additional factor VIII infused. Patients should be screened for isoantibodies (eg, by measuring the degree of PTT shortening immediately after mixing the patient’s plasma with an equal volume of normal plasma, and then by repeating the measurement after incubation for 1 h), especially before an elective procedure that requires replacement therapy. If isoantibodies are present, their titers can be measured by determining the extent of factor VIII inhibition by serial dilutions of patient plasma.
Treatment:
The main treatment for hemophilia is called replacement therapy. Concentrates of clotting factor VIII (for hemophilia A) or clotting factor IX (for hemophilia B) are slowly dripped or injected into a vein. These infusions help replace the clotting factor that’s missing or low.
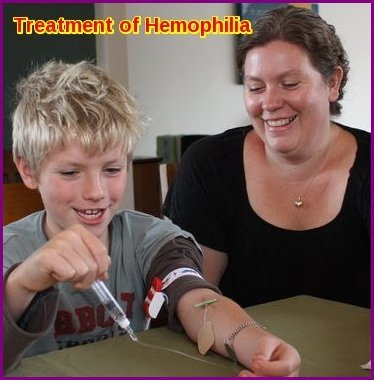
Prophylaxis through regular home-based factor infusion is the gold standard for preventing joint bleeds.
Clotting factor concentrates can be made from human blood. The blood is treated to prevent the spread of diseases, such as hepatitis. With the current methods of screening and treating donated blood, the risk of getting an infectious disease from human clotting factors is very small.
To further reduce the risk, you or your child can take clotting factor concentrates that aren’t made from human blood. These are called recombinant clotting factors. Clotting factors are easy to store, mix, and use at home—it only takes about 15 minutes to receive the factor.
You may have replacement therapy on a regular basis to prevent bleeding. This is called preventive or prophylactic therapy. Or, you may only need replacement therapy to stop bleeding when it occurs. This use of the treatment, on an as-needed basis, is called demand therapy.
Demand therapy is less intensive and expensive than preventive therapy. However, there’s a risk that bleeding will cause damage before you receive the demand therapy.
Complications of replacement therapy include:
- Developing antibodies that attack the clotting factor.
- Developing viral infections from human clotting factors.
- Damage to joints, muscles, or other parts of the body resulting from delays in treatment.
If symptoms suggest bleeding, treatment should begin immediately, even before diagnostic tests are completed. For example, treatment for a headache that might indicate intracranial hemorrhage should begin before CT is completed.
Replacement of the deficient factor is the primary treatment. In hemophilia A, the factor VIII level should be raised transiently to:
- 30% of normal to prevent bleeding after a dental extraction or to abort an incipient joint hemorrhage.
- 50% of normal if severe joint or intramuscular bleeding is already evident.
- 100% of normal before major surgery or if bleeding is intracranial, or otherwise life-threatening.
Repeated infusions at 50% of the initial calculated dose should then be given every 8 to 12 hours to keep trough levels > 50% for 7 to 10 days after major surgery or life-threatening hemorrhage. Each unit/kg of factor VIII increases the factor VIII level by about 2%. Thus, to increase the level from 0% to 50%, about 25 units/kg are required.
Factor VIII can be given as purified factor VIII concentrate, which is derived from multiple donors. It undergoes viral inactivation, but inactivation may not eliminate parvovirus or hepatitis A virus. Recombinant factor VIII is free of viruses and is usually preferred unless patients are already seropositive for HIV or for hepatitis B or C virus.
In hemophilia B, factor IX can be given as a purified or recombinant viral-inactivated product every 24 h. The target levels of factor correction are the same as in hemophilia A. However, to achieve these levels, the dose must be higher than in hemophilia A because factor IX is smaller than factor VIII and, in contrast to VIII, has an extensive extravascular distribution. The formula to determine units required to obtain desired factor IX level:
Number of factor IX units required = patient weight (in kg) x desired factor IX level increase (as % or units/dL) x 1 unit/kg
For example, to attain an 80% level in a 70 kg patient who has a baseline level of 20%: Number of factor IX units needed = 70 kg x 60% x 1 unit/kg = 4200 units.
Note: If the patient has severe hemophilia (ie, baseline factor IX level is or presumed to be <1%), then may just use “desired factor IX level” instead of “desired factor IX level increase”.
In Hemophilia C, factor XI concentrate is available in lyophilized (powder) form in glass containers. It is readily administered with a small volume of liquid. Only very rarely will patients develop allergic reactions to the product. On the other hand, factor XI concentrates are known to pose a risk of promoting development of intra-vascular clots, which, though rarely, can endanger a patient’s life.
Each International Unit (IU) of factor XI per kilogram increases the concentration of factor XI in the blood by 2.4%; the level of blood factor drops by half every two days, until it returns to the base level.
When factor XI proves to be the treatment of choice to prevent or treat bleeding, it is essential that it be closely supervised by a team of experts in a hemophilia treatment centre.
