Principles of Cancer Therapy
The principles of cancer therapy (which can be curative or palliative) are fundamentally based on the tumor’s characteristics and the patient’s ability to tolerate the treatment. Modern anticancer therapy often includes a combination of modalities like surgery, chemotherapy, immunotherapy, radiation therapy, and/or targeted therapy.
Cancer treatment is one of the greatest healthcare concerns globally. Once cancer is confirmed, further diagnostics to assess the tumor grade and tumor stage are required, along with a comprehensive assessment of the patient’s general health (e.g., nutritional status, social support, and mental health).
Complications are frequent, arising from either cancer progression or the treatment itself; therefore, proactive management (e.g., prophylactic antiemetic regimens) is essential to improve patients’ quality of life. The majority of cancer care is managed by oncologists, but all healthcare providers should be familiar with common treatments, complications, and the critical need for multidisciplinary cancer care.
If cancer is suspected, an accurate and comprehensive assessment of both the patient and the disease must be undertaken before a treatment decision is reached.
Assessment of the tumor:
Histological nature of the tumor:
Tumor specimens, usually obtained through fine-needle biopsy, core biopsy, surgical biopsy, or excision of a mass or lesion, are examined to confirm: High-grade, poorly differentiated tumors tend to have a poorer outcome than low-grade, well-differentiated tumors.
The biological behaviour of the tumor:
Tumor markers produced by cancers may be a useful adjunct to histological classification and staging and can be used to influence and monitor the efficacy of treatment. However, tumor markers can be raised in many non-malignant conditions.
Anatomical extent of the tumor:
Usually determined through a combination of clinical, radiological, biochemical and surgical assessment. Routine blood tests including liver function tests and bone profiles may also indicate the presence of metastases.
Curing cancer requires eliminating all cancer cells.
The major modalities of therapy are:
Surgery (for local and local-regional disease):
Surgery is the removal of the tumor and surrounding tissue during an operation. A doctor who specializes in treating cancer using surgery is called a surgical oncologist. Surgery is the oldest type of cancer therapy and remains an effective treatment for many types of cancer today.
The goals of surgery vary. However, it is used for the following reasons:
* To diagnose cancer.
* To remove all or some of the cancer or tumor.
* Find out where the cancer is located.
* Find out if the cancer has spread or is affecting the functions of other organs in the body.
* To restore the body’s appearance or function.
* To relieve side effects.
The location where you have surgery depends on the extent of the surgery and how much recovery you need. You may have surgery in a doctor’s office, clinic, surgery center, or hospital. Outpatient surgery means that you do not need to stay overnight in the hospital before or after surgery. Inpatient surgery means that you do need to stay in the hospital overnight or longer to recover after the surgery.
Types of conventional surgery:
Diagnostic: For most types of cancer, a biopsy is the only way to make a definitive diagnosis. During a surgical biopsy, the surgeon makes a cut called an incision in the skin. Then, he or she removes some or all of the suspicious tissue.
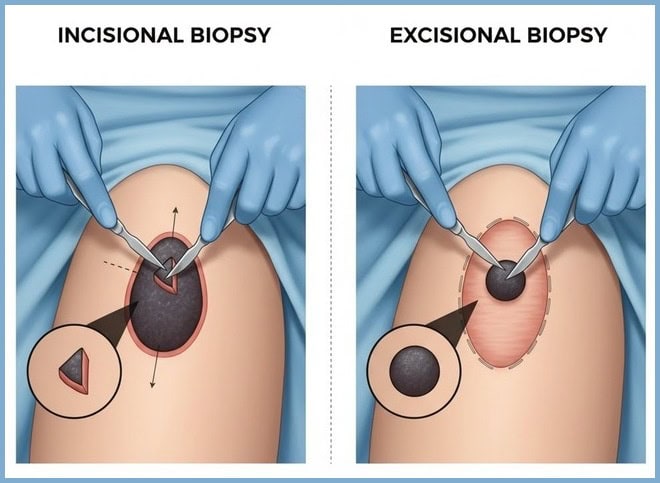
There are 2 main types of surgical biopsies:
* An incisional biopsy is the removal of a piece of the suspicious area for examination.
* An excisional biopsy is the removal of the entire suspicious area, such as an unusual mole or a lump.
