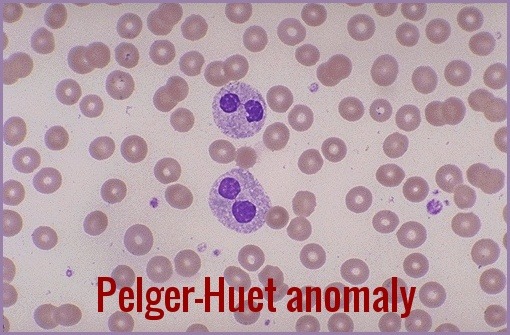
Pelger-Huët Anomaly
Pelger-Huët Anomaly (PHA) is a rare, typically benign hereditary disorder of neutrophil nuclear segmentation, characterised by bilobed or peanut-shaped nuclei instead of the normal multilobed appearance. This distinctive morphology affects neutrophils and other granulocytes and is often discovered incidentally on a peripheral blood smear. Although most individuals with true (inherited) PHA remain asymptomatic with normal immune function, similar morphological changes—termed pseudo–Pelger-Huët anomaly—can also arise in association with myelodysplastic syndromes, severe infections, medications, or other underlying haematological conditions, making accurate differentiation clinically important.
Pelger-Huët anomaly is caused by a mutation in the LBR (lamin B receptor) gene, which encodes a protein involved in the nuclear envelope structure and function. The mutation leads to abnormal nuclear shape and chromatin organization in WBCs, but not in other cell types. Pelger-Huët Anomaly is inherited in an autosomal dominant pattern, which means that a person with one copy of the mutated gene (heterozygous) has a 50% chance of passing it on to each of their offspring. However, most cases of PHA are sporadic, meaning that the mutation occurs spontaneously in a person with no family history of the disorder.
The characteristic leukocyte appearance was first reported in 1928 by Pelger, a Dutch hematologist, who described leukocytes with dumbbell-shaped bilobed nuclei, a reduced number of nuclear segments, and coarse clumping of the nuclear chromatin.
In 1931 Huet, a Dutch pediatrician, identified it as an inherited disorder.
Neutrophils contain not more than two lobes to the nuclei (spectacle-shaped nuclei or peanut-shaped nuclei instead of the normal trilobed shape) and “band forms” are frequent. In the inherited anomaly, affected neutrophils with bilobed nuclei makeup 60-90% of the neutrophils seen.
PHA can be diagnosed by examining blood smears under a microscope and identifying the characteristic bilobed or peanut-shaped nuclei of neutrophils and other granulocytes. The anomaly can be quantified by calculating the Pelger-Huët ratio, which is the percentage of neutrophils with abnormal nuclei among all neutrophils counted. A ratio of 10% or higher is considered diagnostic of PHA. However, the diagnosis should be confirmed by genetic testing to exclude other causes of nuclear segmentation abnormalities, such as myelodysplastic syndromes (MDS) or acute myeloid leukemia (AML).
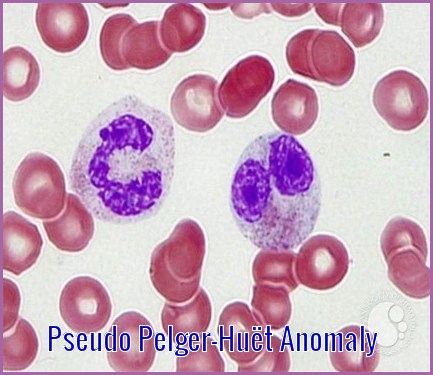
Anomalies resembling Pelger-Huët anomaly that are acquired rather than familial have been described as pseudo Pelger-Huët anomaly (PPHA). The morphologic characteristic seen in pseudo Pelger-Huët anomaly is similar to PHA. The acquired pseudo Pelger-Huët anomaly has been associated with pathologic conditions including acute and chronic myeloid leukemias, myelodysplastic syndrome (MDS), severe infections and some other toxic conditions.
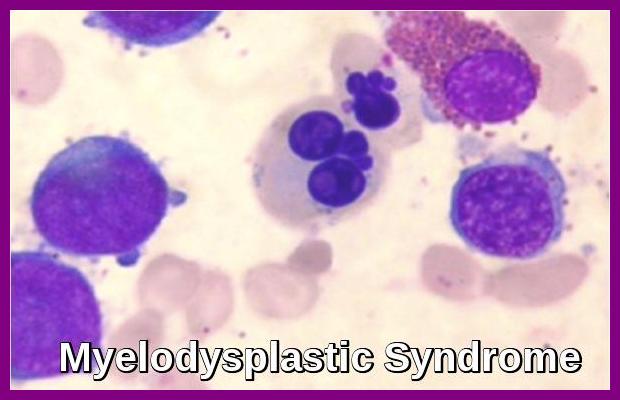
Pseudo Pelger-Huët anomaly and micromegakaryocytes are highly characteristic features of myelodysplastic syndromes and, when present together, are considered strongly suggestive—though not absolutely pathognomonic—of MDS.

Peripheral blood smear showing marked red cell poikilocytosis and a hypogranular neutrophil with a pseudo Pelger-Huët nucleus in a patient with myelodysplastic syndrome.
