Tumor Lysis Syndrome

Tumor Lysis Syndrome illustrating rapid destruction of malignant cells with release of potassium, phosphate, and uric acid into the bloodstream.
Tumor Lysis Syndrome (TLS) is a severe and potentially life-threatening oncologic emergency that occurs during or shortly after cancer treatment. It results from the rapid destruction of malignant cells, leading to the release of intracellular contents such as potassium, phosphate, and uric acid into the bloodstream. Tumor Lysis Syndrome is most commonly seen in hematologic malignancies, including leukemia and lymphoma, but may also occur in patients with bulky solid tumors. This condition can lead to serious complications such as acute kidney injury, cardiac arrhythmias, and metabolic disturbances if not promptly recognized and managed. This article reviews the causes, symptoms, diagnosis, prevention, and management of Tumor Lysis Syndrome. The ICD-10 code for Tumor Lysis Syndrome is E88.3.
TLS is typically triggered by the initiation of cancer therapy, either through chemotherapy or radiation. Rapid tumor cell destruction releases large amounts of cellular components, including nucleic acids, potassium, phosphate, and uric acid, into the bloodstream. The body’s standard mechanisms for processing and excreting these substances can be overwhelmed, leading to metabolic imbalances.
Tumor lysis syndrome (TLS) is one of the most common disease-related oncologic emergencies encountered by physicians managing patients with hematologic malignancies. Although it occurs most frequently in individuals with non-Hodgkin lymphoma or acute leukemia, its incidence is increasing among patients with solid tumors that were previously considered low risk for this complication. TLS results from the rapid destruction of malignant cells, leading to the release of intracellular contents into the bloodstream. When this process overwhelms renal excretory capacity, it results in metabolic disturbances that can cause serious complications.
Clinically, the syndrome is characterized by rapid development of hyperuricemia, hyperkalemia, hyperphosphatemia, hypocalcemia, and acute kidney injury.
Tumor lysis syndrome arises most commonly after the start of initial chemotherapeutic treatment, but spontaneous cases have increasingly been documented in patients with high-grade hematologic malignancies. It can occur within a few hours of treatment, but it is most often seen 48–72 hours (2–3 days) after treatment starts.
TLS is not limited to systemic chemotherapy, which travels throughout the body to destroy cancer cells. It can also occur with intrathecal chemotherapy, which is given directly into the fluid-filled space around the brain and spinal cord. TLS can develop after chemoembolization, which is a procedure that stops blood flow to a tumor and delivers chemotherapy directly to the tumor.
Tumor lysis syndrome (TLS) has also been associated with other treatment modalities, including radiation therapy, corticosteroids, hormonal therapy, and biological agents.
Although tumor lysis syndrome has been reported across a wide range of tumor types, it is most commonly associated with bulky, rapidly proliferating, and treatment-responsive malignancies, particularly acute leukemias and high-grade non-Hodgkin lymphomas such as Burkitt lymphoma. It has also been described in other hematologic malignancies and in solid tumors, including hepatoblastoma and stage IV neuroblastoma.
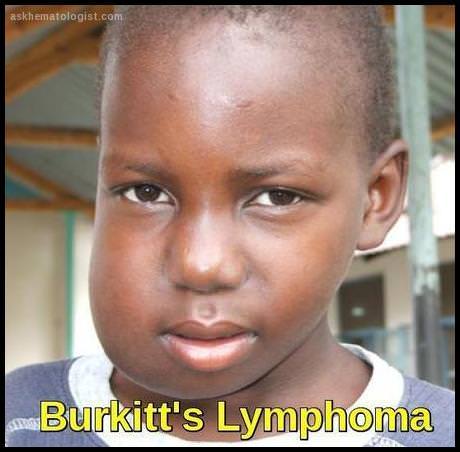
Burkitt lymphoma presenting with characteristic jaw involvement in a pediatric patient, a classic feature of the endemic form.
Because tumor lysis syndrome is potentially life-threatening, the main principles of management include the identification of high-risk patients, initiation of preventive therapy, early recognition of metabolic and renal complications, and prompt administration of supportive care, including hemodialysis.
