Polycythemia Rubra Vera

Polycythemia Rubra Vera: Clinical presentation with red skin, hepatosplenomegaly, and hypercellular marrow.
Polycythemia Rubra Vera (PRV) is a chronic myeloproliferative neoplasm characterized by an increased red blood cell (RBC) production and RBC mass often manifested as an increased hematocrit (Hct). There is a malignant transformation of the multipotential stem cell with excess production of red blood cells in the bone marrow and blood, causing the blood to thicken. The number of white blood cells and platelets may also increase. The extra blood cells may collect in the spleen and cause it to become enlarged. They may also cause bleeding problems and make clots form in blood vessels.
Extramedullary hematopoiesis may occur in the spleen, liver, and other sites with the potential for blood cell formation. There is no Philadelphia chromosome.
The major symptoms are related to hypertension or vascular abnormalities caused by the increased red cell mass. The cause is unknown. The ICD-10-CM Diagnosis Code for Polycythemia Rubra Vera (PRV) or Primary Polycythemia Vera is D45. With currently available treatment, the median survival exceeds 10 years (WHO 2001).
PRV is the most common of the myeloproliferative neoplasms, with incidence increasing with age. PRV may be slightly more common in men. The mean age at diagnosis is around 60 years. The disease is very rare in children.
Clinical Features:
Develop gradually and include:
- Headaches, dizziness, tinnitus and visual problems.
- The face may be red (plethoric), and the retinal veins engorged. The palms and feet may be red, warm, and painful.
- Thrombotic episodes e.g. (neurologic deficits with stroke or transient ischemic attack, DVTs, unilateral vision loss with retinal vascular occlusion).
- Bleeding episodes, especially peptic ulcers.
- Generalized pruritus, particularly after a hot bath.
- Gout.
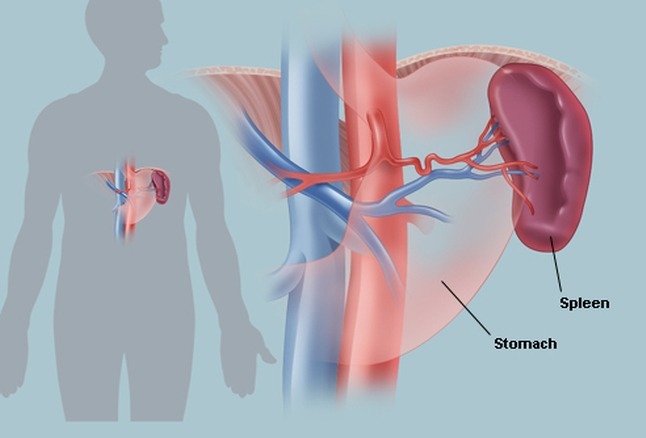
- Hepatomegaly is common, and splenomegaly is present in 75% of cases.
- One-third of patients are hypertensive.
- Hypermetabolism can cause low-grade fevers and weight loss and suggests disease progression.
Investigations:
- Raised Hb.
- Raised red cell mass. This is essential for the diagnosis to differentiate it from pseudo-polycythemia (relative polycythemia) due to a low plasma volume.
- White cell count is usually raised.
- Platelet count is often raised.
- NAP score is raised.
- JAK2 V617F mutation.
- JAK2 exon 12 mutation.
- CXR.
- Abdominal USS.
- In some cases, the diagnosis of PRV must be made by carefully excluding Secondary Polycythemia. This includes a CXR, IV urogram, blood gases, Hb electrophoresis and O2 dissociation curve.
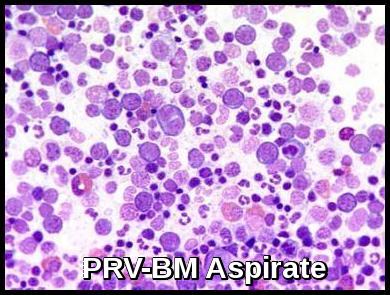
Polycythemia Rubra Vera: Hypercellular bone marrow aspirate with increased erythroid and myeloid precursors.
Diagnostic criteria for PRV as per the 2008 revised World Health Organization (WHO) guidelines include both major and minor criteria. Diagnosis requires the presence of both major criteria and one minor criterion or the presence of the first major criterion together with two minor criteria.
Major WHO criteria are as follows:
- Hemoglobin >18.5 g/dL in men and >16.5 g/dL in women, or other evidence of increased red blood cell volume.
- Presence of JAK2 V617F or other functionally similar mutation, such as JAK2 exon 12 mutation.
