Chemotherapeutic Agents
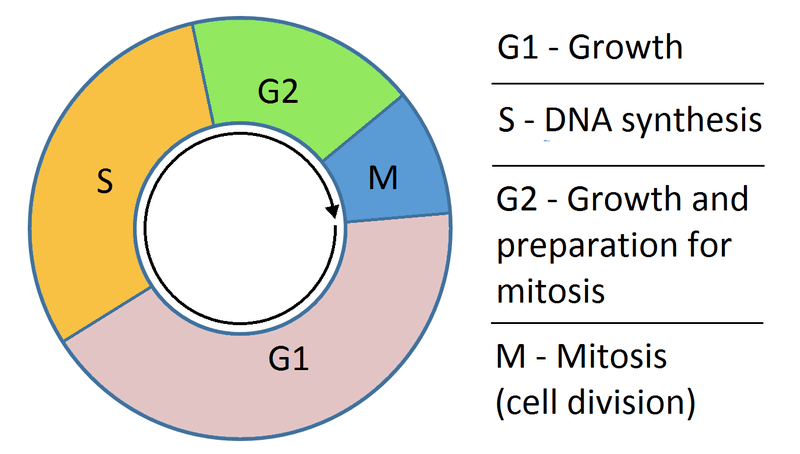
The eukaryotic cell cycle consists of distinct phases (G1, S, G2, and M) that regulate cellular growth and division, serving as critical targets for cell-cycle-specific and cell-cycle-independent chemotherapeutic agents.
This article reviews the foundational principles of chemotherapy to support the safe, effective clinical administration of various cytotoxic agents. We provide an in-depth analysis of cellular mechanisms of action, pharmacological drug classifications, and standardized combination regimens essential for the systemic management of neoplastic diseases and malignant tumors. Mastering these core hematology and oncology concepts is critical for optimizing patient treatment outcomes, calculating precise dosages, and anticipating acute or chronic toxicities.
Introduction:
The long-term understanding of cancer growth is that it is a net result of uncontrolled multiplication of cells that out paces the rate of natural cell death (apoptosis) within the tumor mass. The ultimate aim of the tumor is survival by overcoming the many barriers in its path. The imperative need for continual supply of nutrients to new growth areas, for instance, is met through sustained angiogenesis (formation of new blood vessels).
Another major factor for growth and survival is limitless replicative potential, requiring continued biosynthesis of the genetic material to provide a complementary set of chromosomes in each of the daughter cells following cell division. Inhibiting DNA replication, therefore, affords a logical approach for retarding tumor growth. For this reason, DNA has become a critical target in cancer chemotherapy. Indeed, many of the antitumor agents currently in the cancer armamentarium are DNA-interactive.
Cancer is the uncontrolled growth of cells coupled with malignant behavior: invasion and metastasis (among other features). It is caused by the interaction between genetic susceptibility and environmental factors. These factors lead to accumulations of genetic mutations in oncogenes (genes that control the growth rate of cells) and tumor suppressor genes (genes that help to prevent cancer), which gives cancer cells their malignant characteristics, such as uncontrolled growth.
In the broad sense, most chemotherapeutic drugs work by impairing mitosis (cell division), effectively targeting fast-dividing cells. As these drugs cause damage to cells, they are termed cytotoxic. They prevent mitosis by various mechanisms including damaging DNA and inhibition of the cellular machinery involved in cell division. One theory as to why these drugs kill cancer cells is that they induce a programmed form of cell death known as apoptosis.
Chemotherapy is a category of cancer treatment that uses one or more anti-cancer drugs (chemotherapeutic agents) as part of a standardized chemotherapy regimen.
Chemotherapeutic agents can be used to increase both the length and the quality of the lives of patients with a wide variety of neoplastic diseases.
Chemotherapy may be given with a curative intent (which almost always involves combinations of drugs), or it may aim to prolong life or to reduce symptoms (palliative chemotherapy).
The therapeutic index for many of these agents is very narrow; thus, close attention must be paid to proper dosing and timing of treatment. Proper patient selection is also critical for successful chemotherapy treatment. If the clinician is familiar with the agents being used, then toxicities can often be anticipated and then treated, or in some cases prevented, to ensure the continued quality of life.
By common usage, the term chemotherapy has come to connote the use of rather non-specific intracellular poisons, especially related to inhibiting the process of cell division known as mitosis, and generally excludes agents that more selectively block extracellular growth signals (i.e. blockers of signal transduction). To avoid these connotations, recently developed therapies (against specific molecular or genetic targets) which inhibit growth-promoting signals coming from classic endocrine hormones (primarily estrogens for breast cancer and androgens for prostate cancer) are called hormonal therapies, while the inhibition of other growth-promoting influences (especially those associated with receptor tyrosine kinases) is known as targeted therapy.
Importantly, the use of drugs (whether chemotherapy, hormonal therapy or targeted therapy) constitutes systemic therapy for cancer in that they are introduced into the bloodstream and are therefore in principle able to address cancer at any anatomic location in the body. Systemic therapy is often used in conjunction with other modalities that constitute local therapy (i.e. treatments whose efficacy is confined to the anatomic area where they are applied) for cancer such as radiation therapy, surgery or hyperthermia therapy.
