Blood Morphology
Blood morphology adds greatly to the value of a routine blood count. A skilful examination of a well-made blood smear constitutes the most valuable single procedure in the hematology laboratory. Despite normal blood count figures, careful observation of blood morphology suggested previously unsuspected disorders. For example, in some cases, the finding of hypersegmented neutrophils on the blood smear was the first hematologic clue to a significant deficiency of vitamin B12 or folate, the erythrocytes lacking the characteristic macrocytosis associated with such deficiencies.
The reporting of blood morphology has been improving in recent years, although in many clinical laboratories, it still receives little attention. The blood count report form frequently leaves only a tiny area for morphologic comments!
A blood smear is a drop of blood spread thinly onto a glass slide that is then treated with a special stain and examined under a microscope by a trained laboratorian. It is a snapshot of the cells that are present in the fluid portion of the blood (plasma) at the time the sample is obtained. The results of a blood smear typically include a description of the appearance of the red blood cells, white blood cells, and platelets as well as any abnormalities that may be seen on the slide.
Red Blood Cell Morphology
Red blood cells (erythrocytes) are biconcave disks with a diameter of 7-8 microns, which is similar to the size of the nucleus of a resting lymphocyte. In normal red blood cells, there is an area of central pallor that measures approximately 1/3 the diameter of the cell. Though reference ranges vary between laboratories and in different age groups, normocytic red blood cells typically have a mean corpuscular volume (MCV) between 80-100 fL.
Spherocytes are formed when there is a loss of part of the red blood cell membrane. This may occur in the setting of immune-mediated hemolysis or congenital red cell membrane defects such as hereditary spherocytosis. Spherocytes are smaller than normal red blood cells and lack central pallor. They are less deformable and less able to navigate through small vessels, leading to increased destruction in the spleen.

Peripheral blood smear demonstrating numerous spherocytes lacking central pallor, characteristic of hereditary spherocytosis and immune hemolysis.
Microcytic red blood cells measure 6 microns or less in diameter. The mean corpuscular volume is generally less than 80 fL, though the normal range varies slightly between laboratories and in different age groups. In contrast to spherocytes, which are also decreased in diameter, microcytes retain their central pallor. In microcytosis due to iron deficiency, the central pallor is increased (more than 1/3 the diameter of the cell).

Peripheral blood smear showing microcytic red blood cells with two lymphocytes for accurate size comparison, typical of iron deficiency anemia and thalassemia.
Teardrop cells in a peripheral blood smear from a patient whose bone marrow was extensively replaced by B lymphoblastic leukemia. Teardrop cells may be seen in the setting of marrow infiltration (by fibrosis, granulomatous inflammation, hematologic or metastatic malignancy), splenic abnormalities, megaloblastic anemia, and thalassemia. True teardrop cells have slightly rounded or blunted ends. In contrast, teardrop cells that are formed as an artefact of smear preparation have very sharp points, all facing in the same direction.

Peripheral blood smear demonstrating teardrop-shaped red blood cells (dacrocytes), commonly associated with myelofibrosis and marrow infiltration.
Cabot rings are thin, threadlike, red to violet rings or “figure 8” shaped inclusions in red blood cells. Cabot rings are remnants of the mitotic spindle and can be seen in megaloblastic anemia, medication effect, myelodysplasia and other forms of dyserythropoiesis. In this image of a blood smear from a patient with vitamin B12 deficiency, the Cabot ring is visible as a faint ring-shaped inclusion in the polychromatophilic cell in the center of the field.

Peripheral blood smear demonstrating a Cabot ring inside a red blood cell, typically associated with dyserythropoiesis, megaloblastic anemia, and severe anemia.
Peripheral blood smear of a 38-year-old female with long-standing Crohn’s disease (CD) and development of microcytic anemia. The smear shows numerous target cells and a spur cell (top right). All liver function tests were abnormal indicating that the target cells are due to liver disease secondary to CD. This patient originally had a concomitant iron deficiency. Spur red cells have elongated projections while Burr cells are red cells with circumferential blunted borders. The former is typically seen in liver disease while the latter is seen in uremia. The “Burr” morphology, in this case, is artifactual related to slide preparation and not related to uremia.
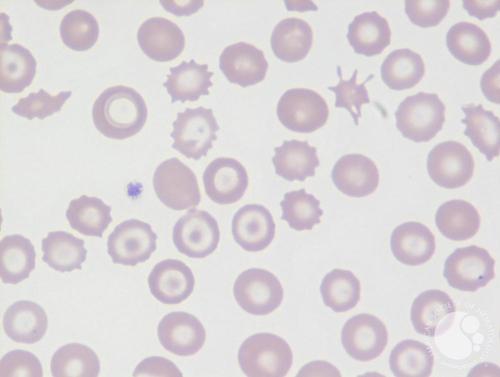
Peripheral blood smear demonstrating target cells (codocytes) and spur cells (acanthocytes), characteristic red cell abnormalities associated with liver disease.
Acanthocytes in two patients with liver disease. Acanthocytes (also called spur cells) are spiculated cells with irregular, pointed or club-like projections that are unevenly distributed on the cell surface. Central pallor is absent. Acanthocytes form as a result of membrane lipid abnormalities and can be seen in liver disease, neuroacanthocytosis, severe malnutrition, and abetalipoproteinemia.

Peripheral blood smear demonstrating acanthocytes, red blood cells with irregular spicules typically associated with liver disease, abetalipoproteinemia, and neuroacanthocytosis.
Sickle cells (drepanocytes) are elongated red blood cells with pointed ends. They are seen in sickling hemoglobinopathies such as sickle cell anemia (homozygous hemoglobin SS), hemoglobin SD disease, and hemoglobin S/beta-thalassemia.

Peripheral blood smear demonstrating sickle-shaped red blood cells, characteristic of sickle cell disease and sickling crises.
Echinocytes (Burr Cells) have multiple short, blunt projections evenly spaced over the cell surface. The central pallor is retained. Echinocytes can be seen in uremic patients. They can also be seen as an artefact of slide preparation or prolonged specimen storage.