Minor WHO criteria are as follows:
- Bone marrow biopsy showing hypercellularity for age with trilineage growth (panmyelosis) with prominent erythroid, granulocytic, and megakaryocytic proliferation.
- The serum erythropoietin level is below the reference range for normal.
- Endogenous erythroid colony formation in vitro.
JAK2 V617F mutation and erythropoietin (EPO) level are key in diagnosing erythrocytosis. If the JAK2 V617F mutation is positive and the EPO level is low, then it confirms the diagnosis of PRV (JAK2 V617F mutation is positive in 97% of PRV patients).
JAK2 mutations also occur in about 60% of patients with essential thrombocythemia. In patients who are positive for JAK2 and whose hemoglobin/hematocrit level is diagnostically equivocal, bone marrow examination is necessary to distinguish the two conditions.
If the JAK2 V617F mutation is absent but the EPO level is low, then testing for JAK2 exon 12 and 13 mutations would be helpful for making a diagnosis of PRV in the 2-3% of PRV patients who are negative for JAK2 V617F mutation.
Patients who are negative for JAK2 mutations and have a normal or high EPO level have secondary polycythemia.
Treatment:
Repeated phlebotomies (venesections) aiming to keep Hct < 0.45

Polycythemia Rubra Vera: Venesection is a first-line treatment to control hematocrit and reduce thrombotic risk.
Cytoreductive therapy e.g. Hydroxyurea (Hydroxycarbamide). In patients who are refractory to or intolerant of hydroxyurea, interferon-alpha can be used as an alternative. Busulfan is also an option for patients older than 65 years.
Aspirin, 75 mg daily, unless contraindicated by major bleeding or gastric intolerance.
Allopurinol to prevent gout.
JAK inhibitors: the JAK1/2 inhibitor Ruxolitinib (Jakavi) is approved for treating PRV in patients who have had an inadequate response to or are intolerant of hydroxyurea.
Radioactive phosphorus (rarely used).
Control of blood pressure if raised.
Untreated PRV is fatal within several years. The long-term risks of PRV include acute leukemia and myelofibrosis transformation, which occurs in fewer than 5% and 10% of patients, respectively, at 10 years. Current treatment modalities do not change these outcomes. Instead, treatment for PRV is intended to decrease the risk of arterial and venous thrombotic events, which could be approximately 20%.
Summary:
Polycythaemia Rubra Vera (PRV) is a chronic myeloproliferative neoplasm marked by uncontrolled overproduction of red blood cells, often accompanied by elevated platelets and white cells. The resulting increase in blood volume and viscosity contributes to symptoms such as headache, pruritus, and erythromelalgia, and significantly raises the risk of thrombosis. Diagnosis relies on detecting the JAK2 V617F mutation—present in approximately 97% of cases—alongside a characteristically low erythropoietin (EPO) level and supportive bone marrow findings. Management focuses on maintaining hematocrit <45% and preventing thrombotic events through therapeutic phlebotomy, low-dose aspirin, and cytoreductive therapy such as hydroxyurea or ruxolitinib in high-risk patients. PRV carries long-term risks including bleeding, myelofibrotic transformation, and progression to acute leukemia, necessitating ongoing monitoring.
Questions and Answers:
What are the early symptoms of Polycythaemia Rubra Vera (PRV)?
Early symptoms commonly include facial redness, headaches, dizziness, blurred vision, pruritus after a hot shower, and fatigue due to increased blood viscosity.
Why does PRV cause red or flushed skin?
PRV leads to excess red-blood-cell mass, increasing blood flow to the skin and causing persistent facial redness, plethoric complexion, and erythromelalgia.
How is PRV diagnosed?
Diagnosis relies on identifying the JAK2 V617F mutation (present in 97% of cases), low EPO levels, hypercellular bone marrow with trilineage growth, and elevated Hb/Hct meeting WHO criteria.
What is the role of the JAK2 mutation in PRV?
The JAK2 V617F mutation drives uncontrolled erythropoiesis and is the key molecular marker, helping confirm PRV and distinguish it from secondary polycythaemia.
Why is blood so thick in PRV?
Excess red blood cells increase blood viscosity, slowing circulation and contributing to symptoms such as headache, tinnitus, visual disturbances, and thrombotic risk.
How is hematocrit controlled in PRV?
Therapeutic phlebotomy is first-line to maintain hematocrit below 45%, reducing blood viscosity and lowering the risk of stroke, DVT, and cardiovascular events.
When is hydroxyurea used in PRV?
Hydroxyurea is prescribed for high-risk patients (age >60 or prior thrombosis) to reduce red-cell and platelet production, lowering thrombotic and symptomatic burden.