Traditional chemotherapeutic agents are cytotoxic by means of interfering with cell division (mitosis) but cancer cells vary widely in their susceptibility to these agents. To a large extent, chemotherapy can be thought of as a way to damage or stress cells, which may then lead to cell death if apoptosis is initiated.
Many of the side effects of chemotherapy can be traced to damage to normal cells that divide rapidly and are thus sensitive to anti-mitotic drugs e.g. cells in the bone marrow, digestive tract and hair follicles. This results in the most common side-effects of chemotherapy: myelosuppression (decreased production of blood cells, hence also immunosuppression), mucositis (inflammation of the lining of the digestive tract), and alopecia (hair loss).

Chemotherapy-induced oral mucositis is a common, painful complication of cytotoxic systemic therapy resulting from direct mucosal toxicity and damage to rapidly dividing epithelial cells.
Because of the effect on immune cells (especially lymphocytes), chemotherapy drugs often find use in a host of diseases that result from harmful overactivity of the immune system against self (so-called autoimmune disorders). These include rheumatoid arthritis, systemic lupus erythematosus, multiple sclerosis, vasculitis and many others.
History of cancer chemotherapy:
The first use of small-molecule drugs to treat cancer was in the early 20th century, although the specific chemicals first used were not originally intended for that purpose. Mustard gas was used as a chemical warfare agent during World War I and was discovered to be a potent suppressor of hematopoiesis (blood production). A similar family of compounds known as nitrogen mustards was studied further during World War II at the Yale School of Medicine. It was reasoned that an agent that damaged the rapidly growing white blood cells might have a similar effect on cancer. Therefore, in December 1942, several patients with advanced lymphomas (cancers of the lymphatic system and lymph nodes) were given the drug by vein, rather than by breathing the irritating gas. Their improvement, although temporary, was remarkable.
Concurrently, during a military operation in World War II, following a German air raid on the Italian harbor of Bari, several hundred people were accidentally exposed to mustard gas, which had been transported there by the Allied forces to prepare for possible retaliation in the event of German use of chemical warfare. The survivors were later found to have very low white blood cell counts. After World War II was over and the reports declassified, the experiences converged and led researchers to look for other substances that might have similar effects against cancer. The first chemotherapy drug to be developed from this line of research was mustine. Since then, many other drugs have been developed to treat cancer, and drug development has exploded into a multibillion-dollar industry, although the principles and limitations of chemotherapy discovered by the early researchers still apply.
Treatment strategies:
There are a number of strategies in the administration of chemotherapeutic drugs used today.
Induction chemotherapy: is the first line treatment of cancer with a chemotherapeutic drug. This type of chemotherapy is used for curative intent.
Combined modality chemotherapy: is the use of drugs with other cancer treatments, such as surgery, radiation therapy, or hyperthermia therapy.
Consolidation chemotherapy: is given after remission in order to prolong the overall disease-free time and improve overall survival. The drug that is administered is the same as the drug that achieved remission.
Intensification chemotherapy: is identical to consolidation chemotherapy but a different drug than the induction chemotherapy is used.
Combination chemotherapy: involves treating a patient with a number of different drugs simultaneously. The drugs differ in their mechanism and side-effects. The biggest advantage is minimising the chances of resistance developing to any one agent. Also, the drugs can often be used at lower doses, reducing toxicity.
Neoadjuvant chemotherapy: is given prior to a local treatment such as surgery, and is designed to shrink the primary tumor. It is also given to cancers with a high risk of micrometastatic disease.
Adjuvant chemotherapy: is given after a local treatment (radiotherapy or surgery). It can be used when there is little evidence of cancer present, but there is a risk of recurrence. It is also useful in killing any cancerous cells that have spread to other parts of the body. These micrometastases can be treated with adjuvant chemotherapy and can reduce relapse rates caused by these disseminated cells.
Maintenance chemotherapy: is a repeated low-dose treatment to prolong remission.
Salvage chemotherapy or palliative chemotherapy: is given without curative intent, but simply to decrease tumor load and increase life expectancy. For these regimens, in general, a better toxicity profile is expected.
All chemotherapy regimens require that the patient be capable of undergoing the treatment. Performance status is often used as a measure to determine whether a patient can receive chemotherapy, or whether dose reduction is required.
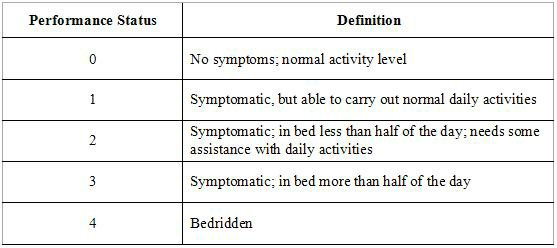
Chart illustrating Performance Status (e.g., ECOG scale), a crucial factor for determining patient eligibility and appropriate chemotherapy dosage.
Because only a fraction of the cells in a tumor die with each treatment (fractional kill), repeated doses must be administered to continue to reduce the size of the tumor. Current chemotherapy regimens apply drug treatment in cycles, with the frequency and duration of treatments limited by toxicity to the patient.