After a biopsy, a pathologist uses a microscope to examine the tissue removed. A pathologist is a doctor who specializes in interpreting laboratory tests and evaluating cells, tissues, and organs to diagnose disease. The pathologist provides a pathology report to the surgeon or oncologist, who makes the diagnosis.
Staging: Staging surgery is performed to find out the size of the tumor and if or where it has spread. During this surgery, the doctor often removes some lymph nodes near the cancer to find out if it has spread. Lymph nodes are tiny, bean-shaped organs that help fight infection.
Your health care team uses the results from this surgery along with the results of other tests to guide the treatment options. These tests can also help predict a patient’s prognosis, which is the chance of recovery. The stage of cancer may also be included on the pathology report.
Tumor removal also called curative or primary surgery: Surgery is most commonly used to remove the tumor and some of the surrounding healthy tissue. The tissue surrounding the tumor is called the margin.
Tumor removal may be the only treatment, or it may be combined with Chemotherapy, Radiation therapy or Other treatments, which may be given before or after surgery.
With conventional surgery, the surgeon makes large incisions through skin, muscle, and sometimes bone. In some situations, surgeons can use less invasive surgical techniques. These less-invasive techniques may speed recovery and reduce pain afterwards.
Debulking: Sometimes, the surgeon cannot remove the entire tumor or removing it might cause too much damage to the body. In these situations, surgery is used to remove as much of the tumor as possible. Other treatments, such as radiation therapy or chemotherapy, may also be used to shrink the remaining cancer. These treatments may also be given before surgery to help shrink the tumor so it can be removed.
Palliation: Palliative surgery is used to relieve side effects caused by a tumor. It plays an important role in improving the quality of life for patients with advanced cancer or widespread disease. For example, surgery may be used to relieve pain or restore physical function when a tumor causes the following complications:
* Pressure on a nerve or the spinal cord.
* Blockage of the bowel or intestines.
* Pressure or blockage elsewhere in the body.
* Stop bleeding. Certain cancers are more likely to cause bleeding, including cancers in areas with a high concentration of blood vessels, such as the uterus, cancers in organs that are fragile and can easily bleed when food and waste products pass through, such as the oesophagus, stomach, and bowel. Bleeding may also be a side effect of some drugs used to treat cancer. When surgery is needed to stop bleeding, a common technique is suture ligation. This surgery involves tying blood vessels using surgical thread.
* Insert a feeding tube or tubes that deliver medications if the cancer or treatment has made it difficult to eat. A feeding tube is inserted directly into the stomach or intestine through the abdominal wall. Or, a tube may be inserted into a vein to deliver pain medication or chemotherapy.
* Prevent broken bones. Bones weakened by cancer or cancer treatment can break easily and often heal slowly. Inserting a metal rod may help prevent fractures of weak bones and relieve pain during healing.
Reconstruction: After primary cancer surgery, surgery may be an option to restore the body’s appearance or function. This is called reconstructive or plastic surgery. Reconstructive surgery may be done at the same time as surgery to remove the tumor. Or, it may be done later after a person has healed or received additional treatment. Examples of reconstructive surgery include breast reconstruction after a mastectomy and surgery to restore a person’s appearance and function after surgery to the head and neck area.
Prevention: Some surgery is performed to reduce the risk of developing cancer. For example, doctors often recommend the removal of precancerous polyps in the colon to prevent colon cancer. In addition, women with a strong family history of breast or ovarian cancers or known mutations to the BRCA1 and BRCA2 breast and ovarian cancer genes may decide to have a mastectomy or oophorectomy to lower the risk of developing breast or ovarian cancer. A mastectomy is the removal of a breast. An oophorectomy is the removal of the ovaries.
Types of minimally invasive surgery:
As mentioned above, conventional surgery often requires large incisions. In some situations, a surgeon can use one or more small incisions. These surgeries typically result in shorter recovery times and less pain afterwards. Below are some examples of minimally invasive procedures and surgeries:
Laparoscopic surgery: The doctor performs surgery through small incisions in the skin using a thin, lighted tube with a camera. The term laparoscopy refers to a minimally invasive surgery of the abdomen. The terms, mediastinoscopy and thoracoscopy are used when the same type of procedure is performed in the chest. Laparoscopic surgery may also be done with robotic assistance for some surgeries. During this type of surgery, the surgeon directs robotic instruments to perform the surgery. This surgical procedure may be used for the kidneys, a man’s prostate, or a woman’s uterus or ovaries.