Peripheral blood smear demonstrating burr cells (echinocytes), characterized by uniform short spicules, commonly seen in renal disease, liver disease, and artifact.
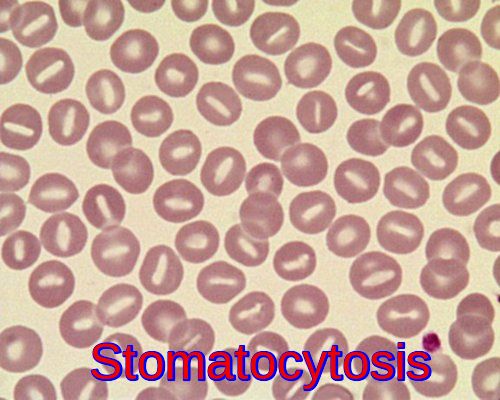
Stomatocytes are red cells with a slit-like or “fish-mouth” central pallor. Stomatocytes may be seen in patients with alcoholic liver disease, hereditary stomatocytosis, or Rh null disease, among other conditions. They may form in vitro in the presence of certain cationic medications or low pH.

Peripheral blood smear demonstrating stomatocytes, red blood cells with a characteristic mouth-shaped (stoma-like) central pallor, seen in hereditary stomatocytosis, liver disease, and alcohol-related disorders.
Red cell fragments (schistocytes) in a patient with microangiopathic hemolysis due to thrombotic thrombocytopenic purpura (TTP). Small triangulocytes and larger, crescent-shaped helmet cells are present. Both of these are red cell fragments and would be included in the schistocyte count. When numerous small schistocytes are present, automated cell counters may count the small red cell fragments as platelets, leading to a falsely elevated automated platelet count.

Peripheral blood smear demonstrating schistocytes, triangulocytes, and helmet cells—fragmented erythrocytes characteristic of microangiopathic hemolytic anemia (MAHA), DIC, TTP, and mechanical hemolysis.
Oxidative hemolysis induced by furosemide in a patient with G6PD deficiency. In oxidative hemolysis, the peripheral smear may show irregularly contracted red blood cells, hemighost or blister cells, and spherocytes. Irregularly contracted cells lack central pallor, and the hemoglobin appears condensed and irregularly distributed in the red blood cell.

Peripheral blood smear demonstrating irregularly contracted cells, blister cells, and hemighost forms commonly associated with oxidative hemolysis such as G6PD deficiency.
Clumping (agglutination) of red blood cells is frequently caused by cold agglutinins. Cold agglutinins are IgM antibodies that may arise following viral or Mycoplasma infections, or in the setting of plasma cell or lymphoid neoplasms. Agglutination of red cells can interfere with red blood cell indices. The red blood cell count may be falsely decreased, and the MCV falsely increased, as clumps of red cells are measured as single cells. The hemoglobin level will be accurate, as this parameter is measured after lysing the red cells.

Peripheral blood smear demonstrating marked red cell agglutination, a classic finding in cold agglutinin disease and immune-mediated erythrocyte clumping.
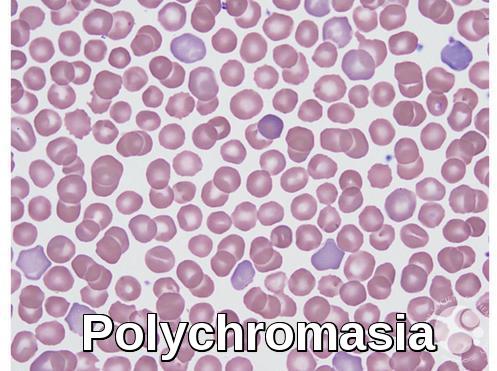
Peripheral blood smear demonstrating polychromasia with increased reticulocytes, indicating active erythropoiesis and response to anemia or hemolysis.
Polychromasia (polychromatophilic cells) in a neonate. Polychromatophilic cells are young red blood cells that have been recently released from the bone marrow. They are larger than mature red cells, and are bluish in color. Polychromasia is increased in hemolysis, blood loss, and marrow infiltration. Normal neonates have a higher number of polychromatophilic cells than older children and adults.
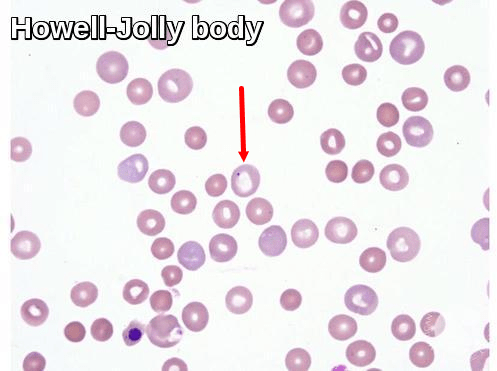
Peripheral blood smear demonstrating a Howell–Jolly body, a basophilic nuclear remnant typically seen in asplenia, hyposplenism, and megaloblastic anemia.
Howell-Jolly body: the red blood cell in the center of the image contains a Howell-Jolly body. Howell-Jolly bodies are small (0.5-1 micron) purple inclusions that contain DNA. They are thought to represent chromosomes that have separated from the mitotic spindle that is left behind when the red cell nucleus is extruded. These inclusions are generally removed by the spleen. Patients with asplenia or hyposplenism may have increased Howell-Jolly bodies on their peripheral blood smear. A nucleated red blood cell is also present at the bottom left side of the image.
Blood Morphology – Erythroid Precursors
Proerythroblasts (also called pronormoblasts) are the earliest erythroid precursors. These are large cells with basophilic, agranular cytoplasm, round nuclei, and high nuclear-cytoplasmic ratios. The chromatin is evenly dispersed but is slightly more dense than myeloblast chromatin. One or more nucleoli may be visible. A perinuclear clear area (hof) may also be seen. A single proerythroblast is seen in the center of this image. Polychromatophilic and orthochromic normoblasts are present on the right side of the field.
Basophilic normoblasts (also called basophilic erythroblasts or early erythroblasts) are smaller than proerythroblasts, with more condensed chromatin and lower nuclear-cytoplasmic ratios. The cytoplasm is deep blue, and a pale perinuclear halo may present. The two cells in the center of the field are basophilic normoblasts.

Bone marrow smear demonstrating early erythroblasts with basophilic cytoplasm and centrally placed nuclei, representing an early stage of erythroid maturation.
Orthochromic normoblasts (also called orthrochromatophilic normoblasts, orthrochromatophilic erythroblasts, or late erythroblasts) are slightly larger than mature red blood cells. They have small, round nuclei and dense, pyknotic chromatin. The cytoplasm is generally slightly more basophilic than the cytoplasm of a mature red blood cell.