Pathophysiology:
Tumor lysis syndrome (TLS) occurs when the release of intracellular contents, including potassium, phosphate, nucleic acids, and cytokines, during rapid tumor cell lysis exceeds the body’s capacity to maintain metabolic homeostasis.
Renal excretion is the primary route for eliminating uric acid, xanthine, and phosphate. These solutes may precipitate within the renal tubules and collecting system, contributing to acute kidney injury. Although the kidneys can normally excrete these metabolites efficiently, clinical tumor lysis syndrome is more likely to develop when renal function is impaired or when the metabolic load overwhelms excretory capacity.
Crystal-induced tissue injury in tumor lysis syndrome occurs when calcium phosphate, uric acid, and xanthine precipitate within the renal tubules, leading to inflammation and tubular obstruction. Crystal formation is promoted by a high solute load, reduced solubility, low urine flow, and the presence of co-crystallizing substances, all of which contribute to disease severity.
Patients with elevated levels of both uric acid and phosphate are at particularly high risk of crystal-associated acute kidney injury. Uric acid precipitation is facilitated by the presence of calcium phosphate, and conversely, calcium phosphate deposition is enhanced in the presence of uric acid. Urine pH also plays an important role: higher pH increases uric acid solubility but reduces the solubility of calcium phosphate, thereby influencing the predominant type of crystal formation.
In patients treated with Allopurinol, accumulation of xanthine, a poorly soluble precursor of uric acid, may occur and can lead to xanthine nephropathy or urolithiasis.
Calcium phosphate deposition is not limited to the kidneys and may occur in multiple tissues. The risk of ectopic calcification is increased in patients receiving intravenous calcium. When deposition occurs within the cardiac conduction system, it may result in serious and potentially fatal arrhythmias.
Epidemiology:
The incidence and severity of tumor lysis syndrome (TLS) are influenced by tumor burden, the intrinsic susceptibility of malignant cells to lysis, patient-related factors, and the quality of supportive care. A larger tumor burden is associated with a greater release of intracellular contents following effective anticancer therapy.
Malignancies with a high risk of TLS include high-grade lymphomas, acute leukemias, and other rapidly proliferating tumors. However, TLS is increasingly recognized in cancers that were previously considered low risk, including endometrial cancer, hepatocellular carcinoma, chronic lymphocytic leukemia, and chronic myeloid leukemia.
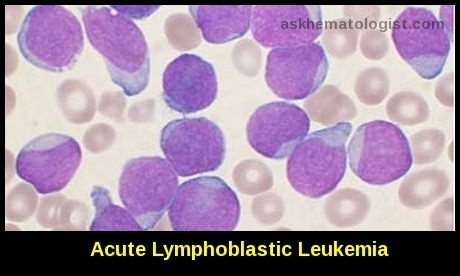
Acute lymphoblastic leukemia demonstrating circulating lymphoblasts on peripheral blood smear, a key diagnostic feature.
Patient-related factors that increase the risk of tumor lysis syndrome (TLS) include pre-existing chronic kidney disease, oliguria, dehydration, hypotension, and acidic urine.
The adequacy of supportive care, particularly fluid management, plays a critical role in both the development and severity of TLS. Severe cases have been reported in patients with non-hematologic malignancies who received effective anticancer therapy without appropriate prophylaxis, including intravenous hydration and close monitoring, due to failure to anticipate TLS.
In contrast, patients with high-risk malignancies such as bulky Burkitt lymphoma often have a lower incidence of clinical TLS in settings where proactive management is standard practice. Routine use of aggressive hydration and Rasburicase significantly reduces the risk of complications. In pediatric Burkitt lymphoma, treatment with rasburicase has been associated with a substantially lower need for dialysis compared with Allopurinol, highlighting the impact of optimal supportive care even in patients with otherwise similar risk profiles.
Clinical picture:
History:
In tumor lysis syndrome, a constellation of clinical signs and symptoms may develop prior to the initiation of chemotherapy or, more commonly, within 72 hours after administration of cytotoxic therapy.