Fresh frozen plasma contains factors VIII and IX. However, unless plasma exchange is done, sufficient whole plasma usually cannot be given to patients with severe hemophilia to raise factor VIII or IX to levels that prevent or control bleeding. Fresh frozen plasma should, therefore, be used only if rapid replacement therapy is necessary and factor concentrate is unavailable or the patient has a coagulopathy that is not yet defined precisely.
A recombinant factor VIII-Fc fusion protein, as well as a recombinant factor IX-Fc fusion protein and a pegylated recombinant factor IX, with longer in vivo survival times have recently been reported to successfully control bleeding in hemophilia A and B.
In patients with hemophilia who develop a factor VIII inhibitor, treatment is best accomplished using recombinant activated factor VII (VIIa) in repeated high doses (eg, 90 mcg/kg).
Both VWF and factor VIII are stored in the endothelial cells, and secreted in response to endothelial cell stimulation. Adjunctive therapy for hemophilia A thus includes in vivo stimulation of patient endothelial cells with the synthetic vasopressin analogue DDAVP (deamino-D–arginine vasopressin, also known as Desmopressin).
Desmopressin may temporarily raise factor VIII (and VWF) levels. The patient’s response should be tested before desmopressin is used therapeutically. Its use after minor trauma or before elective dental surgery may obviate replacement therapy. Desmopressin should be used only for patients with mild hemophilia A (basal factor VIII levels ≥ 5%) who have demonstrated responsiveness.
An antifibrinolytic agent (aminocaproic acid 2.5 to 4 g po qid for 1 week or tranexamic acid 1.0 to 1.5 g po tid or qid for 1 week) may also be used as adjunctive therapy in hemophilia A or B to prevent late bleeding after dental extraction or other oropharyngeal mucosal trauma (eg, tongue laceration).
Prevention:
Patients should avoid aspirin and NSAIDs (both inhibit platelet function).
Regular dental care is essential so that tooth extractions and other dental surgery can be avoided.
Drugs should be given orally or IV; intramuscular injections can cause hematomas and should be avoided.
Patients with hemophilia should be vaccinated against hepatitis B.
Summary:
Hemophilia is a rare, inherited bleeding disorder characterized by a deficiency or dysfunction of specific clotting factors—primarily Factor VIII (Hemophilia A) or Factor IX (Hemophilia B). Because the genes for these factors are located on the X chromosome, the condition predominantly affects males, while females typically serve as carriers.
Clinical presentation often includes easy bruising, prolonged bleeding after minor trauma, and hemarthrosis (bleeding into joints), particularly affecting the knees and elbows. Diagnosis is confirmed through specialized screening tests like APTT and definitive clotting factor assays to determine the exact severity—ranging from mild (>5%) to severe (<1%). While there is currently no cure, modern management through prophylactic replacement therapy (using recombinant or plasma-derived factors) has transformed hemophilia from a life-threatening disease into a manageable chronic condition, allowing patients to lead near-normal lives.
Over the past decade, the management of hemophilia has evolved significantly with the introduction of novel hematology drugs that extend beyond traditional factor replacement therapy. Recombinant factor VIII products such as Afstyla (lonoctocog alfa), Kovaltry (octocog alfa), and Nuwiq (simoctocog alfa), along with extended half-life factor IX agents including Idelvion (albutrepenonacog alfa) and Rebinyn (nonacog beta pegol), have improved prophylactic strategies by reducing infusion frequency while maintaining effective hemostasis. In parallel, non-factor therapies have transformed care, most notably Hemlibra (emicizumab), a bispecific monoclonal antibody that mimics factor VIII activity and has demonstrated substantial reductions in bleeding rates, even in patients with inhibitors. These advances, together with ongoing developments in gene therapy and rebalancing agents, are reshaping the therapeutic landscape, offering more convenient, effective, and individualized treatment approaches that improve long-term outcomes and quality of life for patients with hemophilia.
Questions and Answers:
What causes hemophilia?
Hemophilia is caused by inherited deficiency of clotting factor VIII (hemophilia A) or factor IX (hemophilia B), most often due to mutations in the respective gene on the X chromosome, leading to impaired blood clotting and increased bleeding risk.
What are the main symptoms of hemophilia?
The primary symptoms of hemophilia include prolonged bleeding after injury, spontaneous bleeding into joints (hemarthrosis), muscle bleeds, easy bruising, and prolonged bleeding after surgery or dental procedures.
How is hemophilia diagnosed?
Diagnosis of hemophilia is based on clinical history and characteristic bleeding, prolonged activated partial thromboplastin time (aPTT), and specific clotting factor assays showing low factor VIII or IX activity levels.
What are the differences between hemophilia A and B?
Hemophilia A is caused by factor VIII deficiency and is more common, while hemophilia B is due to factor IX deficiency. Both conditions present similarly, but they involve different clotting factors and genetic mutations.
How is hemophilia treated?