Bone marrow smear demonstrating orthochromic normoblasts, late-stage erythroid precursors with condensed nuclei and hemoglobinized cytoplasm.
Erythroid precursors at various stages of maturation. Basophilic normoblasts are present at the center of the field. Polychromatophilic normoblasts and orthochromic normoblasts are present near the bottom of the field. As erythroid precursors mature, the cell size and nuclear-cytoplasmic ratio decrease, and the chromatin becomes progressively more condensed. The cytoplasm changes color from deep blue to gray-blue to gray-pink as the hemoglobin content increases.
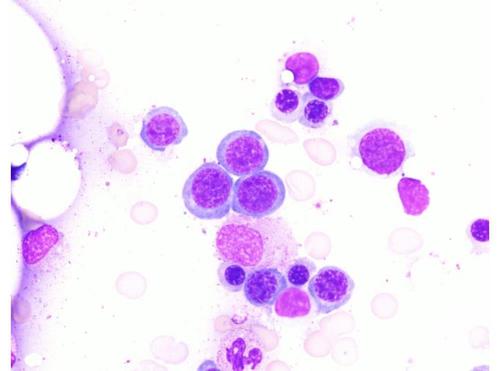
Bone marrow smear demonstrating a range of erythroid precursors at different maturation stages, reflecting active erythropoiesis.
A deficiency of either vitamin B12 or folic acid results in megaloblastic erythroid cells-megaloblasts. These deficiencies result in a decrease in DNA synthesis which slows and inhibits DNA replication (nuclear division). Nuclear maturation is slowed whereas cytoplasmic maturation (largely dependent on RNA function) is unaffected. The impaired nuclear maturation is seen as open, loose, immature chromatin (cut-salami pattern). In contrast to the nucleus, the cytoplasm of megaloblastic cells is abundant with normal hemoglobinization. This disparity between the nucleus and cytoplasm is known as nuclear-cytoplasmic asynchrony. Although most noticeable in erythroid cells failure of DNA synthesis also affects myeloid and megakaryocytes. Giant bands and hypersegmented polymorphonuclear neutrophils are common.
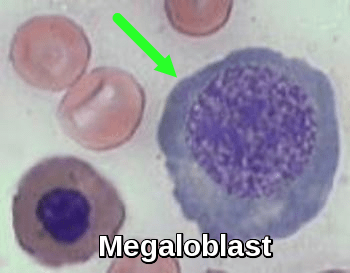
BM smear demonstrating a megaloblast, an enlarged erythroid precursor with nuclear–cytoplasmic asynchrony typical of vitamin B12 or folate deficiency.
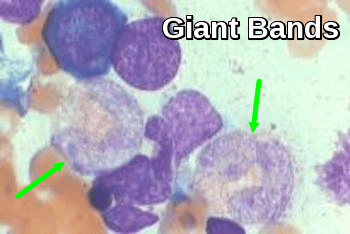
BM smear demonstrating giant band neutrophils, an abnormal granulocyte form often seen in megaloblastic anemia and myelodysplastic syndromes.
Vacuolated erythroid precursors can be seen in copper deficiency, Pearson syndrome, and myelodysplastic syndromes. In this image, a vacuolated erythroid precursor is adjacent to another dysplastic erythroid precursor with megaloblastic features (nuclear-cytoplasmic asynchrony) and nuclear irregularities. The patient had acute myeloid leukemia with myelodysplasia-related changes.
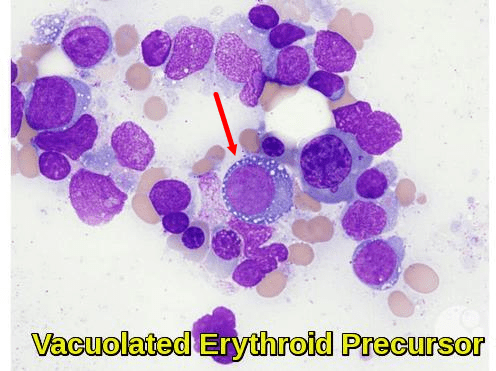
Bone marrow smear demonstrating a vacuolated erythroid precursor, a feature commonly associated with myelodysplastic syndromes, copper deficiency, or severe nutritional anemia.
White Blood Cell Morphology
Mature segmented neutrophils have condensed chromatin and 2 to 5 nuclear lobes separated by thin filaments. The cytoplasm is pale pink and contains numerous specific granules. In healthy adults, approximately 40-80% of peripheral blood leukocytes are segmented neutrophils.
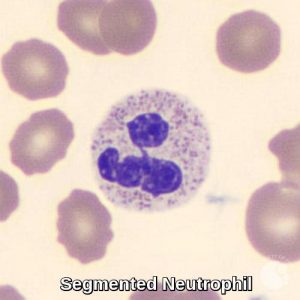
Peripheral blood smear demonstrating a segmented neutrophil with a multilobed nucleus, representing a mature granulocyte.
Band neutrophils are slightly less mature than segmented neutrophils and have indented, unsegmented “C” or “S” shaped nuclei. Band neutrophils normally account for approximately 5-10% of peripheral blood leukocytes. An increased proportion of band neutrophils can be seen in infectious and inflammatory conditions.
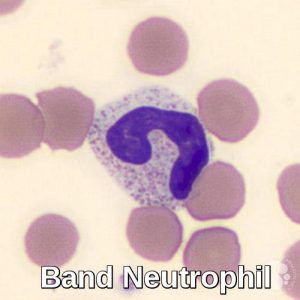
Peripheral blood smear demonstrating a band neutrophil, an immature granulocyte with a curved, unsegmented nucleus.
Toxic granulation in neutrophils is found in inflammatory states. The toxic granules are azurophilic and may be found in the promyelocyte, metamyelocyte, band (shown here), and mature stages.
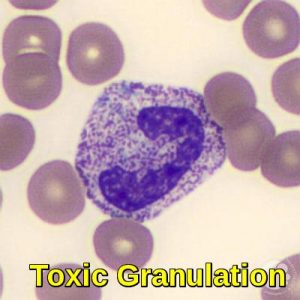
Peripheral blood smear demonstrating a neutrophil with toxic granulation, typically seen in severe infection, inflammation, or marrow stress.
Hypersegmented neutrophil in a patient with vitamin B12 deficiency. Hypersegmented neutrophils have 6 or more nuclear lobes. They are typically seen in megaloblastic anemia due to vitamin B12 or folic acid deficiency, but may also be present in myelodysplastic syndromes and rare congenital conditions.
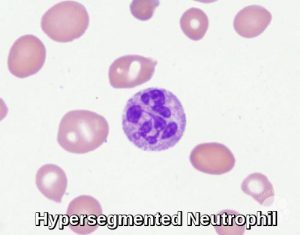
Peripheral blood smear demonstrating a hypersegmented neutrophil, a classic morphological feature of vitamin B12 and folate deficiency.
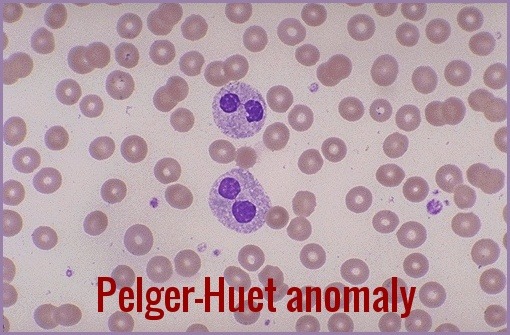
Pelger-Huet anomaly is a congenital autosomal dominant anomaly in which neutrophil nuclei fail to segment normally. In homozygotes, the nucleus is round. In heterozygotes, most granulocytes have bilobed nuclei (“pince-nez” cells) resembling bands.
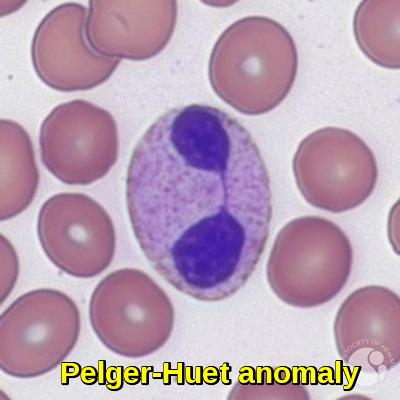
Peripheral blood smear demonstrating Pelger–Huët anomaly, characterized by a bilobed neutrophil nucleus with mature chromatin, seen in inherited PHA and pseudo–Pelger changes in MDS.
Neutrophils with prominent hypolobation and Pseudo Pelger-Huet cytomorphology in a patient seen in the posttransplant setting. This patient was on mycophenolate which is sometimes known to induce such changes. One must be cautious not to make a diagnosis of myelodysplastic syndrome in the setting.