Inquiries should be made with regard to the following:
- Time of onset of symptoms of malignancy.
- Presence of abdominal pain and distension.
- Presence of urinary symptoms – Such as oliguria, flank pain, and hematuria.
- Symptoms of hyperkalemia – Such as weakness and paralysis.
- Occurrence of any symptoms of hypocalcemia – Such as anorexia, vomiting, cramps, seizures, spasms, altered mental status, and tetany.
Other manifestations of tumor lysis syndrome include the following:
- Lethargy
- Edema
- Fluid overload
- Congestive heart failure
- Cardiac dysrhythmias
- Syncope
- Sudden death
Physical Examination:
Symptoms reflect the severity of underlying metabolic abnormalities.
Hyperkalemia can cause paresthesia, weakness, and fatal cardiac arrhythmias.
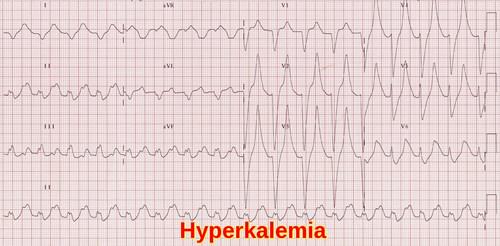
Hyperkalemia ECG demonstrating classic tall peaked T waves, an early and important sign of elevated serum potassium.
Severe hypocalcemia can lead to the following signs and symptoms:
- Paresthesia and tetany with positive Chvostek and Trousseau signs
- Anxiety
- Carpal and pedal spasms
- Bronchospasm
- Seizures
- Cardiac arrest
Deposition of calcium phosphate in various tissues may be responsible for the following signs and symptoms:
- Pruritus
- Gangrenous changes of the skin
- Iritis
- Arthritis
Uremia can produce the following signs and symptoms:
- Fatigue
- Weakness
- Malaise
- Nausea
- Vomiting
- Anorexia
- Metallic taste
- Hiccups
- Neuromuscular irritability
- Difficulty concentrating
- Pruritus
- Restless legs
- Ecchymoses
As uremia progresses, paresthesia and evidence of pericarditis may develop, as well as signs of drug toxicity from medications eliminated by the kidney. Features of volume overload, such as dyspnea, pulmonary rales, edema, and hypertension, may develop.
Elevated uric acid levels may produce lethargy, nausea, and vomiting. Rapidly increasing uric acid levels may lead to arthralgia and renal colic.
Management:
Prophylaxis of Tumour Lysis Syndrome (TLS):
All patients with hematologic malignancies scheduled to receive chemotherapy should undergo formal risk assessment for tumor lysis syndrome (TLS) prior to treatment initiation.
Low-risk patients can be managed with close monitoring of fluid balance and serial laboratory parameters, with a low threshold for initiating intravenous hydration and considering Allopurinol if indicated.
Intermediate-risk patients should receive prophylaxis with allopurinol for up to 7 days, along with enhanced hydration starting at treatment initiation and continuing until the risk of TLS has resolved.
High-risk patients require more intensive prophylaxis, including aggressive intravenous hydration and administration of Rasburicase.

Rasburicase (Fasturtec) used in the prevention and treatment of tumor lysis syndrome by rapidly lowering serum uric acid levels.
Rasburicase is contraindicated in patients with Glucose-6-phosphate dehydrogenase deficiency due to the risk of hemolysis. In such cases, management should consist of adequate hydration, allopurinol therapy, and close biochemical monitoring.
Urate samples obtained from patients receiving Rasburicase must be transported to the laboratory on ice to prevent in vitro degradation of uric acid and falsely low assay results.
In high-risk adults without established clinical or laboratory tumor lysis syndrome (TLS), prophylaxis can be achieved in the majority of patients with a single fixed dose of 3 mg rasburicase. This should be followed by close clinical and biochemical monitoring, with repeat dosing as required.
In high-risk pediatric patients without established TLS, prophylaxis is typically achieved with a single dose of rasburicase at 0.2 mg/kg.
Ongoing management requires close laboratory and clinical monitoring for evidence of evolving or progressive TLS. Although a fixed 3 mg dose may be considered in children, similar to adults, current evidence is insufficient to support a firm recommendation.
