Essential Thrombocythemia
Essential thrombocythemia (ET) is a myeloproliferative neoplasm (MPN) characterized by megakaryocyte hyperplasia and thrombocytosis due to a multipotent hematopoietic stem cell clonal abnormality.
From the genetic perspective, ET patients harbour mutations in JAK2 (50–60%), CALR (15–30%) and MPL (1–5%) genes.
Platelet survival is normal in ET.
Approximately one-third of patients with ET are asymptomatic at diagnosis.
ET usually occurs with bimodal peaks between ages 50 and 70 years and a separate peak among young females.
Patients particularly suffer from combined bleeding and clotting tendency, as well as the general features of myeloproliferative disorders such as anemia, headaches, and malaise.
Bleeding is usually mild and manifests as epistaxis, easy bruising, or GI bleeding. However, thrombosis is a major cause of morbidity and mortality in ET.
Recent studies indicate an elevated leukocyte count is a major independent risk factor for thromboses.
Erythromelalgia (burning pain in hands and feet, with warmth, erythema, and sometimes digital ischemia) may occur.
Mild splenomegaly occurs in < 50% of patients. Hepatomegaly may rarely occur.
Myelofibrosis is common.
Transformation to Acute Myeloid Leukemia (AML) occurs in 0.6-5% of patients with ET; the risk may be comparable to that of the healthy population.
Diagnosis:
The diagnosis can be difficult to differentiate from secondary (reactive) thrombocytosis and is partly a diagnosis of exclusion*.
Platelets are acute-phase reactants; therefore, they increase in response to various stimuli, including systemic infections, inflammatory conditions, bleeding, and tumors.
There is current diagnostic controversy, including heterogeneity within ET and the phenotypic overlap between ET, polycythemia rubra vera, and primary myelofibrosis.
*FBC and peripheral blood smear. The platelet count can be >1,000,000/μL but may be as low as 450,000/μL. The peripheral smear may show platelet aggregates, giant platelets, and megakaryocyte fragments.
*Exclusion of causes of secondary thrombocytosis, e.g. Chronic inflammatory disorders, hemorrhage, acute infection, iron deficiency, splenectomy, and cancer.
*Cytogenetic studies, including Philadelphia chromosome or BCR-ABL assay, should be done.
*JAK2 mutation by PCR. The JAK2 V617F mutation occurs in about 50-60% of patients and, if present, helps distinguish ET from other causes of thrombocytosis.
Polycythemia vera is mainly related to JAK2 mutations, whereas a wider mutational spectrum is detected in ET with mutations in JAK2, the thrombopoietin (TPO) receptor (MPL), and the calreticulin (CALR) genes.
Overall, there is general agreement that 10–30% of all ET patients are wild-type for JAK2, CALR, and MPL mutations when the molecular analyses are performed with DNA from isolated granulocytes or peripheral blood. This subgroup of patients, called ‘triple-negative‘ (TN), has not been extensively studied regarding the presence of JAK2, CALR, and MPL mutations in platelets and RNA from granulocytes.
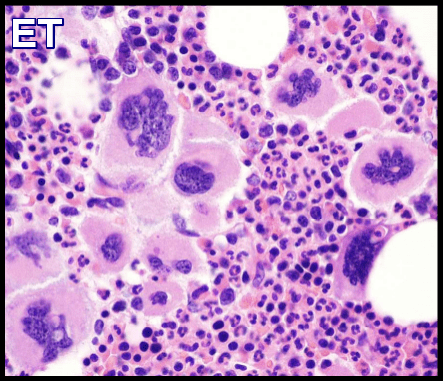
*Possibly bone marrow aspirate and trephine biopsy. The bone marrow shows increased megakaryocyte numbers and drifts of platelets. Megakaryocytes display a spectrum of morphology with predominant large megakaryocytes with hyperlobated nuclei and abundant cytoplasm. The bone marrow iron is present and reticulin is generally not increased.

Atypical megakaryocytes are evident on the bone marrow aspirate smear, including quite large forms with abundant cytoplasm, and deeply lobulated nuclei.
To distinguish ET from other myeloproliferative disorders that cause thrombocytosis, the diagnosis of ET requires a normal Hct, MCV, and iron studies to outrule PRV; the absence of the Philadelphia chromosome and BCR-ABL translocation to outrule CML; and absence of teardrop-shaped RBCs to out rule idiopathic myelofibrosis, although there may be significant increase in bone marrow fibrosis.