Peripheral blood smear demonstrating a pseudo–Pelger–Huët neutrophil, a dysplastic form with a hypolobated nucleus commonly seen in myelodysplastic syndromes and severe infections.
Neutrophilic metamyelocytes have condensed chromatin and a slightly indented nucleus (the indentation is less than half the diameter of the nucleus). The cytoplasm contains abundant specific granules and rare primary (azurophilic) granules. Metamyelocytes are not normally present in the peripheral blood but can be seen in infectious or inflammatory states, and other reactive and neoplastic conditions.

Peripheral blood smear demonstrating a metamyelocyte, an intermediate granulocyte precursor with an indented nucleus and maturing granules.

Peripheral blood smear demonstrating a myelocyte, a granulocytic precursor with a round nucleus and developing cytoplasmic granules.
Myelocytes contain both primary (azurophilic) and secondary/specific (pink or lilac) cytoplasmic granules. The proportion of secondary granules increases as the cell matures. The nucleus is round and lacks a nucleolus. The chromatin is more condensed than that of promyelocytes. Myelocytes are not normally present in peripheral blood, but may be seen in infectious/inflammatory conditions, growth factor effect, marrow infiltration, and myeloid neoplasms.
Promyelocytes are larger than myeloblasts and have basophilic cytoplasm containing primary (azurophilic) granules. A Golgi zone may be visible as a paranuclear hof or clearing. The nuclear chromatin is finely dispersed, and nucleoli may be visible. Promyelocytes comprise approximately 2% of nucleated cells in the bone marrow and do not circulate in peripheral blood under normal conditions.
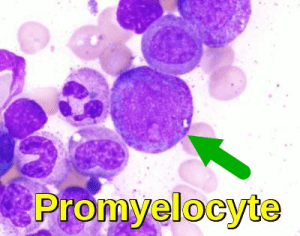
Bone marrow smear demonstrating a promyelocyte with abundant primary granules and an immature nucleus, representing an early stage of granulocyte development.
Myeloblasts are approximately 15-20 microns in size and have high nuclear-to-cytoplasmic ratios. The nucleus is usually round to oval but may be irregular. The chromatin is smooth and open, and one or more nucleoli may be present. The cytoplasm is pale blue. Cytoplasmic granules are not typically seen, though rare small granules may be present. Leukemic myeloblasts may contain a few cytoplasmic granules or Auer rods, which are reddish, linear structures composed of fused primary granules. The presence of Auer rods indicates myeloid malignancy.
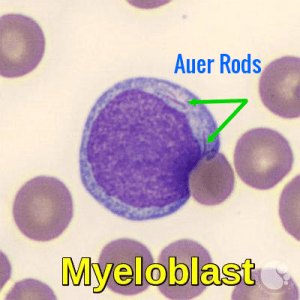
Peripheral blood smear demonstrating a myeloblast containing Auer rods, a hallmark feature of acute myeloid leukemia.
Eosinophils measure 10-17 microns in diameter and have abundant, slightly basophilic cytoplasm containing numerous coarse, reddish-orange cytoplasmic granules. Most eosinophils have bilobed nuclei, but occasional forms with trilobed nuclei may be seen. The chromatin is condensed. Eosinophils usually account for a minor subset of peripheral blood leukocytes. Increased numbers of eosinophils can be seen in parasitic infections, allergic conditions, drug hypersensitivity, myeloid neoplasms, and lymphoproliferative disorders.
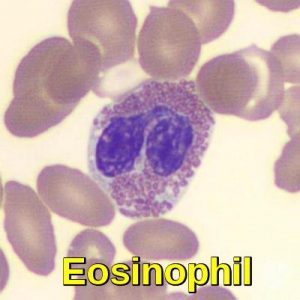
Peripheral blood smear demonstrating an eosinophil with a bilobed nucleus and prominent eosinophilic granules, characteristic of allergic and parasitic responses.
Basophils have segmented nuclei that are often at least partially obscured by abundant coarse, dark blue to purple cytoplasmic granules. Basophil granules are water soluble, and may wash out during staining. Basophils normally circulate in low numbers. Basophilia may be seen in inflammatory and allergic conditions, hypothyroidism, and myeloproliferative neoplasms.

Peripheral blood smear demonstrating a basophil with coarse basophilic granules, characteristic of hypersensitivity reactions and myeloproliferative disorders.
Normal resting lymphocytes are small cells with condensed chromatin and a small amount of pale basophilic cytoplasm. The nucleus of a resting lymphocyte is just slightly larger than a red blood cell.