Dosage:
The efficacy of chemotherapy depends on the type of cancer and the stage. The overall effectiveness ranges from being curative for some cancers, such as some leukemias, to being ineffective, such as in some brain tumors, to being needless in others, like most non-melanoma skin cancers.
Dosage of chemotherapy can be difficult: If the dose is too low, it will be ineffective against the tumor, whereas, at excessive doses, the toxicity (side-effects) will be intolerable to the patient. The standard method of determining chemotherapy dosage is based on calculated body surface area (BSA). The BSA is usually calculated with a mathematical formula or a nomogram, using a patient’s weight and height, rather than by direct measurement of the body area.
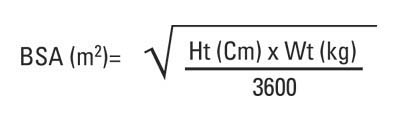
Body Surface Area (BSA) formula used for calculating chemotherapy dosage, showing the variables of height and weight.
Recently, the validity of this method in calculating uniform doses has been questioned. The reason for this is that the formula only takes into account the individual’s weight and height. Drug absorption and clearance are influenced by multiple factors, including age, gender, metabolism, disease state, organ function, drug-to-drug interactions, genetics, and obesity, which has a major impact on the actual concentration of the drug in the patient’s bloodstream.
Types of chemotherapy:
Alkylating agents:
Alkylating agents are the oldest group of chemotherapeutics in use today. Originally derived from mustard gas used in World War I, there are now many types of alkylating agents in use. They are so named because of their ability to alkylate many molecules, including proteins, RNA and DNA. This ability to bind covalently to DNA via their alkyl group is the primary cause for their anti-cancer effects. DNA is made of two strands and the molecules may either bind twice to one strand of DNA (intrastrand crosslink) or may bind once to both strands (interstrand crosslink). If the cell tries to replicate crosslinked DNA during cell division, or tries to repair it, the DNA strands can break. This leads to a form of programmed cell death called apoptosis.
Alkylating agents will work at any point in the cell cycle and thus are known as cell cycle-independent drugs. For this reason, the effect on the cell is dose dependent; the fraction of cells that die is directly proportional to the dose of drug.
The subtypes of alkylating agents are the nitrogen mustards, nitrosoureas, tetrazines, aziridines, cisplatins and derivatives, and non-classical alkylating agents.
Nitrogen mustards include mechlorethamine, cyclophosphamide, melphalan, chlorambucil, ifosfamide, and busulfan.
Nitrosoureas include carmustine (BCNU), lomustine (CCNU) and streptozotocin.
Tetrazines include dacarbazine, mitozolomide, and temozolomide.
Aziridines include thiotepa, mitomycin, and diaziquone (AZQ).
Cisplatin and derivatives include cisplatin, carboplatin, and oxaliplatin. They impair cell function by forming covalent bonds with the amino, carboxyl, sulfhydryl, and phosphate groups in biologically important molecules.
Non-classical alkylating agents include procarbazine and hexamethylmelamine.
Anti-metabolites:
Anti-metabolites are a group of molecules that interfere with DNA and RNA synthesis. Many of them have a similar structure to the building blocks of DNA and RNA. The building blocks are nucleotides; a molecule comprising a nucleobase, a sugar and a phosphate group. The nucleobases are divided into purines (guanine and adenine) and pyrimidines (cytosine, thymine, and uracil). Anti-metabolites resemble either nucleobases or nucleosides (a nucleotide without the phosphate group), but have altered chemical groups.
These drugs exert their effect by either blocking the enzymes required for DNA synthesis or becoming incorporated into DNA or RNA. By inhibiting the enzymes involved in DNA synthesis, they prevent mitosis because the DNA cannot duplicate itself. Also, after misincorporation of the molecules into DNA, DNA damage can occur and programmed cell death (apoptosis) is induced.
Unlike alkylating agents, anti-metabolites are cell cycle dependent. This means that they only work during a specific part of the cell cycle, in this case S-phase (the DNA synthesis phase). For this reason, at a certain dose, the effect plateaus and proportionally no more cell death occurs with increased doses.
Subtypes of the anti-metabolites are the anti-folates, fluoropyrimidines, deoxynucleoside analogues and thiopurines.
The anti-folates include methotrexate and pemetrexed. Methotrexate inhibits dihydrofolate reductase (DHFR), an enzyme that regenerates tetrahydrofolate from dihydrofolate. When the enzyme is inhibited by methotrexate, the cellular levels of folate coenzymes diminish. These are required for thymidylate and purine production, which are both essential for DNA synthesis and cell division. Pemetrexed is another anti-metabolite that affects purine and pyrimidine production, and therefore also inhibits DNA synthesis. It primarily inhibits the enzyme thymidylate synthase, but also has effects on DHFR, aminoimidazole carboxamide ribonucleotide formyltransferase, and glycinamide ribonucleotide formyltransferase.
The fluoropyrimidines include fluorouracil and capecitabine. Fluorouracil is a nucleobase analogue that is metabolized in cells to form at least two active products; 5-fluourouridine monophosphate (FUMP) and 5-fluoro-2′-deoxyuridine 5′-phosphate (fdUMP). FUMP becomes incorporated into RNA and fdUMP inhibits the enzyme thymidylate synthase; both of which lead to cell death. Capecitabine is a prodrug of 5-fluorouracil that is broken down in cells to produce the active drug.