Usually, the congenital form is not associated with thrombocytopenia and leukopenia, so if these features are present more detailed search for myelodysplasia is warranted, as pseudo-Pelger–Huët anomaly can be an early feature of myelodysplasia.
When Pelger-Huët cells are identified, initially attempt to determine if the patient has a benign inherited anomaly or an acquired morphologic feature (ie, pseudo–Pelger-Huët anomaly). In PPHA, cells may appear morphologically similar to inherited PHA, but the absence of these findings in other family members, a low percentage of affected cells (usually 5-20%), and involvement of other cell lines (eg, anemia or thrombocytopenia) suggest an acquired anomaly.
Summary:
Pelger-Huët anomaly is a distinctive neutrophil nuclear segmentation abnormality characterised by bilobed or hypolobated nuclei that may occur as a benign inherited trait or arise as a pseudo Pelger-Huët anomaly in conditions such as myelodysplastic syndromes, severe infections, and drug exposures. Accurate recognition on the peripheral smear is essential to distinguish the harmless congenital form from acquired changes that may signal underlying haematological disease. This post reviews the causes, morphology, clinical relevance, and diagnostic distinctions between true and pseudo Pelger-Huët anomaly, supported by high-resolution smear images for practical interpretation.
Conclusion:
Pelger-Huët anomaly is generally a benign hereditary condition that does not require treatment or follow-up, as it is not associated with clinical symptoms or impaired neutrophil function. However, the pseudo–Pelger-Huët anomaly (PPA) may occur secondary to underlying medical disorders or exposure to certain medications. PPA is most commonly seen in myelodysplastic syndromes but can also appear in other hematologic conditions, including myeloproliferative neoplasms such as chronic myeloid leukemia (CML) and essential thrombocythemia (ET), as well as in various congenital disorders, including Down syndrome and Alagille syndrome. Therefore, when Pelger-Huët–like changes are identified, clinicians should consider the possibility of an acquired cause and investigate accordingly if clinical suspicion exists.
Questions and Answers:
What is Pelger-Huët anomaly?
Pelger-Huët anomaly is a benign inherited neutrophil nuclear segmentation defect in which neutrophils show bilobed (“pince-nez”) or hypolobated nuclei with normal cytoplasmic granulation and preserved function. It is usually asymptomatic and detected incidentally on a peripheral blood smear.
What is the difference between Pelger-Huët anomaly and pseudo Pelger-Huët anomaly?
The inherited form (PHA) is benign, stable, and non-progressive, whereas pseudo Pelger-Huët anomaly (PPA) is an acquired abnormality often associated with myelodysplastic syndromes, acute leukemia, severe infections, or drug effects. PPA typically shows more pronounced hypogranularity and higher diagnostic significance.
What causes pseudo Pelger-Huët anomaly?
Pseudo Pelger-Huët anomaly is most commonly caused by myelodysplastic syndromes but may also occur in myeloproliferative neoplasms, acute leukemia, severe systemic infections, HIV, and following exposure to medications such as G-CSF, valproate, tacrolimus, and chemotherapeutic agents.
How is Pelger-Huët anomaly diagnosed?
Diagnosis is usually made on a peripheral blood smear showing bilobed or hypolobated neutrophils with mature chromatin. In hereditary cases, the morphology is consistent and benign; in suspected pseudo forms, further evaluation—including CBC trends, bone marrow examination, and cytogenetics—is required to exclude an underlying hematologic disorder.
Does Pelger-Huët anomaly affect immune function?
Inherited Pelger-Huët anomaly does not impair neutrophil function, chemotaxis, or host defence. Patients remain clinically well. In contrast, immune compromise in PPA is related to the underlying condition, such as MDS or leukemia, rather than the nuclear morphology itself.
Can Pelger-Huët anomaly be seen in myelodysplastic syndrome?
Yes. Pseudo Pelger-Huët anomaly is a classic morphological feature of MDS and often appears alongside hypogranularity, cytopenias, and dysplasia in multiple lineages. Its presence should prompt evaluation for underlying marrow pathology.
How can Pelger-Huët anomaly be distinguished from a left shift?
In a left shift, neutrophils show immature forms with less dense chromatin and increased cytoplasmic basophilia. In Pelger-Huët anomaly, the nuclei may be bilobed or unilobed, but the chromatin is fully mature and clumped, helping to avoid misinterpretation as band forms.
Is Pelger-Huët anomaly associated with genetic syndromes?
True PHA is hereditary and linked to mutations in the LBR gene. Pseudo Pelger-Huët morphology has been reported in congenital disorders such as Down syndrome and Alagille syndrome, typically reflecting underlying marrow stress or dysplasia.
Do patients with Pelger-Huët anomaly require treatment or follow-up?
Inherited PHA requires no treatment or follow-up because it is benign and asymptomatic. Pseudo Pelger-Huët anomaly requires investigation and management of the underlying cause, particularly when associated with MDS, leukemia, or medication effects.
Can medications cause pseudo Pelger-Huët anomaly?