Laser surgery: The doctor uses a narrow beam of high-intensity light to remove cancerous tissue.
Cryosurgery: The doctor uses liquid nitrogen to freeze and kill abnormal cells.
Microscopically controlled surgery: The dermatologist shaves off skin cancer, one layer at a time, until all cells in a layer appear to be normal cells when viewed under a microscope.
Endoscopy: The doctor inserts a thin, flexible tube with a light and camera on the tip into the body. This device is called an endoscope. It may be inserted into the mouth, rectum, or vagina to examine the internal organs. During an endoscopic procedure, it is possible to remove samples of potentially abnormal tissue for further examination. Learn more about types of endoscopy.
Radiation therapy (for local and local-regional disease):
Radiation therapy can cure many cancers, particularly those that are localized or
that can be completely encompassed within the radiation field.
Radiation therapy plus surgery (for head and neck, laryngeal, or uterine cancer) or combined with chemotherapy and surgery (for sarcomas or breast, oesophagal, lung, or rectal cancers) improves cure rates and allows for more limited surgery as compared with traditional surgical resection.
Radiation therapy can provide significant palliation when cure is not possible:
* For brain tumors: Prolongs patient functioning and prevents neurologic complications.
* For cancers that compress the spinal cord: Prevents progression of neurologic deficits.
* For superior vena cava syndromes: Relieves venous obstruction.
* For painful bone lesions: Usually relieves symptoms.
Radiation cannot destroy malignant cells without destroying some normal cells as well. Therefore, the risk to normal tissue must be weighed against the potential gain in treating the malignant cells. The outcome of a dose of radiation depends on numerous factors,
including:
* Nature of the delivered radiation (mode, timing, volume, dose).
* Properties of the tumor (cell cycle phase, oxygenation, molecular properties, inherent sensitivity to radiation).
In general, cancer cells are selectively damaged because of their high metabolic and proliferative rates. Normal tissue repairs itself more effectively, resulting in a greater net destruction of tumor.
Important considerations in the use of radiation therapy include the following:
* Treatment timing (critical)
* Dose fractionation (critical)
* Normal tissue within or adjacent to the proposed radiation field
* Target volume
* Configuration of radiation beams
* Dose distribution
* Modality and energy most suited to the patient’s situation
Treatment is tailored to take advantage of the cellular kinetics of tumor growth, to maximize damage to the tumor while minimizing damage to normal tissues.
Radiation therapy sessions begin with the precise positioning of the patient. Foam casts or plastic masks are often constructed to ensure exact repositioning for serial treatments.
Laser-guided sensors are used. Typical courses consist of large daily doses given over 3 weeks for palliative treatment or smaller doses given once/day 5 days/week for 6 to 8 weeks for curative treatment.
Types of radiation therapy:
There are several different types of radiation therapy.
External beam radiation therapy can involve photons (gamma radiation), electrons, or protons. Gamma radiation using a linear accelerator is the most common type of radiation therapy. Conformal technology, which reduces scatter at the field margins, can limit the radiation dose to adjacent normal tissue.
Electron beam radiation therapy has little tissue penetration and is best for skin or
superficial cancers. Different electron energies are used based on the desired depth of penetration and type of tumor.
Proton therapy, although limited in availability, has advantages over gamma radiation therapy in that it deposits energy at a depth from the surface, whereas gamma radiation damages all tissues along the path of the beam.
Proton beam therapy also can provide sharp margins that may result in less injury to immediately adjacent tissue and is thus particularly useful for tumors of the eye, the base of the brain, and the spine.
Stereotactic radiation therapy is radiosurgery with precise stereotactic localization of a tumor to deliver a single high dose or multiple fractionated doses to a small intracranial or other target. It is frequently used to treat metastatic tumors in the CNS. Advantages include complete tumor ablation where conventional surgery would not be possible and minimal adverse effects. Disadvantages include limitations involving the size of the area that can be treated and the potential danger to adjacent tissues because of the high dose of radiation. In addition, it cannot be used in all areas of the body. Patients must be immobilized and the area kept completely still.
