PET imaging in Hematology
A PET scan machine used in hematology to assess cancer spread, treatment response, and metabolic activity.
Positron emission tomography (PET) imaging in hematology is a key diagnostic modality that provides detailed three-dimensional (3D) functional imaging of metabolic activity within the body. It has become a cornerstone in the evaluation of hematological malignancies, particularly for staging, treatment response assessment, and detection of disease recurrence.
PET is a nuclear medicine imaging technique that uses small amounts of radioactive tracers to identify differences between normal and diseased tissues. The most commonly used tracer is fluorodeoxyglucose (FDG), making FDG-PET the standard modality in clinical practice. Following intravenous administration, FDG accumulates in metabolically active cells, allowing visualization of areas with increased glucose uptake.
Because malignant cells typically exhibit higher metabolic activity than normal tissues, they preferentially uptake FDG. The PET scanner detects the emitted radiation and generates colour-coded images that reflect tissue metabolic activity, enabling differentiation between benign and malignant processes.
Unlike conventional imaging, which primarily depicts anatomical structures, PET provides functional information about tissue activity. This allows early detection of disease and assessment of treatment response before structural changes become apparent.
In clinical practice, PET is most commonly combined with computed tomography (CT) to form PET-CT, which integrates metabolic and anatomical data, significantly improving diagnostic accuracy compared to PET alone.
Modern PET systems achieve a spatial resolution of approximately 5 mm and allow semi-quantitative assessment using the standardized uptake value (SUV), which reflects tracer uptake relative to the injected dose and body parameters. Although an SUV threshold of approximately 2.5 has historically been associated with malignancy, there is considerable overlap between benign and malignant conditions. Increased uptake may also be seen in inflammatory or infectious processes, while some low-grade malignancies may demonstrate low FDG avidity. Therefore, SUV values should always be interpreted in the appropriate clinical context.
PET-CT scans can help to:
- Diagnose cancer
- Stage cancer, enabling accurate assessment of disease extent and dissemination
- Determine suitability for surgical intervention
- Guide selection of the most appropriate treatment strategy
- Identify the site of the primary tumour
- Detect disease recurrence
A PET-CT scan also plays a crucial role in assessing treatment response, allowing clinicians to evaluate metabolic changes before anatomical changes become apparent.
Importantly, PET imaging is increasingly utilised to guide response-adapted therapy, particularly in Hodgkin lymphoma, where early interim PET (iPET) enables treatment stratification through escalation or de-escalation of chemotherapy. Beyond its role in therapeutic decision-making, iPET provides an early assessment of treatment response and carries significant prognostic value, especially in Hodgkin lymphoma and aggressive non-Hodgkin lymphomas. This strategy contributes to optimising clinical outcomes while reducing unnecessary treatment-related toxicity.
PET-CT is incorporated into international guidelines for lymphoma management, including those from the European Society for Medical Oncology and National Comprehensive Cancer Network.
The clinical utility of PET-CT varies across different hematological malignancies:
- Hodgkin lymphoma: PET-CT is the imaging modality of choice for staging and response assessment and is considered the gold standard in modern management.
- Diffuse large B-cell lymphoma: Provides important prognostic information and is widely used for response evaluation.
- Multiple myeloma: Particularly useful for detecting extramedullary disease and assessing metabolically active lesions.
- Leukemia: Has a limited role, but may be useful in selected scenarios, such as suspected extramedullary involvement.
PET scans may also occasionally be combined with MRI (PET-MRI), offering additional advantages in selected clinical scenarios, particularly where superior soft tissue contrast is required.
PET imaging is increasingly used to assess response following chimeric antigen receptor T-cell (CAR-T) therapy, helping to differentiate between residual disease and treatment-related inflammatory changes.
Emerging PET tracers beyond FDG, such as PSMA and CXCR4-targeted imaging, are under investigation in selected hematological malignancies and may expand diagnostic capabilities in the future.