The deoxynucleoside analogues include cytarabine, gemcitabine, decitabine, azacitidine, fludarabine, nelarabine, cladribine, clofarabine, and pentostatin.
The thiopurines include thioguanine and mercaptopurine.
Anti-microtubule agents:
Anti-microtubule agents are plant-derived chemicals that block cell division by preventing microtubule function. Microtubules are an important cellular structure composed of two proteins; α-tubulin and β-tubulin. They are hollow rod-shaped structures that are required for cell division, among other cellular functions. Microtubules are dynamic structures, which means that they are permanently in a state of assembly and disassembly.
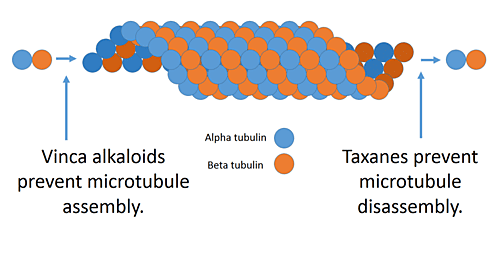
Diagram of Anti-microtubule agents (vinca alkaloids/taxanes) disrupting cell division, illustrating a key chemotherapy mechanism of action.
Vinca alkaloids and taxanes are the two main groups of anti-microtubule agents, and although both of these groups of drugs cause microtubule dysfunction, their mechanisms of action are completely opposite. The vinca alkaloids prevent the formation of the microtubules, whereas the taxanes prevent the microtubule disassembly. By doing so, they prevent the cancer cells from completing mitosis. Following this, cell cycle arrest occurs, which induces programmed cell death (apoptosis). Also, these drugs can affect blood vessel growth; an essential process that tumors utilize in order to grow and metastasize.
Vinca alkaloids are derived from the Madagascar periwinkle, Catharanthus roseus (formerly known as Vinca rosea). They bind to specific sites on tubulin, inhibiting the assembly of tubulin into microtubules. The original vinca alkaloids are natural products that include vincristine and vinblastine. Following the success of these drugs, semi-synthetic vinca alkaloids were produced: vinorelbine (used in the treatment of non-small-cell lung cancer), vindesine, and vinflunine. These drugs are cell cycle-specific. They bind to the tubulin molecules in S-phase and prevent proper microtubule formation required for M-phase.
Taxanes are natural and semi-synthetic drugs. The first drug of their class, paclitaxel, was originally extracted from the Pacific Yew tree, Taxus brevifolia. Now this drug and another in this class, docetaxel, are produced semi-synthetically from a chemical found in the bark of another Yew tree; Taxus baccata. These drugs promote microtubule stability, preventing their disassembly. Paclitaxel prevents the cell cycle at the boundary of G2-M, whereas docetaxel exerts its effect during S-phase. Taxanes present difficulties in formulation as medicines because they are poorly soluble in water.
Podophyllotoxin is an antineoplastic lignan obtained primarily from the American Mayapple (Podophyllum peltatum) and Himalayan Mayapple (Podophyllum hexandrum or Podophyllum emodi). It has anti-microtubule activity, and its mechanism is similar to that of vinca alkaloids in that they bind to tubulin, inhibiting microtubule formation. Podophyllotoxin is used to produce two other drugs with different mechanisms of action: etoposide and teniposide.
Topoisomerase inhibitors:
Because DNA is the blueprint for the human body, any damage to a cell’s DNA can result in cancer. DNA is normally a coiled double helix of two strands; it periodically becomes uncoiled in the process of replication during cell division and in the process transcribing the genetic code to make new proteins. Two enzymes that play roles in this uncoiling and recoiling process are topoisomerase I and topoisomerase II. They also play a significant role in fixing DNA damage that occurs as a result of exposure to harmful chemicals or UV rays. Inhibition of topoisomerase I or II interferes with DNA uncoiling and recoiling processes.
Topoisomerase I inhibitors include irinotecan and topotecan. Topoisomerase II inhibitors include doxorubicin, etoposides and mitoxantrone.
Cytotoxic antibiotics:
The cytotoxic antibiotics are a varied group of drugs that have various mechanisms of action. The common theme that they share in their chemotherapy indication is that they interrupt cell division. The most important subgroup is the anthracyclines and the bleomycins; other prominent examples include mitomycin C, mitoxantrone, and actinomycin.
Among the anthracyclines, doxorubicin and daunorubicin were the first, and were obtained from the bacterium Streptomyces peucetius. Derivatives of these compounds include epirubicin and idarubicin. The mechanisms of anthracyclines include DNA intercalation (molecules insert between the two strands of DNA), generation of highly reactive free radicals that damage intercellular molecules and topoisomerase inhibition.
Delivery:
Most chemotherapy is delivered intravenously, although a number of agents can be administered orally (e.g., melphalan, chlorambucil, busulfan, capecitabine).
Depending on the patient, the type and stage of cancer, the type of chemotherapy, and the dosage, intravenous chemotherapy may be given on either an inpatient or an outpatient basis. For continuous, frequent or prolonged intravenous chemotherapy administration, various systems may be surgically inserted into the vasculature to maintain access. Commonly used systems are the Hickman line, the Port-a-Cath, and the PICC line. These have a lower infection risk, are much less prone to phlebitis or extravasation, and eliminate the need for repeated insertion of peripheral cannulae.