Treatment:
Treatment in patients with ET should be individualized based on risk factors for thrombo-hemorrhagic complications. Risk factors include the following:
- Age 60 years or older.
- History of thrombosis or transient ischemic attack.
- Platelet count >1.5 million/µL.
- Obesity.
- Cardiovascular risk factors such as smoking, hypertension, and hypercholesterolemia.
- Markers of hypercoagulability such as factor V Leiden and antiphospholipid antibodies.
Treatment includes the following:
- Observation may be appropriate for low-risk patients (ie, those lacking any of the above risk factors).
- Low-dose Aspirin.
- Platelet-lowering drugs i.e. cytoreductive agents (eg, hydroxyurea, anagrelide).
- Plateletpheresis is rarely used in emergencies to achieve a rapid decrease in platelet counts in the setting of acute thrombosis and/or marked thrombocytosis.
- Rarely cytotoxic agents.
- Rarely interferon.
- Rarely stem cell transplantation.
Summary:
Essential thrombocythemia (ET) is a chronic myeloproliferative neoplasm characterized by sustained thrombocytosis and megakaryocytic hyperplasia in the bone marrow, leading to an increased risk of thrombosis, microvascular symptoms, and, less commonly, bleeding. Most cases are driven by mutations in JAK2, CALR, or MPL, which alter signaling pathways that regulate hematopoiesis. JAK2 V617F is the most frequent mutation, found in approximately 50–60% of patients, while CALR mutations account for 15–30% and MPL for 1–5%. When none of these driver mutations are detected, the disease is classified as triple-negative ET, a less common variant that may demonstrate different biological behaviour and risk profiles. Diagnosis requires persistent thrombocytosis, exclusion of reactive causes, and bone marrow morphology consistent with ET, supported by molecular testing to identify (or confirm the absence of) JAK2, CALR, and MPL mutations.
Questions and Answers:
What is Essential Thrombocythemia?
Essential thrombocythemia is a chronic myeloproliferative neoplasm characterized by persistently elevated platelet counts, bone marrow megakaryocytic hyperplasia, and an increased risk of arterial and venous thrombosis, microvascular symptoms, and, occasionally, bleeding.
What causes Essential Thrombocythemia?
Most cases are driven by somatic mutations in JAK2, CALR, or MPL, which activate signaling pathways that promote uncontrolled megakaryocyte proliferation and platelet production. A minority of patients have triple-negative ET without detectable driver mutations.
What are the symptoms of Essential Thrombocythemia?
Common symptoms include headaches, visual disturbances, erythromelalgia, dizziness, tingling in the extremities, fatigue, and symptoms related to thrombosis such as stroke, DVT, or MI. Some patients remain asymptomatic and are diagnosed incidentally.
What is erythromelalgia in Essential Thrombocythemia?
Erythromelalgia is a microvascular complication of ET characterized by burning pain, warmth, and erythema of the hands or feet. It responds dramatically to aspirin, reflecting platelet-mediated microvascular occlusion.
How is triple-negative Essential Thrombocythemia defined?
Triple-negative ET refers to ET cases without mutations in JAK2, CALR, or MPL. These patients may have different clinical characteristics and generally require detailed bone marrow morphology and exclusion of reactive thrombocytosis for diagnosis.
How is Essential Thrombocythemia diagnosed?
Diagnosis requires persistent thrombocytosis, exclusion of secondary causes, bone marrow morphology showing large atypical megakaryocytes, and molecular testing for JAK2, CALR, or MPL mutations. Additional tests include CBC, ferritin, CRP, and BCR-ABL1 when appropriate.
What risks are associated with Essential Thrombocythemia?
The main risks include arterial and venous thrombosis, microvascular ischemic symptoms, bleeding in patients with extreme thrombocytosis, and progression to myelofibrosis or acute leukemia in a minority of cases.
How is thrombotic risk stratified in Essential Thrombocythemia?
Risk stratification is based on age, history of thrombosis, and JAK2 mutation status. Patients are categorized into low-risk or high-risk groups to guide decisions about antiplatelet therapy and cytoreduction.
What defines low-risk Essential Thrombocythemia?