Limitations and Pitfalls of PET Imaging:
- False-positive findings:
FDG uptake is not tumour-specific and may be seen in a variety of benign conditions, including infection, inflammation, and post-treatment changes (e.g., following granulocyte colony-stimulating factor administration), potentially leading to overestimation of disease activity. - False-negative findings:
Certain malignancies, particularly indolent or low-grade lymphomas (e.g., small lymphocytic lymphoma), may demonstrate low FDG avidity, resulting in reduced sensitivity of PET imaging. - Physiological FDG uptake:
Normal physiological uptake in organs such as the brain, myocardium, and urinary tract can obscure or mimic pathological findings, requiring careful interpretation in the appropriate clinical context.
How PET scans work?
PET scanners work by detecting the radiation given off by a substance injected into the arm called a radiotracer as it collects in different parts of the body.
In most PET scans, a radiotracer called fluorodeoxyglucose (FDG) is used, which is similar to naturally occurring glucose, so the body treats it in a similar way.

PET scan setup illustrating the scanner and movable couch used for cancer imaging and staging in hematology.
By analyzing the areas where the radiotracer does and doesn’t build up, it’s possible to determine how well certain body functions work and identify any abnormalities. For example, a concentration of FDG in the body’s tissues can help identify cancerous cells because cancer cells use glucose at a much faster rate than normal cells.

Pre- and post-chemotherapy PET scans demonstrating complete metabolic response in non-Hodgkin lymphoma with resolution of lymphadenopathy and splenic uptake.
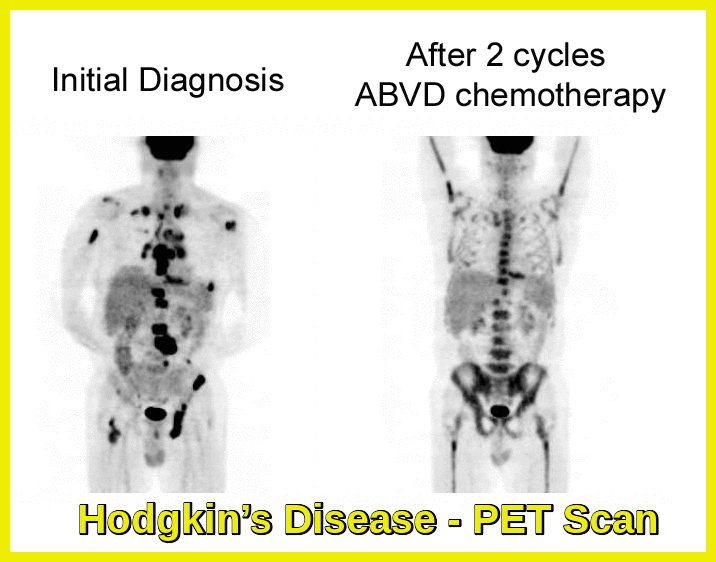
The adoption of the PET scan has been most notable in treating lymphomas, where its incorporation into the care of Hodgkin’s and non-Hodgkin’s lymphomas is now virtually universal. PET scans have clearly impacted the way lymphomas are staged and treated, particularly those types with the potential for cure, such as classical Hodgkin’s lymphoma and large cell and/or aggressive lymphoma.

Baseline and interim PET scans demonstrating treatment response after two cycles of ABVD chemotherapy in Hodgkin lymphoma.
PET scans, because of their enhanced sensitivity and convenience, have almost totally replaced gallium scans. Unlike gallium scans, PET scans are routinely used in staging since the scans can reveal disease in normal-sized lymph nodes (on CT scan), which would otherwise have been interpreted as unremarkable.
PET scanning has altered treatment approaches as well. For instance, bulky masses, despite being sterilized of lymphoma, are often characterized by some residual mass or scar. Using conventional means, it has been difficult to determine whether the residual mass after treatment did or did not contain active disease. As a result, these masses had been routinely irradiated even if a biopsy showed a scar (because of possible sampling error). Although many still advocate irradiating bulky masses, the advent of the PET scan has engendered a rethinking of this approach when no disease activity is apparent.
PET Scans in the Staging of Lymphoma:
Staging is important in treating all malignancies but is critically important for patients with lymphoma. Accurate staging allows the minimization of toxic therapies, such as extended-field radiation or overly aggressive chemotherapy, decreasing the risk of secondary malignancies, which exceeds 10% in several historical series of patients with early-stage Hodgkin’s disease. Patient quality of life during and after treatment may also be improved with tailored therapy defined by staging.