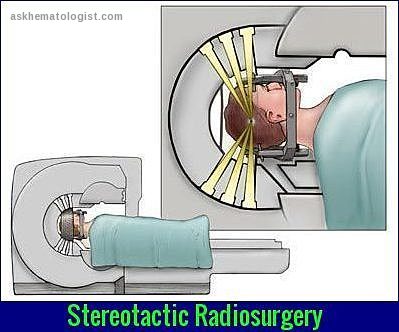
Illustration of stereotactic radiosurgery showing precise radiation beams directed at a brain lesion.
Brachytherapy involves the placement of radioactive seeds into the tumor bed itself (eg, in the prostate or cervix). Typically, placement is guided by CT or ultrasonography. Brachytherapy achieves higher effective radiation doses over a longer period than could be accomplished by the use of fractionated, external beam radiation therapy.

Clinical image showing HDR brachytherapy for prostate cancer using transperineal applicators for precise internal radiation delivery.

X-ray image demonstrating multiple radioactive seed implants used in permanent prostate cancer brachytherapy.
Systemic radioactive isotopes can direct radiation to cancer in organs that have specific receptors for uptake of the isotope (ie, radioactive iodine for thyroid cancer) or when the radionuclide is attached to a monoclonal antibody (eg, iodine-131 plus tositumomab for non-Hodgkin lymphoma). Isotopes can also accomplish palliation of generalized bony metastases (ie, radiostrontium for prostate cancer).
Other agents or strategies, particularly chemotherapy, can sensitize tumor tissue to the delivered radiation and increase efficacy.
Adverse effects:
Radiation can damage any intervening normal tissue.
Acute adverse effects depend on the area receiving radiation and may include:
- Lethargy
- Fatigue
- Mucositis
- Dermatologic manifestations (erythema, pruritus, desquamation)
- Esophagitis
- Pneumonitis
- Hepatitis
- GI symptoms (nausea, vomiting, diarrhea, tenesmus)
- GU symptoms (frequency, urgency, dysuria)
- Cytopenias
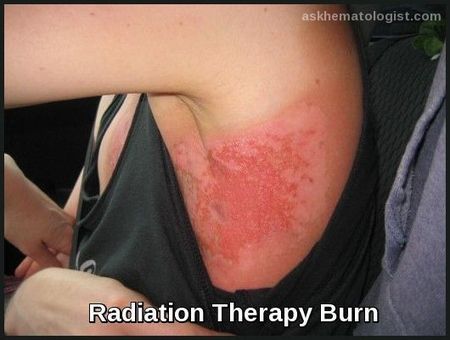
Erythematous skin burn following radiation therapy, demonstrating acute radiotherapy-induced dermatitis.
Early detection and management of these adverse effects is important not only for the patient’s comfort and quality of life but also to ensure continuous treatment; prolonged interruption can allow for tumor regrowth.
Late complications can include cataracts, keratitis, and retinal damage if the eye is in the treatment field. Additional late complications include hypopituitarism, xerostomia, hypothyroidism, pneumonitis, pericarditis, oesophagal stricture, hepatitis, ulcers, gastritis, nephritis, sterility, and muscular contractures. Radiation that reaches normal tissue can lead to poor healing of the tissues if further procedures or surgery is necessary. For example, radiation to the head and neck impairs recovery from dental procedures (eg, restoration, extraction) and thus should be administered only after all necessary dental work has been done.
Radiation therapy can increase the risk of developing other cancers, particularly leukemias, sarcomas in the radiation pathway, and carcinomas of the thyroid or breast. Peak incidence occurs 5 to 20 years after exposure and depends on the patient’s age at the time of treatment. For example, chest radiation therapy for Hodgkin lymphoma in adolescent girls leads to a higher risk of breast cancer than does the same treatment for postadolescent women.
Chemotherapy (for systemic disease):
Chemotherapy is the use of drugs or medications to treat disease, a method of cancer treatment. The medications enter the body and circulate, seeking to destroy cancer cells. The ideal chemotherapeutic drug would target and destroy only cancer cells. Only a few such drugs exist! Chemotherapy can be used alone or as part of a treatment plan that could include surgery, radiation therapy or biotherapy. There are several goals when using chemotherapy; cure, gaining control of cancer, and palliation (relief of symptoms). Some types of cancer respond best to chemotherapy medications, others react best with several treatment options combined.
The most common routes of administration are IV for cytotoxic drugs and oral for targeted drugs. Frequent dosing for extended periods may necessitate subcutaneously implanted venous access devices (central or peripheral), multilumen external catheters, or peripherally inserted central catheters.