Low-risk ET is defined by age under 60, absence of prior thrombosis, and typically CALR-positive or JAK2-negative disease. These patients usually receive low-dose aspirin unless contraindicated, and cytoreductive therapy is not routinely required.
What defines high-risk Essential Thrombocythemia?
High-risk ET is defined by age over 60, history of arterial or venous thrombosis, or the presence of a JAK2 mutation that increases thrombotic risk. These patients commonly require cytoreductive therapy such as hydroxyurea or interferon in addition to low-dose aspirin.
How is Essential Thrombocythemia treated?
Treatment focuses on preventing thrombosis and managing symptoms. Low-dose aspirin is used for most patients, while cytoreductive therapy—such as hydroxyurea or interferon—is reserved for high-risk patients or those with significant symptoms.
What does the bone marrow look like in Essential Thrombocythemia?
Bone marrow examination shows increased numbers of large, mature megakaryocytes with hyperlobulated nuclei and loose clustering, without significant granulocytic or erythroid proliferation, consistent with ET morphology.
What does the blood film show in Essential Thrombocythemia?
Peripheral blood film typically shows marked thrombocytosis with large and giant platelets. Leukocytosis may be present, but red cell morphology usually remains normal.
Can Essential Thrombocythemia progress to myelofibrosis or leukemia?
ET can evolve into post-ET myelofibrosis or, rarely, acute myeloid leukemia. This risk is low but increases with disease duration, age, long-term cytoreductive therapy, and certain mutational profiles.
References:
Thiele J, Kvasnicka HM. The 2008 WHO diagnostic criteria for polycythemia vera, essential thrombocythemia, and primary myelofibrosis. Curr Hematol Malig Rep. 2009;4:33–40.
Harrison CN, Bareford D, Butt N, et al; British Committee for Standards in Haematology. Guideline for investigation and management of adults and children presenting with a thrombocytosis. Br J Haematol. 2010;149:352–375.
Fenaux P, Simon M, Caulier MT, et al. Clinical course of essential thrombocythemia in 147 cases. Cancer. 1990;66:549–556.
Toyama K, Karasawa M, Yamane A, Irisawa H, Yokohama A, Saitoh T, et al. JAK2-V617F mutation analysis of granulocytes and platelets in chronic myeloproliferative disorders: advantages of platelet testing. Br J Haematol. 2007;139:64–69.
Cazzola M, Kralovics R. From Janus kinase 2 to calreticulin: the clinically relevant genomic landscape of myeloproliferative neoplasms. Blood. 2014;123:3714–3719.
Vora AJ, Lilleyman JS. Secondary thrombocytosis. Arch Dis Child. 1993;68(1):88–90.
Hoffman R, Benz EJ Jr, Shattil SJ, et al, eds. Primary thrombocythemia. In: Hematology: Basic Principles and Practice. 3rd ed. Philadelphia: Churchill Livingstone; 2000:1188–1204.
Courville EL. Essential Thrombocythemia. Available from: https://imagebank.hematology.org/reference-case/16/essential-thrombocythemia
Campbell PJ, Green AR. The myeloproliferative disorders. N Engl J Med. 2006;355(23):2452–2466. doi:10.1056/NEJMra063728
Rumi E, Pietra D, Ferretti V, et al. JAK2 or CALR mutation status defines subtypes of essential thrombocythemia with different clinical course and outcomes. Blood. 2014;123(12):1544–1551. doi:10.1182/blood-2013-11-539098
Keywords:
essential thrombocythemia, ET, thrombocythemia, myeloproliferative neoplasm, JAK2 mutation, CALR mutation, MPL mutation, triple negative ET, platelet overproduction, megakaryocyte hyperplasia, thrombocytosis, erythromelalgia, ET blood film, bone marrow aspirate, ET diagnosis, ET treatment, thrombosis risk, cytoreductive therapy, hydroxyurea, hydroxycarbamide, interferon therapy, aspirin therapy ET, ET complications, ET prognosis, ET vs PV, ET vs prefibrotic MF, ET risk stratification, low-risk ET, high-risk ET, WHO diagnostic criteria ET, ET bone marrow morphology, ET platelet morphology, ET clinical features

Request Online Consultation With Dr M Abdou
Fee: US$100
Secure payment via PayPal (credit and debit cards accepted)
Pay Now





well organized topic.