When rasburicase is used for the treatment or prophylaxis of TLS, concomitant use of Allopurinol is not recommended, as it may reduce the efficacy of rasburicase.
Urinary alkalinization is not recommended for TLS prophylaxis.
Treatment of Established Tumour Lysis Syndrome (TLS):
Management of established tumor lysis syndrome (TLS) requires a multidisciplinary approach involving hematology, nephrology, and intensive care teams.
If appropriate facilities for intensive monitoring and management are not available locally, early transfer to a high-dependency or intensive care unit, or to a specialist hematology center, should be considered in accordance with British Committee for Standards in Haematology guidance.
Potassium should not be added to intravenous fluids.
Urinary alkalinization is not recommended in the treatment of TLS.
Although Allopurinol is useful for prophylaxis, it is not the treatment of choice in established TLS, except in patients with Glucose-6-phosphate dehydrogenase deficiency or hypersensitivity to rasburicase.
In the absence of contraindications, patients with established TLS should receive Rasburicase at a dose of 0.2 mg/kg/day. The duration of therapy should be guided by clinical and biochemical response.
Asymptomatic hypocalcemia should not be treated. Symptomatic hypocalcemia should be managed with intravenous calcium gluconate at an appropriate dose based on age and weight, with close monitoring of calcium, phosphate, and renal function.
Patients with serum potassium ≥6 mmol/L or a ≥25% increase from baseline should undergo continuous cardiac monitoring.
Indications for renal replacement therapy include refractory fluid overload, hyperkalemia, hyperuricemia, hyperphosphatemia, or symptomatic hypocalcemia.
Peritoneal dialysis is not recommended for the management of TLS.
Renal replacement therapy should be continued until there is recovery of renal function, correction of electrolyte abnormalities, and restoration of adequate urine output.
Intravenous isotonic fluids should be administered to maintain a urine output of >4 mL/kg/hour in infants and >100 mL/m²/hour in older children and adults.
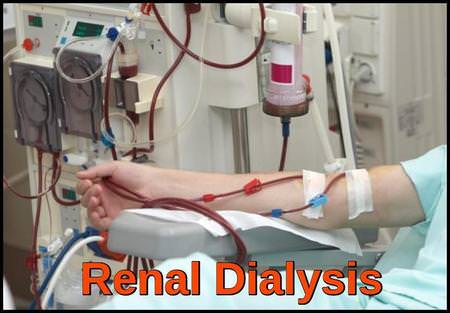
Renal dialysis (hemodialysis) used in the management of severe electrolyte imbalance and acute kidney injury in tumor lysis syndrome.
Summary:
Tumor Lysis Syndrome (TLS) is a life-threatening oncologic emergency caused by rapid destruction of malignant cells, leading to the release of potassium, phosphate, and nucleic acids into the bloodstream. This results in severe metabolic disturbances including hyperkalemia, hyperphosphatemia, hypocalcemia, and hyperuricemia, which may precipitate acute kidney injury, cardiac arrhythmias, and neurological complications. TLS most commonly occurs in patients with high-grade lymphomas such as Burkitt lymphoma and acute leukemias, but is increasingly recognized in solid tumors and with modern targeted therapies. Risk factors include high tumor burden, pre-existing renal impairment, dehydration, and inadequate prophylaxis.
Early risk stratification and prevention are essential, with management strategies tailored according to risk level. Low-risk patients require close monitoring and hydration, while intermediate-risk patients benefit from allopurinol prophylaxis. High-risk patients should receive aggressive intravenous hydration and rasburicase, a recombinant urate oxidase that rapidly lowers serum uric acid levels. In established TLS, management requires a multidisciplinary approach, including intensive monitoring, correction of electrolyte abnormalities, and avoidance of urinary alkalinization. Rasburicase is the treatment of choice unless contraindicated, such as in G6PD deficiency. Severe cases with refractory metabolic disturbances or fluid overload may require renal replacement therapy, with hemodialysis playing a critical role. Prompt recognition and evidence-based management significantly reduce morbidity and mortality associated with TLS.