What is the purpose of ruxolitinib in PRV management?
Ruxolitinib is used in hydroxyurea-resistant or intolerant patients to control hematocrit, reduce splenomegaly, improve symptoms, and regulate JAK-STAT pathway activation.
Why are PRV patients at risk of thrombosis?
High hematocrit, increased platelets, leukocytosis, and blood hyperviscosity promote clot formation, making arterial and venous thrombosis major complications.
Can PRV progress to myelofibrosis or leukemia?
Yes. Long-standing PRV can evolve into post-PV myelofibrosis or, less commonly, transform to acute myeloid leukemia, especially after prolonged cytotoxic therapy.
Is itching after a hot shower a sign of PRV?
Yes. Aquagenic pruritus is a hallmark symptom caused by abnormal mast-cell activation and increased histamine release.
Does PRV cause enlarged spleen?
Many patients develop splenomegaly due to extramedullary hematopoiesis and increased blood cell turnover.
What lifestyle changes help patients with PRV?
Adequate hydration, regular physical activity, smoking cessation, weight control, and avoiding iron supplementation help reduce symptoms and thrombotic risk.
How often should PRV patients be monitored?
Regular monitoring includes CBC, hematocrit level, symptom assessment, and spleen size evaluation, with follow-up frequency depending on risk category and therapy.
References:
Adamson JW, Fialkow PJ, Murphy S, et al. Polycythemia vera: stem-cell and probable clonal origin of the disease. N Engl J Med. 1976;295:913-916.
James C, Ugo V, Le Couedic JP, et al. A unique clonal JAK2 mutation leading to constitutive signalling causes polycythaemia vera. Nature. 2005;434:1144-1148.
Landolfi R, Marchioli R, Kutti J, et al. Efficacy and safety of low-dose aspirin in polycythemia vera. N Engl J Med. 2004;350:114-124.
The Patient Story. Polycythemia Vera Skin Conditions. Available at: https://thepatientstory.com/mpn/polycythemia-vera/symptoms/skin
Vannucchi AM, Kiladjian JJ, Griesshammer M, et al. Ruxolitinib versus standard therapy for the treatment of polycythemia vera. N Engl J Med. 2015;372:426-435.
Marchioli R, Finazzi G, Specchia G, et al. Cardiovascular events and intensity of treatment in polycythemia vera. N Engl J Med. 2013;368:22-33.
Tefferi A, Vardiman JW. Classification and diagnosis of myeloproliferative neoplasms: The 2008 World Health Organization criteria and point-of-care diagnostic algorithms. Leukemia. 2008;22(1):14-22.
Arber DA, Orazi A, Hasserjian R, et al. The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood. 2016;127(20):2391-2405.
Barbui T, Barosi G, Birgegard G, et al. Philadelphia-negative classical myeloproliferative neoplasms: critical concepts and management recommendations from European LeukemiaNet. J Clin Oncol. 2011;29(6):761-770.
Keywords:
Polycythemia Vera, Polycythemia Rubra Vera, PRV, polycythemia vera symptoms, facial redness polycythemia vera, polycythemia vera itching, aquagenic pruritus PV, polycythemia vera diagnosis, JAK2 V617F polycythemia vera, low EPO polycythemia vera, polycythemia vera treatment, venesection polycythemia vera, phlebotomy polycythemia vera, hematocrit control PV, polycythemia vera aspirin, hydroxyurea polycythemia vera, hydroxycarbamide polycythemia vera, ruxolitinib polycythemia vera, Jakafi PRV treatment, polycythemia vera prognosis, polycythemia vera life expectancy, polycythemia vera complications, polycythemia vera thrombosis risk, polycythemia vera hypercellular marrow, bone marrow aspirate polycythemia vera, PRV bone marrow findings, erythrocytosis PV, polycythemia vera WHO criteria, PRV diagnostic criteria, PRV vs secondary polycythemia, polycythemia vera lab values, hemoglobin high polycythemia vera, hematocrit high PV, platelet count high PV, PRV leukocytosis, polycythemia vera splenomegaly, polycythemia vera hepatosplenomegaly, post polycythemia vera myelofibrosis, post polycythemia vera AML, polycythemia vera cancer, polycythemia vera ICD10, polycythemia vera management guidelines, European LeukemiaNet PV, polycythemia vera clotting risk, erythromelalgia PRV, polycythemia vera hot shower itching, polycythemia vera fatigue, polycythemia vera headache, polycythemia vera dizziness, polycythemia vera treatment guidelines, polycythemia vera images, polycythemia vera skin, polycythemia vera face redness


Request Online Consultation With Dr M Abdou
Fee: US$100
Secure payment via PayPal (credit and debit cards accepted)
Pay Now


Was wondering if having primary PCV can contribute to bone weakening ? I had a seizure in March and found out later that I had some compression fractures in my spine. I had no other trauma. I’ve been in pain since March but the fractures, from what one spine specialist told me, aren’t large enough to be treated. I’m awaiting a new CT scan to see if the fractures have gotten better or worse since my July scan.