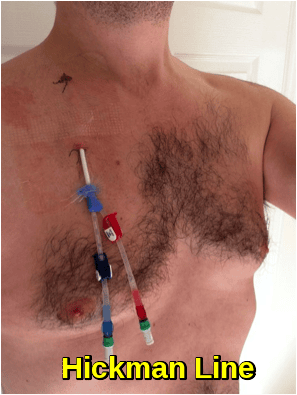
Hickman Line, a type of central venous catheter (CVC) used for the long-term, safe administration of chemotherapy agents.
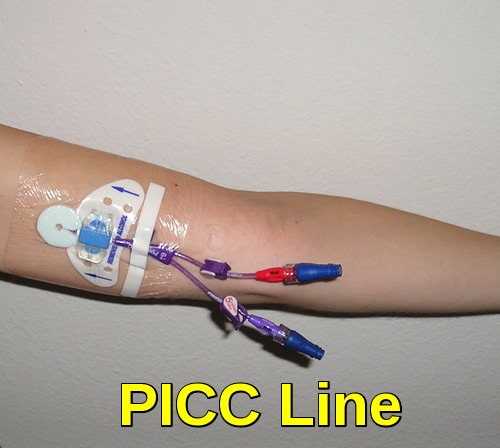
PICC Line (Peripherally Inserted Central Catheter), commonly used for long-term chemotherapy administration and venous access in oncology patients.
Isolated limb perfusion (often used in melanoma), or isolated infusion of chemotherapy into the liver or the lung have been used to treat some tumors. The main purpose of these approaches is to deliver a very high dose of chemotherapy to tumor sites without causing overwhelming systemic damage. These approaches can help control solitary or limited metastases, but they are by definition not systemic, and, therefore, do not treat distributed metastases or micrometastases.
Topical chemotherapies, such as 5-fluorouracil, are used to treat some cases of non-melanoma skin cancer.
If cancer has central nervous system involvement, or with meningeal disease, intrathecal chemotherapy may be administered.
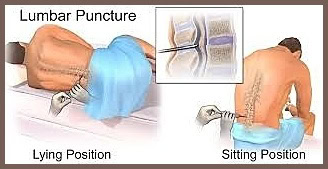
Administration of Intrathecal Chemotherapy into the subarachnoid space for treating CNS involvement in cancer.
Adverse Effects:
Cancer cells tend to grow fast, and chemotherapy drugs kill fast-growing cells. But because these drugs travel throughout the body, they can affect normal, healthy cells that are fast-growing, too. Damage to healthy cells causes side effects.
The normal cells most likely to be damaged by chemotherapy are :
- Blood-forming cells in the bone marrow.
- Hair Follicles.
- Cells in the mouth, digestive tract, and reproductive system.
- Some chemotherapy drugs can damage cells in the heart, kidneys, bladder, lungs, and nervous system.
Chemotherapy-related toxicities can occur acutely after administration, within hours or days, or chronically, from weeks to years.
Fatigue can result from a number of factors, including anemia, depression, and radiation therapy.
Hair loss (alopecia) affects many patients and can be one of the most disturbing side effects, sometimes preventing people from receiving chemotherapy. Hair usually starts to regrow a few weeks after the last treatment, and can sometimes change color, texture, thickness, and style. Sometimes hair has a tendency to curl after regrowth, resulting in “chemo curls”. Severe hair loss occurs most often with drugs such as doxorubicin, daunorubicin, paclitaxel, docetaxel, cyclophosphamide, ifosfamide, and etoposide. Chemotherapy induces hair loss in women more often than men. Permanent thinning or hair loss can result from some standard chemotherapy regimens.
Virtually all chemotherapeutic regimens can cause immune suppression and myelosuppression, often by paralyzing the bone marrow and leading to a decrease of white blood cells, red blood cells, and platelets. Anemia and thrombocytopenia, when they occur, are improved with red cell and platelet transfusion. Neutropenia (a decrease of the neutrophil granulocyte count below 0.5 x 109/litre) can be improved with synthetic G-CSF (granulocyte-colony-stimulating factor, e.g., filgrastim, lenograstim). In very severe myelosuppression, which occurs in some regimens, almost all the bone marrow stem cells are destroyed, meaning allogenic or autologous bone marrow cell transplants are necessary. Although patients are encouraged to wash their hands, avoid sick people, and take other infection-reducing steps, about 85% of infections are due to naturally occurring microorganisms in the patient’s own gastrointestinal tract (including oral cavity) and skin. This may manifest as systemic infections, such as sepsis, or as localized outbreaks, such as Herpes simplex, shingles, or other members of the Herpesviridea. The risk of illness and death can be reduced by taking common prophylactic antimicrobials such as quinolones, trimethoprim/sulfamethoxazole, and aciclovir before any fever or sign of infection appears.