Yes. Several medications—including G-CSF, valproate, tacrolimus, mycophenolate, and chemotherapeutic agents—may induce pseudo Pelger-Huët changes. Morphology often returns to normal after discontinuation of the offending drug.
References:
Peter Maslak, Susan McKenzie. Pseudo Pelger-Huët Cells (II) – 1. ASH Image Bank, American Society of Hematology. Available at: https://imagebank.hematology.org/image/1848/pseudo-pelger-huet-cells-ii–1
Bain BJ. Blood Cells: A Practical Guide. 5th ed. Wiley-Blackwell; 2015.
Bux J, Behrens G, Jaeger G, et al. Diagnosis and clinical course of autoimmune neutropenia in infancy: analysis of 240 cases. Blood. 1998;91(1):181–186.
Rao LVM, Gowda V. Pelger-Huët anomaly: a rare inherited anomaly of leukocytes. Blood Cells Mol Dis. 2014;53(4):223–227.
Rosinol L, Cibeira MT, Blade J, et al. Pelger-Huët anomaly in multiple myeloma: a new prognostic factor? Eur J Haematol. 2001;66(6):383–386.
Wikipedia Contributors. Pelger–Huët anomaly. Wikipedia, The Free Encyclopedia. Last updated April 25, 2022. Available at: https://en.wikipedia.org/wiki/Pelger%E2%80%93Hu%C3%ABt_anomaly
Keywords:
Pelger-Huët anomaly, pseudo Pelger-Huët anomaly, Pelger-Huët neutrophils, pince-nez neutrophils, hypolobated neutrophils, neutrophil nuclear segmentation defect, inherited Pelger-Huët anomaly, LBR gene mutation, pseudo-Pelger changes in MDS, Pelger-Huët in myelodysplastic syndrome, Pelger-Huët in leukemia, neutrophil dysplasia, blood smear morphology, granulocyte nuclear abnormalities, hematology smear interpretation, Pelger-Huët syndrome, acquired Pelger-Huët, peripheral smear findings, Pelger-Huët diagnosis, Pelger-Huët vs pseudo Pelger-Huët

Request Online Consultation With Dr M Abdou
Fee: US$100
Secure payment via PayPal (credit and debit cards accepted)
Pay Now


Mutation here involve which Gene?
Hello,
Thank you for your message.
Pelger-Huët anomaly is a benign, dominantly inherited defect of terminal neutrophil differentiation secondary to mutations in the lamin B receptor (LBR) gene.
Regards,
My brother and my oldest son both have been verified as having PHA. Other things they have in common are Dyslexia and a few other learning issues. Could these possibly be connected somehow? I think I read somewhere that certain mutations in the LBR gene can cause learning issues.
Hi Suzanne,
Thank you for your comment.
I really don’t know if there is a link between PHA and Dyslexia but I think this article may answer some of your queries with this matter:
https://rarediseases.info.nih.gov/diseases/9148/pelger-huet-anomaly
BW,
I went to the hospital recently for heart issue due to over stress. This test came back in my patient portal.
Smear Exam by Pathologist
Note:
Mar 02, 2022 11:18 a.m. EST
Comment:
Red blood cells show minimal variation in shape and size with no inclusions and less than one schistocyte per 100 red blood cells seen. No nucleated red blood cells are seen. The white blood cell differential count is confirmed. Normal morphological features are seen; rare Pelger Huet cells seen. The platelet count shows decreased numbers. No platelet clumping is seen. Peripheral Smear Review: Mild macrocytic anemia NOTE: Vit B12/Folate levels and if normal, considerations for a marrow study is recommended.
Hi Pamela,
Thank you for your comment.
From your blood film comment they are querying ?possible myelodysplasia in view of the macrocytic anaemia and the Pelger-Huët anomaly. I would suggest initially to check your serum B12, Folate, GGT, and LDH.
BW,
Hello, I was wondering if the following results of “rare hypograndular neutrophil” is the same as PHA or APHA.
Normochromic, normocytic RBCs. Platelets adequately granulated. Rare hypogranular neutrophil of uncertain significance in this setting. No other features of dysplasia. No blasts.
Hi Christine,
Thank you for your comment.
As long as the hypogranular neutrophils are not seen frequently on the blood film and your CBC is normal you can safely ignore it.
BW,
Is this “condition “ related to Eosinophilic?
I am not medically educated but fascinated with diagnoses and medical vocabulary.
Thank you for your kind response.
Hi Ila,
Thank you for reaching out.
Pelger-Huët anomaly (PHA) does not directly relate to eosinophilia. PHA is a genetic disorder characterized by abnormal nuclear shape in neutrophils, where the nuclei appear hyposegmented.
This condition is typically benign and does not affect the function of the neutrophils.
Eosinophilia, on the other hand, refers to an elevated number of eosinophils in the blood and can be caused by a variety of conditions, including allergic reactions, parasitic infections, and certain hematologic disorders.
There is no known direct association between PHA and eosinophilia.
Best wishes,
Dr. M. Abdou
I don’t ordinarily comment but I gotta admit appreciate it for the post on this perfect one.