Most PET studies involve patients with either Hodgkin’s disease or diffuse large B-cell non-Hodgkin’s lymphoma. PET detects more disease sites above and below the diaphragm on lymphoma staging than gallium scintigraphy and may have particular utility in evaluating the spleen. Perhaps more importantly, PET may help to characterize a residual mass on anatomic imaging following therapy as either fibrosis or residual active lymphoma. Moreover, persistently positive PET scans during and after chemotherapy appear to have a high sensitivity for predicting subsequent relapse. A negative PET scan at the end of therapy provides very favorable prognostic information.
Lymphoma Histology and FDG Uptake:
Most studies evaluating FDG-PET in lymphoma include patients with diffuse large B-cell non-Hodgkin’s lymphoma (NHL) or Hodgkin’s disease. There is limited data available on the role of PET in other histologies. A retrospective review of 172 patients with various types of lymphoma who underwent FDG-PET imaging was completed at the University of Pennsylvania. Only 6% of those patients had no evidence of disease on PET scans. FDG-PET accurately detected disease in patients with diffuse large B-cell NHL, mantle cell lymphoma, follicular lymphoma, and Hodgkin’s disease. PET was less reliable at detecting marginal zone lymphoma, a finding that has been confirmed by other groups, particularly in the case of extranodal marginal zone lymphomas.
In another study, limited to patients with indolent B-cell lymphomas, PET appeared to have the potential to contribute to managing patients with follicular NHL. Still, sites of disease in patients with small lymphocytic lymphomas were only detected approximately 50% of the time. Small series have suggested that using SUV to determine the intensity of FDG uptake, PET may be able to predict the histologic transformation of indolent lymphoma; however, the relationship among pathological subtype, mitotic rate, and SUV remains controversial.
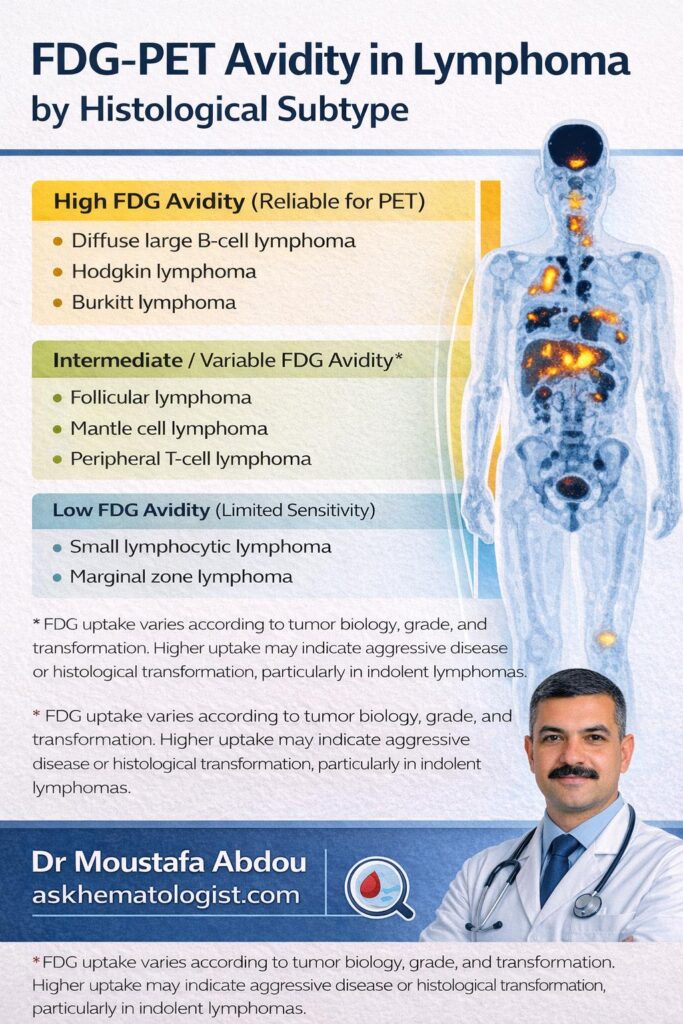
FDG-PET avidity patterns across lymphoma subtypes highlighting high, intermediate, and low uptake for clinical interpretation.