Peripheral blood smear demonstrating a mature lymphocyte with a dense round nucleus and scant cytoplasm, typical of normal adaptive immune cells.
Reactive lymphocytes show a range of morphologic features. Reactive lymphocytes with immunoblast-like morphology are large cells with high nuclear-cytoplasmic ratios, condensed chromatin, and deeply basophilic cytoplasm. Another type of reactive lymphocyte has less condensed chromatin and abundant, pale blue cytoplasm that may appear to “hug” adjacent red blood cells. These cells are also called Downey type II cells. They can be seen in a variety of conditions, but are often increased in infectious mononucleosis due to EBV infection.

Peripheral blood smear demonstrating a reactive lymphocyte, characterized by an enlarged size, irregular nucleus, and abundant basophilic cytoplasm, commonly seen in viral infections such as EBV.
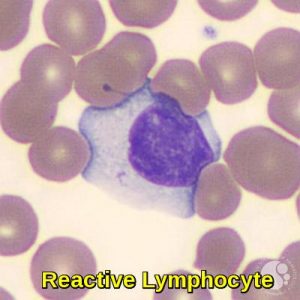
Peripheral blood smear demonstrating a reactive lymphocyte with abundant pale basophilic cytoplasm that molds around neighboring red cells, typical of infectious mononucleosis and other viral infections.
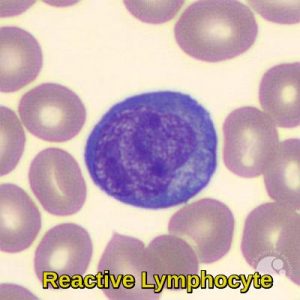
Peripheral blood smear demonstrating a reactive lymphocyte with intensely basophilic cytoplasm and an irregular nucleus, typically seen in viral infections and heightened immune responses.
Lymphocytes with peripherally clumped chromatin and often deep blue cytoplasm similar to plasma cells are termed plasmacytoid lymphocytes. These transitional forms between lymphocytes and plasma cells are seen in the blood of patients with viral infections. These cells are variously known as atypical lymphocytes, lymphocytoid plasma cells or plasmacytoid lymphocytes.
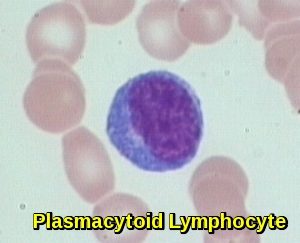
Peripheral blood smear demonstrating a plasmacytoid lymphocyte with eccentric nucleus and basophilic cytoplasm, seen in viral infections, immune activation, and some lymphoproliferative disorders.
Monocytes measure 12-20 microns in diameter and have abundant greyish-blue cytoplasm and fine, azurophilic cytoplasmic granules. Cytoplasmic vacuoles may be present. The nucleus is irregular, indented, or kidney-shaped. Although the chromatin is condensed, it is somewhat finer than the chromatin of a mature neutrophil or resting lymphocyte.
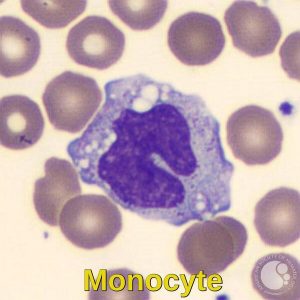
Peripheral blood smear demonstrating a monocyte with a folded nucleus and ample gray-blue cytoplasm, characteristic of the largest normal circulating leukocyte.
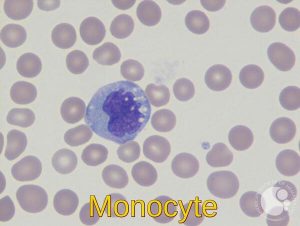
Peripheral blood smear demonstrating a monocyte with an indented nucleus and characteristic gray-blue cytoplasm.
Normal monocyte in circulation with folded nuclei and abundant vacuolated cytoplasm.
Acute monoblastic/monocytic leukemia: blasts may vary in appearance. The cytoplasm may contain fine granules or vacuoles.
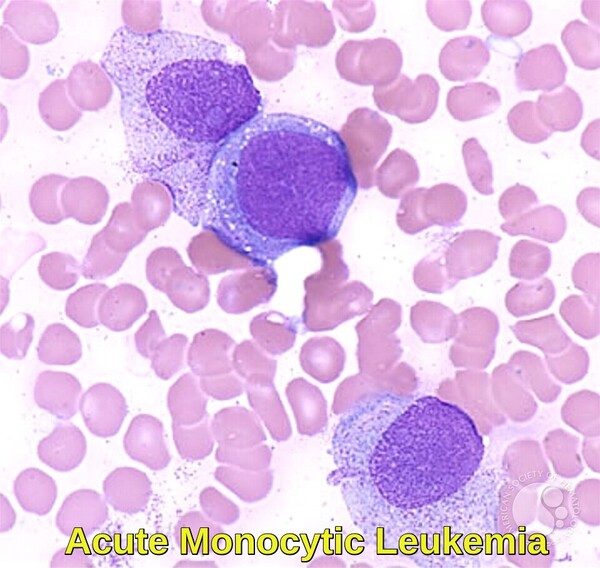
Peripheral blood smear demonstrating acute monocytic leukemia with prominent monoblasts and immature monocytic cells, characteristic of AML M5.
Peripheral smear from a patient with monocytic leukemia. The monocytes are dysplastic with vacuolated and irregular cytoplasm.

Peripheral blood smear demonstrating dysplastic monocytes with nuclear and cytoplasmic abnormalities, a feature seen in acute myeloid leukemia with monocytic differentiation.
Platelet Morphology

Peripheral blood smear demonstrating normal platelets, appearing as small purple cytoplasmic fragments distributed among red blood cells.
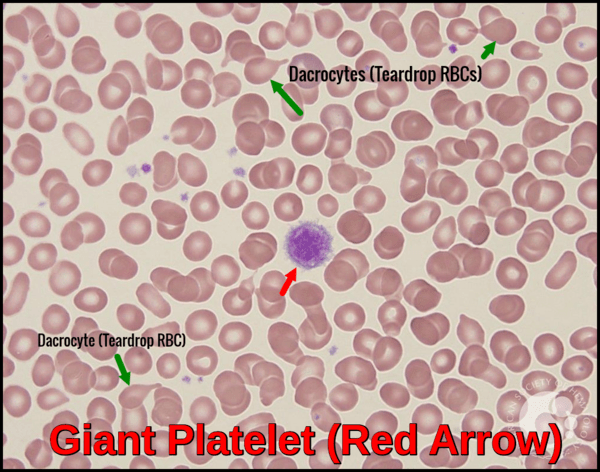
Peripheral blood smear demonstrating a giant platelet (red arrow) and multiple dacrocytes (green arrows), findings seen in myeloproliferative neoplasms and marrow infiltration.
*Large platelets have a diameter greater than 4 microns. Giant platelets have a diameter greater than 7 microns (larger than a normal red blood cell). Large platelets can be seen when platelet turnover is increased (as in immune thrombocytopenic purpura). They may also be present in myeloproliferative neoplasms, myelodysplasia, and some congenital thrombocytopenia syndromes, including Bernard-Soulier syndrome and MYH9-related disorders. Giant platelets may fall outside the upper size threshold for platelets on hematology analyzers. If sufficient numbers of giant platelets are present, the automated platelet count may be falsely decreased.