Questions and Answers:
What is Tumor Lysis Syndrome (TLS)?
Tumor Lysis Syndrome (TLS) is a life-threatening oncologic emergency caused by rapid destruction of cancer cells, leading to the release of potassium, phosphate, and nucleic acids into the bloodstream, resulting in metabolic disturbances and acute kidney injury.
What causes Tumor Lysis Syndrome?
TLS occurs when malignant cells, particularly in high-grade lymphomas and acute leukemias, break down rapidly either spontaneously or following chemotherapy, releasing intracellular contents that overwhelm the body’s metabolic and renal capacity.
Which cancers are most commonly associated with TLS?
TLS is most commonly seen in aggressive hematologic malignancies such as Burkitt lymphoma and acute leukemias, but it is increasingly recognized in solid tumors and in patients receiving targeted therapies.
What are the key risk factors for Tumor Lysis Syndrome?
Major risk factors include high tumor burden, rapidly proliferating and treatment-sensitive cancers, pre-existing renal impairment, dehydration, hypotension, and inadequate prophylaxis.
What are the main biochemical abnormalities in TLS?
TLS is characterized by hyperkalemia, hyperphosphatemia, hyperuricemia, and secondary hypocalcemia, which can lead to cardiac arrhythmias, seizures, and renal failure.
How does Tumor Lysis Syndrome cause acute kidney injury?
Acute kidney injury in TLS results from precipitation of uric acid and calcium phosphate crystals within renal tubules, leading to obstruction, inflammation, and impaired renal function.
What are the ECG changes seen in hyperkalemia associated with TLS?
Hyperkalemia may cause characteristic ECG changes, including tall peaked T waves, PR prolongation, QRS widening, and potentially life-threatening arrhythmias.
How is Tumor Lysis Syndrome prevented?
Prevention involves risk stratification, aggressive hydration, close monitoring, and pharmacologic prophylaxis with allopurinol or rasburicase depending on the patient’s risk level.
What is the role of rasburicase in TLS?
Rasburicase rapidly lowers serum uric acid by converting it to allantoin, making it highly effective in both prevention and treatment of TLS, particularly in high-risk patients.
When is allopurinol used in Tumor Lysis Syndrome?
Allopurinol is mainly used for prophylaxis in low- to intermediate-risk patients and is not the preferred treatment in established TLS unless rasburicase is contraindicated.
Why is rasburicase contraindicated in some patients?
Rasburicase is contraindicated in patients with Glucose-6-phosphate dehydrogenase deficiency due to the risk of severe hemolysis.
Is urinary alkalinization recommended in TLS?
Urinary alkalinization is not recommended, as it may promote calcium phosphate precipitation and worsen renal complications.
When is dialysis required in Tumor Lysis Syndrome?
Renal replacement therapy is indicated in cases of refractory hyperkalemia, severe metabolic disturbances, fluid overload, or acute kidney injury unresponsive to medical management.
What is the role of hydration in TLS management?
Aggressive intravenous hydration enhances renal perfusion and promotes excretion of uric acid and electrolytes, reducing the risk of crystal formation and kidney injury.
Why must uric acid samples be handled carefully in patients receiving rasburicase?
Blood samples must be transported on ice to prevent in vitro degradation of uric acid by rasburicase, which can lead to falsely low laboratory results.
References:
MOORE AJ, VU MA, STRICKLAND SA. Supportive care in hematologic malignancies. In: GREER JP, ARBER DA, GLADER B, et al., editors. Wintrobe’s Clinical Hematology. 13th ed. Philadelphia: Wolters Kluwer/Lippincott Williams & Wilkins; 2013:1426–1466.