PET CT Scan in Myeloma:
18F-FDG PET/CT is commonly used to detect myeloma bone disease and extra-medullary sites of metabolically active myeloma foci with relatively high sensitivity and specificity. Furthermore, follow-up PET/CT scans can be used to monitor treatment response and are of prognostic value for survival. This imaging modality uses enhanced glucose metabolic activity to visualize areas of interest and detect more lesions than whole-body X-rays. A decrease in 18F-FDG uptake correlates with the chemotherapeutic response, and residual 18F-FDG uptake after completion of therapy may serve as an accurate marker for assessing minimal residual disease (MRD) comparable to next-generation sequencing or multi-colour flow cytometry. According to the International Myeloma Working Group (IMWG) response criteria, developing new bony lesions or soft tissue plasmacytoma on imaging modalities, including PET/CT, indicates progressive disease (PD) independent of serum monoclonal protein or light chain concentration.

The image depicts a myeloma pretreatment PET scan showing widespread axial skeletal disease and diffuse marrow 18F-FDG uptake. Post-treatment PET scan shows the response with therapy in the axial skeleton and bone marrow.
Deauville Criteria:
The Deauville 5-point scoring system is an internationally accepted method for interpreting FDG-PET findings in both Hodgkin lymphoma and non-Hodgkin lymphoma. It provides a standardized approach by comparing lesion uptake to physiological reference regions (mediastinum and liver), and is incorporated into modern response criteria such as the Lugano Classification.
Scoring system:
- Score 1: No residual uptake above background
- Score 2: Uptake ≤ mediastinal blood pool
- Score 3: Uptake > mediastinum but ≤ liver
- Score 4: Uptake moderately greater than liver
- Score 5: Markedly increased uptake compared to liver and/or new lesions
- Score X: New areas of uptake unlikely to be related to lymphoma (e.g., infection or inflammation)
Clinical interpretation:
- Scores 1–3 are generally considered consistent with complete metabolic response (CMR), particularly in Hodgkin lymphoma
- Scores 4–5 suggest residual active disease, although interpretation should always consider clinical context and timing of imaging
- Score 3 represents an intermediate category and, in many patients with Hodgkin lymphoma, is associated with a favourable prognosis when treated with standard therapy
Preparation:
Your doctor will give you detailed instructions on preparing for your scan. A general rule is to not eat anything for at least 6 hours before the scan. You will be encouraged to drink water. Wear comfortable clothes.
During the Examination:
A nurse or technologist will take you to a special room where you will receive an intravenous (IV) injection of the radioactive drug. Sometimes, you will be asked to inhale the drug instead. Then, you will wait 30 to 90 minutes for the drug to travel through your body and accumulate in the tissues being studied. During this time, you will rest quietly and avoid movement. You won’t be able to feel the drug in your body.
The PET scanner is a large machine with a hole in the middle. It looks like a doughnut with a table in the middle. You will lie on the table. The table will slide into the machine. You will be asked to remain still during the scan.
Time Required:
30 to 45 minutes.
Noise During Examination:
Buzzing or clicking sounds.
Space During Examination:
You will lie on a narrow table that slides into a circular opening of the scanner. The size of the opening is 27 to 30 inches. How much space you feel you have around you will depend on your body size and the scanner used. A mild sedative may be used to help you feel more comfortable during the exam. Let your doctor know if you are anxious about being in enclosed spaces.
A patient undergoing a PET-CT scan with a healthcare professional, demonstrating the clinical setup used for cancer diagnosis, staging, and treatment response assessment.
Benefits:
The functional information obtained by a PET scan is unique and unavailable using other types of imaging. For many diseases, PET provides the most useful information required to diagnose and determine the most appropriate treatment.
Risks:
Although a radioactive tracer is used during a PET scan, the amount of radiation exposure is generally low and short-lived, and is not sufficient to affect normal body processes. However, PET-CT does expose patients to moderate levels of ionising radiation due to the combined PET and CT components; therefore, its use should always be clinically justified, particularly in younger patients and in situations requiring repeated imaging.