Peripheral blood smear demonstrating platelet clumping, often due to EDTA-related pseudothrombocytopenia or inadequate sample mixing.
*Platelet clumping can lead to a falsely decreased automated platelet count (pseudo thrombocytopenia/spurious thrombocytopenia). At higher magnification, the platelets within the aggregate appear normal in structure and granularity. Causes of platelet clumping include platelet activation (due to traumatic venipuncture) and EDTA-dependent antibodies that react with platelet glycoprotein IIb/IIIa. In the latter case, the clumping can be corrected by using blood collection tubes containing an alternative anticoagulant, such as sodium citrate.

Peripheral blood smear demonstrating platelet satellitism, with platelets forming a rosette around neutrophils—an EDTA-associated artifact that may mimic thrombocytopenia.

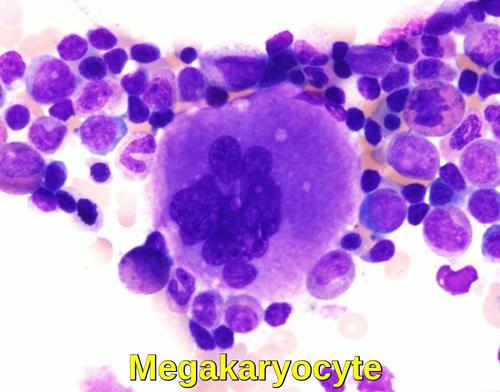
Bone marrow smear demonstrating a mature megakaryocyte with a multilobulated nucleus and extensive granular cytoplasm, responsible for platelet production.

Live-cell GIF showing mature megakaryocytes extending proplatelets and releasing circulating platelets.
Questions and Answers:
What is a blood smear test and what does it detect?
A blood smear (peripheral blood film) evaluates the size, shape, colour, and structural features of red cells, white cells, and platelets. It detects anaemia patterns, hemolysis, infections, leukemia, lymphoma, myelodysplasia, myeloproliferative neoplasms, and artefacts missed by automated analysers.
What do abnormal red blood cell shapes mean on a blood smear?
Abnormal shapes such as spherocytes, schistocytes, target cells, teardrop cells, elliptocytes, and sickle cells each point toward specific conditions including haemolysis, microangiopathy, thalassaemia, marrow infiltration, hereditary membrane defects, or sickle cell disease.
What causes schistocytes in a blood smear?
Schistocytes indicate red cell fragmentation, commonly seen in thrombotic microangiopathies (TTP, HUS), DIC, mechanical valve haemolysis, severe burns, and malignant hypertension. Their presence is a red-flag finding requiring urgent evaluation.
What does anisopoikilocytosis indicate?
Marked variation in red cell size and shape suggests iron deficiency, mixed nutritional deficiencies, myelodysplasia, marrow infiltration, advanced haemolytic anaemia, or combined disease states.
Why do hypersegmented neutrophils appear in blood morphology?
Neutrophils with ≥6 lobes are classic for B12 or folate deficiency but may also appear in myelodysplastic syndromes or medication-related marrow suppression.
What do blasts on a peripheral blood film mean?
The presence of blasts suggests acute leukemia, myelodysplastic transformation, or severe marrow stress. This warrants urgent specialist review and bone marrow evaluation.
What are the causes of giant platelets on a blood smear?
Giant platelets occur in ITP, myeloproliferative neoplasms, myelodysplasia, Bernard-Soulier syndrome, and MYH9-related disorders. They can cause falsely low automated platelet counts.
Why do platelets clump on a blood smear?
Clumping is usually due to EDTA-induced artefact (pseudo-thrombocytopenia), traumatic venipuncture, or platelet activation. It causes inaccurate automated platelet counts and should prompt repeat sampling (e.g., in citrate).
Can blood morphology be abnormal if the full blood count is normal?
Yes. A normal FBC can still hide early dysplasia, nutritional deficiency, early haemolysis, or subtle malignant changes visible only on morphology, making smear review clinically indispensable.
What are teardrop cells and what conditions cause them?
Teardrop cells (dacrocytes) are associated with myelofibrosis, marrow infiltration (metastasis), severe megaloblastic anaemia, thalassaemia, and extramedullary haematopoiesis. True teardrops have smooth, not sharply pointed, ends.
How can a blood smear differentiate reactive from malignant lymphocytosis?
Reactive lymphocytes vary in size with abundant cytoplasm, whereas malignant lymphocytes appear small, clonal, and mature-looking (CLL) or display atypia depending on subtype. Combined smear + flow cytometry is the diagnostic pathway.
What is the significance of target cells on a blood smear?
Target cells suggest thalassaemia, liver disease, haemoglobinopathies, or post-splenectomy states, and they guide further haemoglobin studies.
Why does cold agglutinin disease affect blood smear interpretation?
Cold agglutinins cause RBC clumping, falsely elevating MCV and reducing RBC count. Warming the sample corrects the artefact.
References:
Teresa Scordino. ASH Image Bank – American Society of Hematology. Available at: https://imagebank.hematology.org/
Girish Venkataraman. ASH Image Bank – American Society of Hematology. Available at: https://imagebank.hematology.org/
Peter Maslak. ASH Image Bank – American Society of Hematology. Available at: https://imagebank.hematology.org/
John Lazarchick. ASH Image Bank – American Society of Hematology. Available at: https://imagebank.hematology.org/
Perla Vicari. ASH Image Bank – American Society of Hematology. Available at: https://imagebank.hematology.org/
Wikipedia Contributors. Megakaryocyte. Wikipedia, The Free Encyclopedia. Updated 13 September 2023.
Available at: https://en.wikipedia.org/wiki/Megakaryocyte
Wikimedia Commons Contributors. Platelets release in mature megakaryocytes (GIF). Wikimedia Commons.
Available at: https://commons.wikimedia.org/wiki/File:Platelets_release_in_mature_megakaryocytes_.gif
Paul C. Hattersley, MD, Judith L. Engels, MT. The Reporting of Blood Morphology.
Available at: https://goo.gl/zr26TK
Paul G. Hattersley, Judith L. Engels. Neutrophilic Hypersegmentation Without Macrocytic Anemia. Western Journal of Medicine. 1974;121:179–184.
G.C. De Gruchy. Clinical Hematology in Medical Practice. 2nd ed. Philadelphia: F.A. Davis Co.; 1964:63.
Paul G. Hattersley, D. Ragusa. Don’t Forget the Morphology: The Importance of Evaluation of Blood Smears. California Medicine. 1965;103:175–177.
Department of Pathology, University of Virginia, School of Medicine. Megaloblastic Anemia – RBC Disorders.
Available at: https://www.med-ed.virginia.edu/courses/path/innes/rcd/mega.cfm
Keywords:
blood morphology, peripheral blood smear, blood film examination, red blood cell morphology, white blood cell morphology, platelet morphology, blood smear interpretation, blood cell morphology, abnormal blood morphology, RBC morphology abnormalities, WBC morphology abnormalities, platelet morphology disorders, blood cell identification, poikilocytosis, anisocytosis, schistocytes, spherocytes, target cells, tear drop cells, burr cells, acanthocytes, stomatocytes, sickle cells, Howell–Jolly bodies, Cabot rings, toxic granulation, hypersegmented neutrophils, Pelger–Huët anomaly, pseudo-Pelger–Huët cells, reactive lymphocytes, blasts on blood smear, myeloblasts, promyelocytes, myelocytes, metamyelocytes, erythroid precursors, proerythroblasts, megakaryocytes, thrombopoiesis, giant platelets, large platelets, platelet size abnormalities, increased platelet turnover, immune thrombocytopenic purpura ITP, myeloproliferative neoplasms MPN, myelodysplastic syndromes MDS, congenital thrombocytopenia, Bernard–Soulier syndrome, MYH9-related disorders, falsely decreased platelet count, automated platelet count errors, EDTA-induced platelet clumping, pseudo-thrombocytopenia, spurious thrombocytopenia, platelet glycoprotein IIb/IIIa antibodies, platelet activation, traumatic venipuncture, citrate tube anticoagulant, platelet satellitism, platelet clumping, hematology microscopy, RBC morphology in anemia, leukocyte morphology, clinical significance of blood morphology, Ask Hematologist, Dr Moustafa Abdou, Dr Abdou Hematologist, AskHematologist.com