Drug resistance can occur to chemotherapy. Identified mechanisms include overexpression of target genes, mutation of target genes, development of alternative pathways, drug inactivation by tumor cells, defective apoptosis in tumor cells, and loss of receptors for hormonal agents. For cytotoxic drugs, one of the best-characterized mechanisms is overexpression of the MDR-1 gene, a cell membrane transporter that causes efflux of certain drugs (eg, vinca alkaloids, taxanes, anthracyclines). Attempts to alter MDR-1 function and thus prevent drug resistance have been unsuccessful.
Cytotoxic drugs:
Traditional cytotoxic chemotherapy, which damages cell DNA, kills many normal cells in addition to cancer cells. Antimetabolites, such as 5-fluorouracil and methotrexate, are cell cycle–specific and have no linear dose-response relationship. In contrast, other chemotherapeutic drugs (eg, DNA cross-linkers, also known as alkylating agents) have a linear dose-response relationship, producing more tumor-killing as well as more toxicity at higher doses. At their highest doses, DNA cross-linkers may cause bone marrow aplasia, necessitating bone marrow/stem cell transplantation to restore bone marrow function.
Single-drug chemotherapy may cure selected cancers (eg, choriocarcinoma, hairy cell leukemia). More commonly, multidrug regimens incorporating drugs with different mechanisms of action and different toxicities are used to increase the tumor cell kill, reduce dose-related toxicity, and decrease the probability of drug resistance. These regimens can provide significant cure rates (eg, in acute leukemia, testicular cancer, Hodgkin lymphoma, non-Hodgkin lymphoma, and, less commonly, solid tumors such as small cell lung cancer and nasopharyngeal cancer). Multidrug regimens typically are given as repetitive cycles of a fixed combination of drugs. The interval between cycles should be the shortest one that allows for recovery of normal tissue. Continuous infusion may increase cell kill with some cell cycle–specific drugs (eg, 5-fluorouracil).
For each patient, the probability of significant toxicities should be weighed against the likelihood of benefit. The end-organ function should be assessed before chemotherapeutic drugs with organ-specific toxicities are used (eg, echocardiography before doxorubicin use). Dose modification or exclusion of certain drugs may be necessary in patients with chronic lung disease (eg, bleomycin), renal failure (eg, methotrexate), or hepatic dysfunction (eg, taxanes).
Despite these precautions, adverse effects commonly result from cytotoxic chemotherapy. The normal tissues most commonly affected are those with the highest intrinsic turnover rate: bone marrow, hair follicles, and the GI epithelium.
Imaging (eg, CT, MRI, PET) is frequently done after 2 to 3 cycles of therapy to evaluate response to treatment. Therapy continues if there is a clear response. If the tumor progresses despite therapy, the regimen is often amended or stopped. If the disease remains stable with treatment and the patient can tolerate therapy, then a decision to continue is reasonable with the understanding that the disease will eventually progress.
Understandably, you may be anxious about starting chemotherapy. Gaining a better understanding of how the mediations work, what they do to your body, and what changes they may cause can help you overcome some of your fears and anxiety.
Other important methods include:
Hormonal therapy:
Hormone therapy is a treatment that uses medicines to block or lower the amount of hormones in the body to slow down or stop the growth of cancer.
Hormones are natural substances made by glands in our bodies. They are carried around our body in our bloodstream and act as messengers between one part of our body and another.
Hormones are responsible for many functions in our body, including the growth and activity of certain cells and organs. The endocrine system is the network of glands that make hormones.
How does hormone therapy work?
Some cancers use hormones to grow or develop. This means the cancer is hormone sensitive or hormone dependent.
Hormone therapy for cancer uses medicines to block or lower the amount of hormones in the body to stop or slow down the growth of cancer.
Hormone therapy stops hormones being made or prevents hormones from making cancer cells grow and divide. It does not work for all cancers.