Treatment centers on replacement of the deficient clotting factor using factor VIII or IX concentrates, prophylactic therapy to prevent bleeds, and supportive care such as antifibrinolytic agents. Emerging therapies include extended half-life products and non-factor agents like bispecific antibodies.
Can people with hemophilia lead a normal life?
With modern management including regular prophylaxis, prompt treatment of bleeds, safe activity planning, and multidisciplinary care, many people with hemophilia can maintain a high quality of life and engage safely in daily activities.
What complications can arise from hemophilia?
Frequent joint bleeds can lead to chronic arthropathy and disability. Other complications include target joints, muscle compartment syndrome, intracranial hemorrhage, and inhibitor development to factor replacement therapy.
Is hemophilia inherited?
Yes, hemophilia is usually inherited in an X-linked pattern, typically affecting males, while females are carriers. Genetic counseling can help at-risk families understand inheritance and testing options.
What is hemarthrosis in hemophilia?
Hemarthrosis is bleeding into a joint space, commonly affecting knees, elbows, or ankles in hemophilia patients. It causes swelling, pain, and restricted range of motion, and recurrent episodes can lead to joint degeneration.
References:
Blanchette V.S., Key N.S., Ljung L.R., Manco-Johnson M.J., van den Berg H.M., Srivastava A. Definitions in Hemophilia: Communication from the SSC of the ISTH Subcommittee on Factor VIII, Factor IX, and Rare Coagulation Disorders. Journal of Thrombosis and Haemostasis. 2014;12(11):1935–1939. doi:10.1111/jth.12772.
Srivastava A., Santagostino E., Dougall A., et al. WFH Guidelines for the Management of Hemophilia, 3rd edition. Haemophilia. 2020;26(S6):1-158. doi:10.1111/hae.14046. ← (Strongly recommended addition)
Centers for Disease Control and Prevention (CDC). Diagnosis and Management of Hemophilia. National Center on Birth Defects and Developmental Disabilities.
National Heart, Lung, and Blood Institute (NHLBI), NIH. How Is Hemophilia Treated?
BMJ Best Practice. Hemophilia – Clinical Overview and Management.
Merck Manual Professional Edition. Hemophilia – Hematology and Oncology Section.
Lewandowska M, Nasr S, Shapiro AD. Emerging therapies in hemophilia: improving equitable access to care. Haemophilia. 2025. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC11849425/
Shapiro AD, Pipe SW, et al. Emicizumab prophylaxis in hemophilia A (HAVEN clinical trials): significant reduction in bleeding rates and improved outcomes. N Engl J Med. 2017–2018. Summary available from: https://en.wikipedia.org/wiki/Emicizumab
European Haemophilia Consortium. Novel treatments in haemophilia and other bleeding disorders: periodic review. 2023. Available from: https://haemophilia.ie/wp-content/uploads/2023/02/Novel-Treatments-in-Haemophilia-Other-Bleeding-Disorders-A-Periodic-Review-2023-Issue-1.pdf
Pipe SW, Shapiro AD, et al. Advances in hemophilia A therapy including bispecific antibodies and novel prophylactic agents. EHC Novel Treatment Review. 2025. Available from: https://academy.ehc.eu/wp-content/uploads/2025/06/NTR-January-2025-final.pdf
World Federation of Hemophilia. Guidelines for the management of hemophilia, 3rd edition. 2020. Available from: https://www1.wfh.org/publications/files/pdf-1863.pdf
Keywords:
Hemophilia, hemophilia A, hemophilia B, inherited bleeding disorder, factor VIII deficiency, factor IX deficiency, hemophilia symptoms, hemophilia diagnosis, hemophilia treatment, hemarthrosis, hemophilic arthropathy, acquired hemophilia, prophylaxis in hemophilia, gene therapy for hemophilia, female carriers hemophilia, bleeding disorder complications, new hematology drugs, hemophilia drugs, recombinant factor VIII, recombinant factor IX, extended half life factor VIII, extended half life factor IX, emicizumab Hemlibra, non factor therapy hemophilia, bypassing agents hemophilia, inhibitors in hemophilia, hemophilia management guidelines, hemophilia prophylaxis treatment, severe hemophilia treatment, mild hemophilia management, hemophilia A treatment options, hemophilia B treatment options, CAR T cell therapy hematology (contextual internal link support), targeted therapy hematology, bleeding disorders treatment advances

Request Online Consultation With Dr M Abdou
Fee: US$100
Secure payment via PayPal (credit and debit cards accepted)
Pay Now




Are your images of hemophilia copyrighted? Is it possible to get copyright permission from your organization for use.
Hello,
Thank you for your request.
You are free to share, copy and redistribute the material in any medium or format as long as you will include our article link as a source according to our creative commons license:
https://creativecommons.org/licenses/by-sa/2.0/
Regards,
Recently diagnosed with type A, two major bleeds, legs and arm. Painful and debilitating. I’m 65 and yet to get some treatment. Why would that be?
Hello,
Thank you for your message.
What are your factor 8 level and your coagulation profile?