Request Online Consultation With Dr M Abdou
Fee: US$100
Secure payment via PayPal (credit and debit cards accepted)
Pay Now



Merci beaucoup pour cet article très complet.
Please send me things that are new
What are Dohle Bodies on RBC morthphogy blood test.
Hi Diane,
Thank you for your comment.
Döhle bodies are single or multiple blue cytoplasmic inclusions that are remnants of rough endoplasmic reticulum. They are seen in bacterial infections but also following tissue damage including burns, in inflammation, following administration of G-CSF, and during pregnancy.
BW,
Can I ask you a question?
I have iron deficiency anemia. The red blood cell morphology shows polychromasia 2+, anisocytosis 2+, microsytes 2+, hypochromia 1+, schistocytes 1+, ovalocytes 1+, dacrocytes 1+. What came first? The anemia or the irregular numbers? Do these not indicate a blood disorder that would in turn cause anemia, or the other way around? I hope I hear back from you.
Hi Lisa,
Thanks for your comment.
Iron deficiency anaemia can cause red cell abnormalities including hypochromasia, microcytosis, pencil cells, and others. You should identify the cause of the iron deficiency and treat it.
After treating the anaemia with iron supplements repeat your CBC and see if the abnormalities have disappeared or are still present, if they are still present I would suggest checking Hb electrophoresis, hemolysis screen, B12 and Folate.
BW,
Hi Dr.
This article was very useful for me.
Thanks
Hi Faresh,
Thank you, happy to see your comment.
BW,
This article was very useful for medical team in order to provide a proper treatment to our clients or patients.
Thanks
Dear Henry,
Many thanks for your kind words.
I’m pleased that you find this article useful.
BW,
Thank you so much for this article. It is teaching me to be able to identify the Cells and the inclusion of courses really helped.
More of these articles help.
Is there more where you include the CBC parameters for Medical Technicians beginners to have a better understanding of interpretation of slides.
Hi MBali,
Thanks for your comment.
You can find more about blood morphology through the ASH website:
ASH Image Bank: https://imagebank.hematology.org/
BW,
very useful, thank you so much……this article is going to help me alot
Hi Jacob,
Thank you for your comment.
Pleased that you found this article helpful.
BW
I hope someone can help me, I was looking in my chart test results and I found this liste under morphology~
Component Results
Component Your Value
Platelet Estimate Normal
Polychromia Rare
This blood test was done when I was in the hospital with some breathing issues a few yr back ,everytime I had asked my Primary care r she doesn’t know what it means,,, she sent me to a hematologist an all he saqi was Maybe some low iron ,I have no idea what this test was for and what the poly test reading says Rare. I would love to understand this test and find out what the result Rare means >> thank you for your help
Hi Patrice,
Thank you for your comment.
Polychromasia occurs on a lab test when some of your red blood cells show up as bluish-gray when they are stained with a particular type of dye.
This happens when red blood cells are immature because they were released too early from your bone marrow.
These immature cells are called reticulocytes.
Certain blood disorders e.g. anemia can cause your bone marrow to release blood cells early, and these cells show up as polychromasia on a blood test called a peripheral blood smear.
BW
how to identify blood morphology using microscopy?
Hi Curiousjas,
Thank you for your comment.
Importantly, viewing blood smears under the microscope needs to be done shortly after blood collection employing sterile technique (**wearing gloves) from a disinfected site (wiping off 1st drop of blood). Using a high quality clean glass slide (flat, no distortions and corrosion resistant) of 75mm X 25mm and 1mm thickness is ideal. Then place the blood drop 1cm from the end of the slide.
Proceeding with the 45 degree wedge or push slide technique used in manual and automated environments, creates a monolayer blood smear. This is done in a smooth and quick motion. Fixation, staining, washing and air drying are quickly commenced. When staining either Romanowsky, Wright’s or Giemsa stain are used or a combination thereof, usually simply Wright’s stain.
Read More
BW,
Much useful information.
Hi Mujibur,
Thank you for your comment.
Pleased that you found this article helpful.
BW
it was a worth reading. the content was informative. I have enriched myself reading this one.
Dear Dr Hoque,
Thank you for your comment.
Pleased that you found this article helpful.
BW
Very inspiratif, thank you
Hi Deden,
Many thanks for your comment.
Pleased that you found this article helpful.
BW,
Hello Doctor,
I had been hospitalized after closed head impact injury, severe pulsating headache through my skull that felt like I was dying. I had sweating fever, vomiting, slurring, cognitive decline for ten days with walking instability-turned-lower leg paralysis, prior to becoming comatose and given 5% survival rate to live. What started as as small collection LSD amassed to becoming acute on chronic an bilateral subdural hematomas while awaiting craniotomy; I am trying to make sense of the bloodwork resulting in Abnormal RBC Morphology daily. I have read that the presence of slightly immature red blood cells may indicate hemorrhage, but not much more than that. I am trying to understand how Abnormal RBC Morphology results equate to the head trauma if at all.
Hi Jacqueline,
Thank you for your comment. I am sorry to hear about your recent sufferings following a head injury. I hope you are feeling better now.
Regarding your question about the immature RBCs on your blood film, it is not clear from your comment which type of immature RBCs you were referring to. Did you mean reticulocytes and polychromasia or NRBCs?
Reticulocytes are immature RBCs which increase in numbers following hemorrhage and is a physiological body response which shouldn’t cause concerns. However, NRBCs shouldn’t be seen on the blood smear under normal conditions.