Nausea and vomiting are two of the most feared cancer treatment-related side-effects for cancer patients and their families. Chemotherapy-induced nausea and vomiting (CINV) are common with many treatments and some forms of cancer. Since the 1990s, several novel classes of antiemetics have been developed and commercialized, becoming a nearly universal standard in chemotherapy regimens, and helping to successfully manage these symptoms in a large portion of patients. Chemotherapy regimens are classified into four categories according to potential for CINV—highly emetogenic (>90%), moderately emetogenic (30–90%), low emetogenic (10–30%), and minimally emetogenic (<10%). Cisplatin, carboplatin, cyclophosphamide, and doxorubicin-based regimens are moderate/highly emetogenic agents leading to CINV.

Chemotherapy-induced nausea and vomiting (CINV), a common and severe side effect of cytotoxic cancer treatment.
Other gastrointestinal side effects, such as diarrhea, and constipation are also common side effects experienced during chemotherapy.
Sexual dysfunction resulting from chemotherapy and radiation therapy can often result in loss of libido.
Some types of chemotherapy are gonadotoxic and may cause infertility. Chemotherapies with high risk include procarbazine and other alkylating drugs such as cyclophosphamide, ifosfamide, busulfan, melphalan, and chlorambucil. Drugs with medium risk include doxorubicin and platinum analogs such as cisplatin and carboplatin. On the other hand, therapies with low risk of gonadotoxicity include plant derivatives such as vincristine and vinblastine, antibiotics such as bleomycin and dactinomycin, and antimetabolites such as methotrexate, mercaptopurine, and 5-fluorouracil. Female infertility by chemotherapy appears to be secondary to premature ovarian failure by loss of primordial follicles. Patients may choose between several methods of fertility preservation prior to chemotherapy, including cryopreservation of semen, ovarian tissue, oocytes, or embryos.
Chemotherapy is teratogenic during pregnancy, especially during the first trimester, to the extent that abortion usually is recommended if pregnancy in this period is found during chemotherapy. Second- and third-trimester exposure does not usually increase the teratogenic risk and adverse effects on cognitive development, but it may increase the risk of various complications of pregnancy and fetal myelosuppression.
Affecting about 60% to 80% of patients receiving chemotherapy, cutaneous toxicities can include skin rash and mucositis. Mucositis is also one of the common side effects experienced as a result of radiation therapy and chemotherapy treatment e.g. methotrexate, with mouth sores often affecting food intake.
Thrombocytopenia (low platelet count) can affect clotting and cause bleeding and is caused by alkylating, antirheumatic, and antimetabolite agents.
Between 30 and 40 percent of patients undergoing chemotherapy experience chemotherapy-induced peripheral neuropathy (CIPN), a progressive, enduring, and often irreversible condition, causing pain, tingling, numbness and sensitivity to cold, beginning in the hands and feet and sometimes progressing to the arms and legs. Chemotherapy drugs associated with CIPN include thalidomide, vinca alkaloids, taxanes, proteasome inhibitors, and the platinum-based drugs. Whether CIPN arises, and to what degree, is determined by the choice of drug, duration of use, the total amount consumed and whether the patient already has peripheral neuropathy. Though the symptoms are mainly sensory, in some cases motor nerves and the autonomic nervous system are affected. CIPN often follows the first chemotherapy dose and increases in severity as treatment continues, but this progression usually levels off at the completion of treatment. The platinum-based drugs are the exception; with these drugs, sensation may continue to deteriorate for several months after the end of treatment. Some CIPN appears to be irreversible. Pain can often be managed with drug or other treatment but the numbness is usually resistant to treatment.
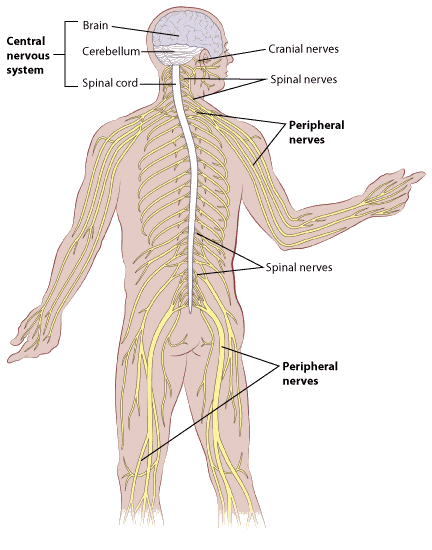
Diagram of the Central Nervous System (CNS), which can be the site of chemotherapy-induced neurotoxicity or require intrathecal administration.
Damage to the optic nerve and the ocular motor nerves can often occur when a patient is receiving antineoplastic chemotherapy. “Chemo-brain” can be a side effect of cancer therapy that causes memory loss and other cognitive changes.
Development of secondary neoplasia after successful chemotherapy or radiotherapy treatment can occur. The most common secondary neoplasm is secondary acute myeloid leukemia, which develops primarily after treatment with alkylating agents or topoisomerase inhibitors. Survivors of childhood cancer are more than 13 times as likely to get a secondary neoplasm during the 30 years after treatment than the general population.
Certain chemotherapy agents, such as antimetabolites, can cause hepatotoxicity and nephrotoxicity.
Alkylating agents e.g. ifosfamide can cause hemorrhagic cystitis, resulting in dysuria, frequency, urgency, nocturia, suprapubic pain, and microscopic or gross hematuria.
One of the most serious side effects of cancer treatment is cardiotoxicity e.g. anthracyclines, which can occur early or late in treatment.