Cancers that can be hormone sensitive include:
breast cancer
prostate cancer
ovarian cancer
endometrial cancer
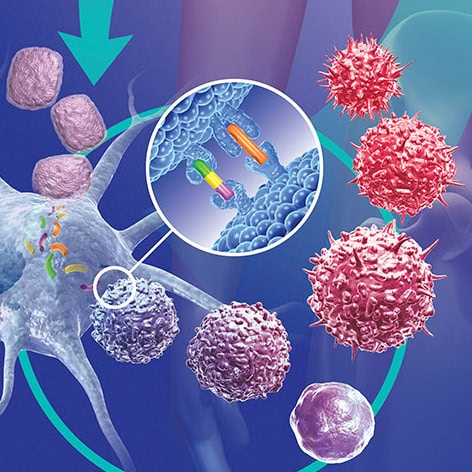
Immunotherapy:
Immunotherapy is a treatment that uses certain parts of a person’s immune system to fight diseases such as cancer. This can be done in a couple of ways:
* Stimulating your own immune system to work harder or smarter to attack cancer cells.
* Giving you immune system components, such as man-made immune system proteins.
Some types of immunotherapy are also sometimes called biologic therapy or biotherapy.
In the last few decades immunotherapy has become an important part of treating some types of cancer. Newer types of immune treatments are now being studied, and they’ll impact how we treat cancer in the future.
Immunotherapy includes treatments that work in different ways. Some boost the body’s immune system in a very general way. Others help train the immune system to attack cancer cells specifically.
Immunotherapy works better for some types of cancer than for others. It’s used by itself for some of these cancers, but for others it seems to work better when used with other types of treatment.
The main types of immunotherapy now being used to treat cancer include:
Monoclonal antibodies: These are man-made versions of immune system proteins. Antibodies can be very useful in treating cancer because they can be designed to attack a very specific part of a cancer cell.
Immune checkpoint inhibitors: These drugs basically take the ‘brakes’ off the immune system, which helps it recognise and attack cancer cells.
Cancer vaccines: Vaccines are substances put into the body to start an immune response against certain diseases. We usually think of them as being given to healthy people to help prevent infections. But some vaccines can help prevent or treat cancer.
Other, non-specific immunotherapies: These treatments boost the immune system in a general way, but this can still help the immune system attack cancer cells e.g. interferons, and other biologic response modifiers.
Differentiating drugs:
Retinoids are essential for the maintenance of epithelial differentiation. As such, they play a fundamental role in chemoprevention of epithelial carcinogenesis and in differentiation therapy.
Physiological retinoic acid is obtained through two oxidation steps from dietary retinol, i.e. retinol→retinal→retinoic acid. The latter retinal→retinoic acid step is irreversible and eventually marks disposal of this essential nutrient, through cytochrome P450-dependent oxidative steps.
Retinoids are effective chemopreventive agents against skin, head and neck, breast, liver and other forms of cancer.
Differentiation therapy of acute promyelocytic leukemia (APL) is based on the ability of retinoic acid to induce differentiation of leukemic promyelocytes.
Patients with relapsed, retinoid-resistant APL are now being treated with arsenic oxide, which results in apoptosis of the leukemic cells. Interestingly, induction of differentiation in promyelocytes and consequent remission of APL following retinoid therapy depends on expression of a chimeric PML–RARα fusion protein resulting from a t(15;17) chromosomal translocation.
The development of new, more effective and less toxic retinoids, alone or in combination with other drugs, may provide additional avenues for cancer chemoprevention and differentiation therapy.
Targeted Cancer Therapies:
Targeted cancer therapies are drugs designed to interfere with specific molecules necessary for tumor growth and progression.
Traditional cytotoxic chemotherapies usually kill rapidly dividing cells in the body by interfering with cell division. A primary goal of targeted therapies is to fight cancer cells with more precision and potentially fewer side effects.
Targeted cancer agents are broadly classified as either monoclonal antibodies or small molecules.
Therapeutic monoclonal antibodies target specific antigens found on the cell surface, such as transmembrane receptors or extracellular growth factors. Examples include bevacizumab, cetuximab and ipilimumab.
In some cases, monoclonal antibodies are conjugated to radio-isotopes or toxins to allow specific delivery of these cytotoxic agents to the intended cancer cell target.
Small molecules can penetrate the cell membrane to interact with targets inside a cell. Small molecules are usually designed to interfere with the enzymatic activity of the target protein. Examples include bortezomib, imatinib and seliciclib.
The overall cancer treatment should be coordinated among a radiation oncologist, surgeon, and medical oncologist, where appropriate. Choice of modalities constantly evolves, and numerous controlled research trials continue. When available and appropriate, clinical trial participation should be considered and discussed with patients.