Please let me know which type of immature RBCs you were referring to so I can provide you with the correct information. If you have any other questions or concerns, please don’t hesitate to ask.
Best regards,
Dr M Abdou
Amazing! I trained in 1958 & looking at all your slides I’m amazed at how much I retained in spite of not working in Haematology since I had the children in the late ’70s. The only change I can see is the current term for poikilocytes! Cheered me up no end. I was originally searching for the name of the arthritis linked disorder that produced a sort of nuclear lysis in white cells – it is on the tip of my tongue but evading me. The method for testing in those days was a right pain & I’m pretty sure there is a nice simple test for it now. Thank you for making my day.
Hi Christine,
Thank you for your comment.
I’m glad to hear that you found the article interesting and that you’ve regained your knowledge of blood morphology.
It’s possible that you were referring to systemic lupus arthritis and LE cells.
If I’m right here is a link to the LE cells
Best regards,
Dr M Abdou
Thank you so much for this article! I’m a biomedical sciences student and this helped a lot for me to identify different cells and different diseases based on cell morphology. I appreciate all the effort you put in and I hope you are doing very well in life!
Hi Chloe,
Thank you for your comment.
I’m glad to hear that you found this article helpful.
Best wishes,
Dr. M Abdou
Hi.
My hemoglobin was 8.3. I was treated for a year and my hemoglobin and iron stores returned to normal. A year after stopping supplements my hemoglobin was back down to 8.2. I took iron supplements again for 1 month and retested. My CBC then showed hemoglobin 11.4, normal lymphocytes, ABN lymphocytes high, anisocytosis 4+, elliptocytes +4, RBC morphology abnormal: tear drop cells present, Dimorphic RBC population present.
I was referred to a hematologist. I’m wondering if this is necessary or if my results are just due to having iron deficiency anemia.
Hi Jen,
Thank you for your comment.
While the abnormalities could be related to your known iron deficiency, it would be advisable to consult a hematologist due to the high lymphocyte count and presence of tear drop red cells.
Best wishes,
Dr. M Abdou
Hi,
I was diagnosed with iron deficiency anemia over two years ago. My hemoglobin was 8.3. After being on iron supplements for 6 months my hemoglobin and iron stores were back to normal, so iron supplements were stopped.
Two months ago I had routine bloodwork done and my hemoglobin level was 8.2. I was put back on iron supplements again and re-test a month later. Those test results showed hemoglobin 11.4, MCV, MCH, MCH, absolute granulocytes, and absolute monocytes all low, RDW unable to report due to abnormal histogram, RBC morphology: tear drop cells and Dimorphic RBC population present, normal lymphocytes but high ABN lymphocytes, platelet evaluation: adequate Large Platelets present, Anisocytosis 4+, and Elliptocytes 4+.
I have now been referred to a hematologist. Could all of these abnormal results just due to the iron deficiency anemia?
Thank you
I recently had CBC w/diff and manual diff drawn. CBC was wnl, but my manual diff showed 3-7/HFP abnormal teardrop cells and poikilocytosis abnormal. Can you tell me what this means and if there should be cause for concern?
Thanks so much,
Valerie
Hi Valerie,
A few teardrop cells and poikilocytosis on a manual differential can be non-specific but may indicate an underlying bone marrow or red blood cell disorder.
If your CBC is otherwise normal and you have no symptoms, this may not be concerning.
However, if you have anemia, other abnormal blood findings, or symptoms like fatigue or weight loss, further evaluation may be warranted.
Best wishes,
Dr M Abdou
Hello,
Over the course of the last 8 months, I have had 2 CBC`s drawn which I have a diagnosis of behcets so I get frequent blood drawn. Both times I have had myelocyctes, metamyelocytes, and promyelocytes in my blood. Level was .3 both times. CBC was otherwise remarkable. PCP does not seem concerned. Any thought?? Thanks
Hello Julia,
Thank you for sharing your results.
The presence of myelocytes, metamyelocytes, and promyelocytes in the peripheral blood is known as a “left shift,” and it usually reflects the bone marrow releasing immature white cells (granulocytes) into the circulation.
In most cases, a small number of immature myeloid cells (such as 0.3%) can be seen in association with infection, inflammation, recovery from marrow suppression, or as part of a reactive process — especially in the context of chronic inflammatory conditions such as Behçet’s disease. Persistent low-level immature granulocytes without other blood count abnormalities often indicate a benign/reactive process rather than a primary bone marrow disorder.
However, if these findings persist on multiple blood films, or if there are any changes in your blood counts (anaemia, low platelets, high white cells), or symptoms such as night sweats, weight loss, or unexplained fevers, a formal haematology review and peripheral smear examination would be appropriate to rule out chronic myeloid or other myeloproliferative disorders. If there is concern about an underlying bone marrow pathology, it would also be reasonable to check a peripheral blood BCR-ABL transcript to exclude chronic myeloid leukaemia.
I would further recommend arranging CRP, ESR, LDH, and an abdominal ultrasound to assess for systemic inflammation and organomegaly.
Since you already have regular follow-up for Behçet’s disease, I would suggest discussing these points with your physician to decide on the most appropriate next steps.
Kind regards,
Dr M Abdou
Good evening,
I had a CBC a few day ago which was WNL for the most part however it said myelocytes, metamyelocytes and promyelocytes were seen. Level was .3 I had a CBC appx 8 months ago and the same was seen. My PCP doesn’t seem concerned. Your thoughts?