Potential risks associated with the tracer include:
- Pregnancy and breastfeeding:
The radioactive tracer may expose a fetus during pregnancy or an infant during breastfeeding. Patients should inform their doctor if they are pregnant or nursing before undergoing the scan. - Allergic reactions:
There is a rare risk of hypersensitivity or allergic reaction to the tracer.
Cost of PET Scan:
The cost of a PET scan can vary significantly depending on several factors, including the geographic location, the type of PET scanner used, the complexity of the procedure, and the healthcare provider. Here is a general overview of the cost of PET scans worldwide:
- The average cost of a PET scan ranges from $1,500 to $3,000 in the United States.
- In the United Kingdom, the cost of a PET scan typically falls between £1,000 and £2,000.
- In Canada, the cost of a PET scan is usually covered by provincial health insurance plans.
- In Australia, the cost of a PET scan can range from $1,000 to $2,500.
- In many developing countries, the cost of a PET scan can be significantly higher due to limited access to advanced medical technology and infrastructure.
It’s important to note that these are just approximate ranges, and the actual cost may vary depending on individual circumstances and specific healthcare providers. If you are considering undergoing a PET scan, it is advisable to consult with your healthcare provider or insurance company to obtain accurate information about the cost and coverage options.
Summary:
Positron emission tomography (PET), most commonly performed as PET-CT, is a cornerstone imaging modality in modern hematology and oncology. By using fluorodeoxyglucose (FDG) to detect metabolic activity, PET imaging enables accurate diagnosis, staging, and treatment response assessment in hematological malignancies such as Hodgkin lymphoma, Diffuse large B-cell lymphoma, and Multiple myeloma.
Unlike conventional imaging, PET provides functional insights into disease biology, allowing early detection of therapeutic response before structural changes occur. It plays a central role in response-adapted therapy, particularly through interim PET (iPET), and is incorporated into international guidelines from organizations such as European Society for Medical Oncology and National Comprehensive Cancer Network. The Deauville scoring system further standardizes PET interpretation in lymphoma.
While highly valuable, PET imaging has limitations, including false positives from inflammation or infection, and reduced sensitivity in low-grade lymphomas. Advances such as PET-MRI and novel tracers continue to expand its clinical applications, including emerging roles in CAR-T therapy assessment.
Frequently Asked Questions About PET Imaging in Hematology:
What is a PET scan and how does it work?
A PET scan is a nuclear medicine imaging technique that uses a radioactive tracer, most commonly FDG, to detect metabolic activity in tissues. Cancer cells typically show increased glucose uptake, allowing PET imaging to identify areas of disease.
What is the role of PET-CT in hematology?
PET-CT is widely used in hematology for diagnosing, staging, assessing treatment response, and detecting relapse in blood cancers, particularly lymphomas and selected cases of myeloma.
Which hematological malignancies are best assessed by PET imaging?
PET imaging is most reliable in FDG-avid diseases such as Hodgkin lymphoma, Diffuse large B-cell lymphoma, and Burkitt lymphoma, while indolent lymphomas show more variable uptake.
What is the Deauville score in PET imaging?
The Deauville score is a 5-point scale used to assess treatment response in lymphoma by comparing FDG uptake in lesions to the mediastinum and liver. It is part of the Lugano Classification.
What is interim PET (iPET) and why is it important?
Interim PET is performed during treatment, usually after 2–3 cycles of chemotherapy, to assess early response. It has prognostic significance and is used to guide response-adapted therapy, especially in Hodgkin lymphoma.
What are the limitations of PET imaging?
PET imaging may produce false-positive results due to infection or inflammation and false-negative results in low-grade lymphomas. Physiological uptake in organs such as the brain and urinary tract can also affect interpretation.
What is SUV in PET scans?
Standardized Uptake Value (SUV) is a semi-quantitative measure of FDG uptake in tissues. While higher SUV values may suggest malignancy, interpretation must always consider clinical context.
Is PET imaging used in multiple myeloma?
Yes, PET imaging is increasingly used in Multiple myeloma to detect active disease, assess treatment response, and identify extramedullary involvement.
What is the role of PET imaging in CAR-T therapy?
PET imaging is emerging as an important tool for assessing response following CAR-T therapy, helping distinguish between residual disease and treatment-related inflammatory changes.
Is PET-CT safe?