Various terms are used to describe the response to treatment. The disease-free interval often serves as an indicator of cure and varies with cancer type. For example, lung, colon, bladder, large cell lymphomas, and testicular cancers are usually cured if a 5-yr disease-free interval occurs. However, breast and prostate cancers may recur long after 5 yr, an event defining tumor dormancy (now a major area of research); thus, a 10-yr disease-free interval is more indicative of cure.
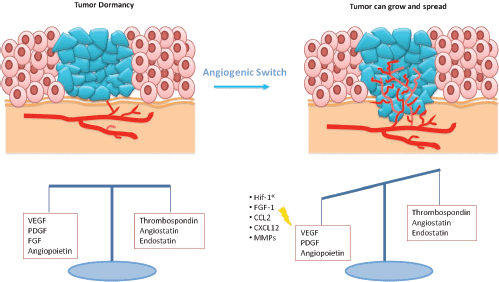
Illustration demonstrating tumor dormancy and how the angiogenic switch activates tumor vascularization, enabling cancer growth and spread.
Treatment decisions should weigh the likelihood of adverse effects against the likelihood of benefit; these decisions require frank communication and possibly the involvement of a multidisciplinary cancer team. Patient preferences for how to live out the end of life should be established early in the course of cancer treatment despite the difficulties of discussing death at such a sensitive time.
References:
Chabner B.A., MD; Thompson E.C., MD, MPH. Overview of Cancer Therapy – Hematology and Oncology. MSD Manual Professional Edition. Available online.
Sticca R., MD. Basics of Cancer Surgery. Cancer.Net. Navigating Cancer Care – Surgery Overview. Approved by the Cancer.Net Editorial Board, July 2016.
Regional Cancer Center (RCC). Introduction to Chemotherapy. Patient Education Resource.
De Vita V.T., Hellman S., Rosenberg S.A. Principles and Practice of Oncology (8th Edition). Lippincott, Williams & Wilkins, 2008.
American Cancer Society. What is Cancer Immunotherapy? Learn More.
Oxford Academic. Retinoids in Chemoprevention and Differentiation Therapy. Carcinogenesis. View Article.
Abramson R. Overview of Targeted Therapies for Cancer. My Cancer Genome. Updated July 14, 2017.
Stapleton S., Carrier C. Laparoscopic Colon Cancer Management May Be Beneficial in Elderly. 2 Minute Medicine. Read Article.
Morton G.C. The Emerging Role of High-Dose-Rate Brachytherapy for Prostate Cancer. ScienceDirect. Full Text.
Cheuck L. Brachytherapy (Radioactive Seed Implantation Therapy) in Prostate Cancer: Practice Essentials, Background, Indications. Medscape. Access Overview.
Watson M., et al. Principles of Cancer Treatment. Cancer Care, Oxford General Practice Library. Oxford University Press; 2010 (online edn 2011). DOI: 10.1093/med/9780199232031.003.0004.
AMBOSS. Principles of Cancer Care. Medical Knowledge Base.
Gelao L., Criscitiello C., Fumagalli L., Locatelli M., Manunta S., Esposito A., Minchella I., Goldhirsch A., Curigliano G. Tumour dormancy and clinical implications in breast cancer. ecancermedicalscience. 2013; 7: 320. doi:10.3332/ecancer.2013.320. Available at ecancer.org
Keywords:
principles of cancer therapy, cancer treatment principles, cancer treatment goals, cancer treatment types, treatment modalities, oncology principles, curative treatment, palliative care, adjuvant therapy, neoadjuvant therapy, chemotherapy, radiation therapy, cancer surgery, tumor staging, localized cancer, systemic cancer, multimodal therapy, cancer diagnosis

Request Online Consultation With Dr M Abdou
Fee: US$100
Secure payment via PayPal (credit and debit cards accepted)
Pay Now





That’s cool that your immune system could be used to fight cancer. I feel like it would be nice to not have surgery or anything. Hopefully, that would help people get through it more easily.
Hi Tyler,
Thank you for your comment.
Our immune system does attack cancer cells. It’s recognising and destroying little cancers as they develop all the time. If we didn’t have an immune system, then we would be developing cancer a lot more often. I suggest reading this interesting article too:
Science Surgery: ‘Why doesn’t the immune system attack cancer cells?’
Regards,