Will any change in diet help my weakened bones ?
Thanks
Hi Maryann,
Thank you for your comment.
First, do you have osteoporosis?
I don’t know how old are you, but if you are older than fifty I would recommend DEXA scan.
It would be also helpful to check your ESR, myeloma screen and Vitamin D blood levels.
I don’t think there is a direct link between PRV and the compression fractures in your spine!
If you are vitamin D deficient you might benefit from some oral vitamin D and Calcium supplements.
Best wishes,
Author
Hello. are labs showing WBC: 10.3, RBC: 5.53, Hemoglobin: 16.1, Hematocrit 49.4, Platelets: 342, Neutrophils: 7.6 Basophils: 0, Eosinophils: .2, Lymphocytes 1.9, Monocytes: .7 indicitive of PVR or any other blood cancer or serious condition? Thank you. – Chris
Also, do these numbers only go up with PV and similar blood cancers, and would you see an increase in values over 70 days?
Hi Chris,
Thanks for your comment.
The figures you mentioned are likely those of 2ry polycythaemia, not primary polycythaemia (PRV).
If you smoke or have a lung disease or are dehydrated that may explain your mild polycythemia.
Give up smoking if you smoke and maintain adequate hydration and repeat your CBC after 8 weeks.
BW,
Hello. Thank you so much for responding. If I may, there is a little more to the story and I would greatly appreciate your further thoughts in case your opinion changes. I had 3 labs done starting 70 days ago for a physical. I am a 48 year old male.
The first lab showed (SEPT 4):
WBC: 10.32, RBC: 5.76, Hemoglobin: 17, Hematocrit: 50.3, Netrophils: 74.6.
we repeated the labs 3 weeks later because I am a very nervous patient (SEPT 20):
WBC: 11.7, RBC: 5.52, Hemoglobin: 16.6, Hematocrit: 49.7, Neutrophils: 9.0
We then waited about two months and repeated the labs again and got the numbers I shared with you in my original post (NOV 17):
WBC: 10.3, RBC: 5.53, Hemoglobin: 16.1, Hematocrit: 49.3, Neutrophils: 7.6.
I am a very nervous individual and patient, so thinking about this does cause me daily stress. I do not smoke and do not live at a high altitude ( Michigan ). The good news is the numbers do seem stable or going down after close to 3 months from the initial lab work. But they are still elevated. I have had a very stressful year, losing both my parents and dealing with their house, so I have had some fatigue and perhaps some blurry vision in my right eye, but those may or may not be connected. Dehydration might factor, as it is possible I hadn’t been consuming as much water as I should. Thanks again for your incite and time. Best – Chris
Hi Chris,
Thanks for the update.
As per my previous reply, your mild polycythemia is likely reactive/relative and you should rest assured.
However, if you would like an additional piece of investigation to do in view of your anxiety, I would suggest checking your blood for the JAK2 mutation. If negative (as expected) you would just need to keep yourself well hydrated.
BW,
Sorry the neutrophils in the first lab ( SEPT 4) were 74.6%, or 7.71. Just to clarify.
Also, sorry for the third post, but forgot to include these values.
test 1, platelets: 354, test 2, platelets: 368, test 3, platelets: 342.
Thanks so much for your reply, that is reassuring. One last and quick question, would 70 days between the first and latest CBC panel generally be enough time to see a trend upward in the values? Thanks again, and I do see a hematologist on Monday.. As you have already detected, I am nervous and this weighs on me.
Hello, thank you for your advise. I did pursue a JAK12 test after talking to my doctor and it was negative, at least V617F. My spleen was also normal upon ultrasound. Thanks again, and I hope this fairly confidently rules out PRV.
Hello. Thank you again for you time. I did get one more test back and I was still a bit concerned. I had my erythropoietin tested and my value was 5. I know from Google that lower values are associated with primary Polycythemia, not secondary.
Even though I had a normal spleen reading and my JAK2V617F was negative,do I need to further test for what they call Exon 12 and 14 mutations? What are the chances I have PVR given my value of Erythropoietin, in your opinion?
As always, I appreciate your thoughts and your time.