PET-CT involves exposure to moderate ionising radiation, but it is generally safe when clinically justified. Special considerations apply to pregnant or breastfeeding patients.
References:
1. Thompson CJ. Instrumentation. In: Wahl RL, editor. Principles and Practice of Positron Emission Tomography. Philadelphia: Lippincott Williams & Wilkins; 2002. p. 48–64.
2. Coleman M. The use of PET scanning in lymphoma: progress and problems. HemOnc Today. 2009 Sep 25. Available from: https://goo.gl/YFdVhK
3. Friedberg JW. PET scans in the staging of lymphoma: current status. The Oncologist. 2003;8(5):438–447. Available from: https://theoncologist.onlinelibrary.wiley.com/doi/full/10.1634/theoncologist.8-5-438
4. Ng AK, Bernardo MV, Weller E, et al. Second malignancy after Hodgkin disease treated with radiation therapy with or without chemotherapy: long-term risks and risk factors. Blood. 2002;100(6):1989–1996. doi:10.1182/blood.V100.6.1989
5. Elstrom R, Guan L, Baker G, et al. Utility of FDG-PET scanning in lymphoma by WHO classification. Blood. 2003;101(10):3875–3876. doi:10.1182/blood-2002-11-3546
6. Hoffmann M, Kletter K, Diemling M, et al. FDG-PET does not reliably visualize extranodal B-cell lymphoma of MALT type. Ann Oncol. 1999;10(10):1185–1189. doi:10.1023/A:1008394900859
7. Jerusalem G, Beguin Y, Najjar F, et al. FDG-PET in staging of low-grade non-Hodgkin lymphoma. Ann Oncol. 2001;12(6):825–830. doi:10.1023/A:1011197427507
8. Barrington SF, et al. Role of imaging in staging and response assessment of lymphoma: consensus of the International Conference on Malignant Lymphomas Imaging Working Group. Eur J Nucl Med Mol Imaging. 2010;37(9):1824–1833. doi:10.1007/s00259-010-1428-0
9. Cheson BD, Fisher RI, Barrington SF, et al. Recommendations for initial evaluation, staging, and response assessment of Hodgkin and non-Hodgkin lymphoma: the Lugano classification. J Clin Oncol. 2014;32(27):3059–3068. doi:10.1200/JCO.2013.54.8800
10. National Comprehensive Cancer Network (NCCN). NCCN Clinical Practice Guidelines in Oncology: Hodgkin Lymphoma & B-Cell Lymphomas. Available from: https://www.nccn.org
11. European Society for Medical Oncology (ESMO). ESMO Clinical Practice Guidelines for lymphoma. Available from: https://www.esmo.org/guidelines
12. Follows GA, et al. Guidelines for the first-line management of classical Hodgkin lymphoma. Br J Haematol. 2014;166(1):34–49. doi:10.1111/bjh.12878
13. Mah K, Caldwell CB. PET-CT in radiotherapy treatment planning. In: Standardized Uptake Value – ScienceDirect Topic Overview. Elsevier; 2008. Available from: https://www.sciencedirect.com/topics/medicine-and-dentistry/standardized-uptake-value
14. Sundaram S, Driscoll J, Fernandez-Ulloa M, de Lima M, Malek E. FDG-PET imaging in multiple myeloma: implications for response assessment. Am J Nucl Med Mol Imaging. 2018;8(6):421–427. PMID: 30697462
15. Durie BG, Waxman AD, D’Agnolo A, Williams CM. Whole-body FDG-PET identifies high-risk myeloma. J Nucl Med. 2002;43(11):1457–1463. Available from: https://jnm.snmjournals.org/content/43/11/1457
16. Bartel TB, Haessler J, Brown TL, et al. F18-FDG PET in multiple myeloma prognosis. Blood. 2009;114(10):2068–2076. doi:10.1182/blood-2009-03-213280
17. Kumar S, Paiva B, Anderson KC, et al. International Myeloma Working Group consensus criteria for response and MRD assessment. Lancet Oncol. 2016;17(8):e328–e346. doi:10.1016/S1470-2045(16)30206-6
Keywords:
PET CT scan, PET imaging in hematology, FDG PET scan, PET scan for cancer diagnosis, PET CT meaning, PET scan meaning, PET CT vs CT scan, difference between PET scan and CT scan, PET scan for lymphoma staging, PET CT in lymphoma, FDG PET in lymphoma, Deauville score PET, PET scan Hodgkin lymphoma, PET scan non-Hodgkin lymphoma, PET CT response assessment lymphoma, interim PET iPET lymphoma, PET scan in multiple myeloma, FDG PET in multiple myeloma, PET CT myeloma imaging, PET scan bone marrow disease, standardized uptake value SUV PET, FDG uptake cancer imaging, PET CT interpretation guidelines, PET CT scan procedure, whole body PET CT scan, PET scan for cancer patients, PET scan results explained, PET CT scan cost, PET imaging cost, PET scan safety radiation, PET CT staging cancer, PET CT restaging lymphoma, PET CT treatment response, PET MRI scan, PET CT vs MRI, functional imaging in hematology, PET scan in blood cancer, PET CT radiotherapy planning, PET imaging clinical applications, PET CT guidelines NCCN ESMO, PET scan CAR-T therapy assessment, PET CT oncology imaging, PET scan metabolic imaging cancer


Request Online Consultation With Dr M Abdou
Fee: US$100
Secure payment via PayPal (credit and debit cards accepted)
Pay Now

I appreciate you helping me learn more how a PET scan works. It is interesting that it can detect the radiation given off by the injected liquid in the body. My nephew is thinking of becoming a scanning technician. He might interested in knowing this.
Hi Elisabeth,
Thank you for your comment.
The PET scan uses a special dye containing radioactive tracers. These tracers are either swallowed, inhaled, or injected into a vein in your arm depending on what part of the body is being examined. Certain organs and tissues then absorb the tracer. When detected by a PET scanner, the tracers help your doctor to see how well your organs and tissues are working. The tracer will collect in areas of higher chemical activity, which is helpful because certain tissues of the body, and certain diseases, have a higher level of chemical activity. These areas of disease will show up as bright spots on the PET scan. The PET scan can measure blood flow, oxygen use, how your body uses sugar, and much more.
Regards,
Dr. Abdou,
My mother is 96 and many test have been done to figure out what is wrong with her lung. Everything has been ruled out except cancer, but after 3 Cscans they can’t make a diagnosis. Would you reccomend a PET scan? They are talking about taking fluid from the outside of her lung to test it for cancer cells. She doesn’t want the procedure where they take specimens from inside the lungs. The pulmonary doctor seems to think because she is 96 just let it fo and die. Would like to hear what you think. Thank you.
Nancy
Hi Nancy,
Thank you for your message.
The PET scan uses a mildly radioactive drug to show up areas of the body where cells are more active than normal. It’s used to help diagnose some conditions including cancer. It can also help to find out where and whether cancer has spread. I encourage doing a PET-CT scan and a CT guided biopsy for your mother to reach a definite diagnosis if she is a good 96 i.e. without multiple comorbidities and has a reasonable performance status (0-2) as long as she can go for the test as some cancers may respond well to treatment like lymphomas for example.
Best wishes,
I have a lot of small dots of blood and medium and large bruises for no reason when I do hit myself instantly I bruise
I am very tired no energy.. no blood thinners. Primary M.D. just tells me I have low red blood cells. Should I request to see a hematologist
?
Hi Caroline,
Thank you for your message.
I would suggest initially to check your FBC, Coagulation Screen, Serum iron/ferritin, B12, and Folate.
Best wishes,
Good evening doctor
My 4 years old son has been diagnosed with NHL (Burkitt) with multiple hepatic liver masses but his bone marrow is negetive now he is reciveing the third cycle (mtx , cytosar) , he completed 2 induction cycle(cyclophosmide , mtx , adrimycin ,vencstrin and rituxmab )and i have some question
1-does liver involved in burkitt lymphoma?
2-is it stage 3 or 4 ?
3-how is the prognoses of burkitt lymphoma with this state ?
FYI he is feeling better now
with recpect
Hi Adnan,
Thank you for your message and wishing your son a speedy recovery Insha Allah.
1- Any organ can get involved.
2- I’ll classify it as stage 4 even if the bone marrow is spared.
3- Prognosis is good and he may achieve a complete cure with treatment.
BW,