In particularly large tumors and cancers with high white cell counts, such as lymphomas, teratomas, and some leukemias, some patients develop tumor lysis syndrome. The rapid breakdown of cancer cells causes the release of chemicals from the inside of the cells. Following this, high levels of uric acid, potassium, and phosphate are found in the blood. High levels of phosphate induce secondary hypoparathyroidism, resulting in low levels of calcium in the blood. This causes kidney damage and the high levels of potassium can cause cardiac arrhythmia. Although prophylaxis with allopurinol or rasburicase is available and is often initiated in patients with large tumors, this is a dangerous side-effect that can lead to death if left untreated.
Limitations:
Chemotherapy does not always work, and even when it is useful, it may not completely destroy cancer.
The blood-brain barrier poses a difficult obstacle to pass to deliver chemotherapy to the brain. This is because the brain has an extensive system in place to protect it from harmful chemicals. Drug transporters can pump out drugs from the brain and brain’s blood vessel cells into the cerebrospinal fluid and blood circulation. These transporters pump out most chemotherapy drugs, which reduces their efficacy for treatment of brain tumors. Only small lipophilic alkylating agents such as lomustine or temozolomide are able to cross this blood-brain barrier.
Blood vessels in tumors are very different from those seen in normal tissues. As a tumor grows, tumor cells furthest away from the blood vessels become low in oxygen (hypoxic). To counteract this they then signal for new blood vessels to grow (angiogenesis). The newly formed tumor vasculature is poorly formed and does not deliver an adequate blood supply to all areas of the tumor. This leads to issues with drug delivery because many drugs will be delivered to the tumor by the circulatory system.
Resistance:
Resistance is a major cause of treatment failure in chemotherapeutic drugs. There are a few possible causes of resistance in cancer, one of which is the presence of small pumps on the surface of cancer cells that actively move chemotherapy from inside the cell to the outside. Cancer cells produce high amounts of these pumps, known as p-glycoprotein, in order to protect themselves from chemotherapeutics.
Another mechanism of resistance is gene amplification, a process in which multiple copies of a gene are produced by cancer cells. This overcomes the effect of drugs that reduce the expression of genes involved in replication. With more copies of the gene, the drug cannot prevent all expression of the gene and therefore the cell can restore its proliferative ability.
Cancer cells can also cause defects in the cellular pathways of apoptosis (programmed cell death). As most chemotherapy drugs kill cancer cells in this manner, defective apoptosis allows survival of these cells, making them resistant.
Targeted Therapies:
Targeted therapies are a relatively new class of cancer drugs that can overcome many of the issues seen with the use of cytotoxics. They are divided into two groups: small molecule and antibodies.
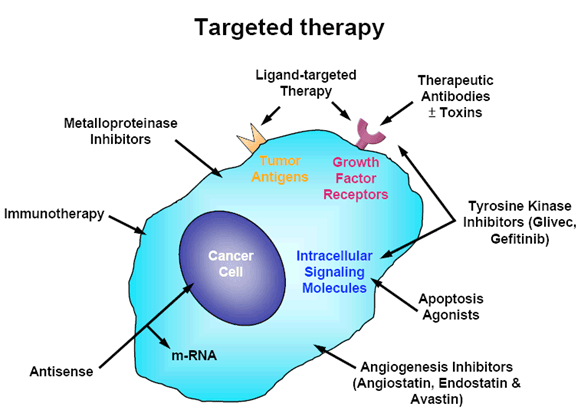
Diagram illustrating the mechanism of Targeted Therapy (monoclonal antibody or TKI), showing selective binding to a specific molecular cancer target.
The massive toxicity seen with the use of cytotoxics is due to the lack of cell specificity of the drugs. They will kill any rapidly dividing cell, tumor or normal.
Targeted therapies are designed to affect cellular proteins or processes that are utilized by the cancer cells. This allows a high dose to cancer tissues with a relatively low dose to other tissues. As different proteins are utilized by different cancer types, the targeted therapy drugs are used on a cancer type specific, or even on a patient specific basis.
Although the side effects are often less severe than that seen of cytotoxic chemotherapeutics, life-threatening effects can occur.
Initially, the targeted therapeutics were supposed to be solely selective for one protein. Now it is clear that there is often a range of protein targets that the drug can bind. An example target for targeted therapy is the protein produced by the Philadelphia chromosome, a genetic lesion found commonly in chronic myeloid leukemia. This fusion protein has enzyme activity that can be inhibited by imatinib, a small molecule drug.
Summary:
This comprehensive clinical guide establishes the fundamental principles of cancer chemotherapy, detailing the pharmacological mechanisms, classification, and administration strategies of cytotoxic agents. It explores the systemic nature of chemotherapy as opposed to localized modalities like surgery and radiation, explaining how these drugs selectively disrupt cell division by targeting rapid cellular replication. The article highlights the historical evolution of systemic therapies from wartime nitrogen mustards to modern combination regimens designed to minimize drug resistance. Key treatment methodologies—including induction, consolidation, neoadjuvant, and adjuvant therapies are systematically outlined alongside the standard practices and limitations of using Body Surface Area (BSA) for clinical dosing. Finally, it addresses the physiological basis behind classic chemotherapy-induced toxicities, such as myelosuppression, alopecia, and oral mucositis, which stem from collateral damage to healthy, rapidly proliferating non-malignant tissues.