MIRRAKHIMOV AE, VOORE P, KHAN M, ALI AM. Tumor lysis syndrome: a clinical review. World J Crit Care Med. 2015;4(2):130–138. https://doi.org/10.5492/wjccm.v4.i2.130
CAIRO MS, COIFFIER B, REITER A, YOUNES A, TLS Expert Panel. Recommendations for the evaluation of risk and prophylaxis of tumour lysis syndrome (TLS) in adults and children with malignant diseases. Br J Haematol. 2010;149(4):578–586. https://doi.org/10.1111/j.1365-2141.2010.08143.x
IKEDA AK, EL-DEIRY WS. Tumor lysis syndrome: clinical presentation, history, and physical examination. Medscape. Available at: https://emedicine.medscape.com/article/282171-clinical
KALEMKERIAN GP, DARWISH B, VARTERASIAN ML. Tumor lysis syndrome in small-cell carcinoma and other solid tumors. Am J Med. 1997;103(5):363–367. https://doi.org/10.1016/S0002-9343(97)00231-9
CANADIAN CANCER SOCIETY. Tumour lysis syndrome. Available at: https://cancer.ca/en/cancer-information/diagnosis-and-treatment/managing-side-effects/tumour-lysis-syndrome
ABU-ALFA AK, YOUNES A. Tumor lysis syndrome and acute kidney injury: evaluation, prevention, and management. Am J Kidney Dis. 2010;55(Suppl 3):S1–S13. https://doi.org/10.1053/j.ajkd.2009.12.044
HOWARD SC, JONES DP, PUI CH. The tumor lysis syndrome. N Engl J Med. 2011;364(19):1844–1854.
HOWARD SC, PUI CH. Pitfalls in predicting tumor lysis syndrome. Leuk Lymphoma. 2006;47(4):782–785. https://doi.org/10.1080/10428190500399560
CHESON BD. Etiology and management of tumor lysis syndrome in patients with chronic lymphocytic leukemia. Clin Adv Hematol Oncol. 2009;7(4):263–271.
JONES GL, WILL A, JACKSON GH, WEBB NJA, RULE S, British Committee for Standards in Haematology. Guidelines for the management of tumour lysis syndrome in adults and children with haematological malignancies. Br J Haematol. 2015;169(5):661–671. https://doi.org/10.1111/bjh.13403
CAIRO MS, BISHOP M. Tumor lysis syndrome: new therapeutic strategies and classification. Br J Haematol. 2004;127(1):3–11. https://doi.org/10.1111/j.1365-2141.2004.05116.x
Keywords:
tumor lysis syndrome, tumor lysis syndrome TLS, TLS treatment, TLS prevention, TLS symptoms, TLS criteria Cairo Bishop, TLS management guidelines, tumor lysis syndrome pathophysiology, tumor lysis syndrome risk factors, tumor lysis syndrome complications, oncologic emergency TLS, hyperuricemia TLS, hyperkalemia ECG changes, hyperphosphatemia TLS, hypocalcemia TLS, acute kidney injury TLS, TLS dialysis indications, rasburicase mechanism uric acid allantoin, rasburicase dosing TLS, allopurinol prophylaxis TLS, TLS prophylaxis guidelines, TLS risk stratification, spontaneous tumor lysis syndrome, TLS laboratory abnormalities, TLS monitoring electrolytes, Burkitt lymphoma tumor lysis syndrome, acute lymphoblastic leukemia TLS, leukemia tumor lysis syndrome, lymphoma TLS risk, targeted therapy tumor lysis syndrome, venetoclax TLS risk, TLS ECG changes hyperkalemia, TLS renal failure mechanism, calcium phosphate precipitation kidney, uric acid nephropathy TLS, xanthine nephropathy allopurinol, G6PD deficiency rasburicase contraindication, TLS hydration protocol, TLS management algorithm, TLS ICU management, TLS pediatric management, TLS adult management, tumor lysis syndrome diagnosis, tumor lysis syndrome prevention strategies


Request Online Consultation With Dr M Abdou
Fee: US$100
Secure payment via PayPal (credit and debit cards accepted)
Pay Now


That is a dangerous desease
It is indeed and prevention is much easier than treatment!
I like what you guys are usually up too. Such clever work and exposure! Keep up the fantastic works guys I’ve added you guys to my own blogroll
Hi Rebeca,
Many thanks for your comment.
Pleased that you found this article helpful.
BW,
Thanks for the article, very interesting