Questions and Answers:
What is the primary mechanism of action of classic cytotoxic chemotherapy agents?
Traditional cytotoxic chemotherapy agents primarily work by disrupting cell division (mitosis) in rapidly proliferating cells. They achieve this through various mechanisms, including directly damaging genetic material, forming crosslinks within or between DNA strands, or inhibiting the essential enzymatic and cellular machinery required for nucleic acid synthesis and replication. This cellular stress and structural disruption often activates programmatic cell death pathways, leading to apoptosis of the malignant cells.
How does systemic cancer therapy differ from localized treatment modalities?
Systemic therapies, which include cytotoxic chemotherapy, hormonal therapies, and targeted molecular therapies, are introduced directly into the bloodstream. This allows the active agents to distribute throughout the entire vascular and tissue architecture of the body, making them capable of treating disseminated disease, macro-metastases, and occult micrometastases at any anatomical site. In contrast, local therapies, such as surgical resection or radiation therapy, confine their therapeutic efficacy strictly to the specific anatomical zone where they are physically applied.
What are the clinical differences between neoadjuvant and adjuvant chemotherapy?
Neoadjuvant chemotherapy is administered prior to local interventions like surgery or radiation with the primary intent of shrinking the primary tumor mass to facilitate a more effective, less invasive resection, while also addressing early micrometastatic disease. Adjuvant chemotherapy is delivered after definitive local therapy has been completed, targeting any remaining microscopic or occult cancer cells that may have shed into the circulation, thereby significantly reducing the clinical risk of disease recurrence or distant relapse.
Why does cytotoxic chemotherapy frequently cause side effects like myelosuppression and oral mucositis?
Traditional chemotherapeutic agents are non-specific intracellular toxins that cannot differentiate between rapidly dividing malignant cells and rapidly dividing healthy cells. Consequently, normal tissues with high baseline turnover rates are susceptible to collateral damage during treatment cycles. Direct injury to the rapidly proliferating hematopoietically active stem cells in the bone marrow causes myelosuppression, while damage to the basal epithelial cells lining the gastrointestinal tract and oral cavity leads to mucosal ulceration, inflammation, and clinical oral mucositis.
What is the clinical rationale behind using combination chemotherapy regimens over single-agent therapies?
Combination chemotherapy involves the simultaneous administration of multiple cytotoxic drugs that feature distinct mechanisms of action and non-overlapping toxicity profiles. The primary clinical advantage of this strategy is that it targets separate biochemical pathways within the heterogeneous tumor cell population, significantly minimizing the mathematical probability of the cancer developing clonal drug resistance. Furthermore, utilizing multiple agents often allows clinicians to optimize therapeutic efficacy at lower, more tolerable doses of individual drugs, reducing the severity of specific organ-toxicities.
How is chemotherapy dosing traditionally calculated, and what are its clinical limitations?
Chemotherapy dosing is traditionally calculated based on a patient’s estimated Body Surface Area (BSA), derived via standardized nomograms or mathematical formulas utilizing height and weight. The major limitation of this conventional method is that it assumes a uniform relationship between surface area and drug handling, completely ignoring critical individual pharmacokinetic variables. Actual drug absorption, systemic concentration, and metabolic clearance are heavily influenced by independent patient factors, including advancing age, gender, genetic polymorphisms, underlying organ function, body composition (obesity), and concurrent drug-to-drug interactions.
References:
Airley R. Cancer Chemotherapy. Chichester: Wiley-Blackwell; 2009. ISBN 978-0470092548. Available from: https://onlinelibrary.wiley.com/book/10.1002/9780470745267
Wood M, Brighton D. The Royal Marsden Hospital Handbook of Cancer Chemotherapy: A Guide for the Multidisciplinary Team. St. Louis (MO): Elsevier Churchill Livingstone; 2005. ISBN 978-0443071010.
Perry MC. The Chemotherapy Source Book. 4th ed. Philadelphia (PA): Wolters Kluwer Health / Lippincott Williams & Wilkins; 2008. ISBN 978-0781773287.
Epstein RJ. Maintenance therapy to suppress micrometastasis: the new challenge for adjuvant cancer treatment. Clin Cancer Res. 2005 Aug 1;11(15):5337-41. doi: 10.1158/1078-0432.CCR-05-0210. PMID: 16061843. Available from: https://aacrjournals.org/clincancerres/article/11/15/5337/190802/Maintenance-Therapy-to-Suppress-Micrometastasis
Skeel RT. Handbook of Cancer Chemotherapy. 6th ed. Philadelphia (PA): Lippincott Williams & Wilkins; 2003. ISBN 978-0781736299.
Nastoupil LJ, Rose AC, Flowers CR. Diffuse large B-cell lymphoma: current treatment approaches. Oncology (Williston Park). 2012 May;26(5):488-95. PMID: 22730635. Available from: https://www.cancernetwork.com/view/diffuse-large-b-cell-lymphoma-current-treatment-approaches
Lind MJ. Principles of cytotoxic chemotherapy. Medicine. 2008 Jan 1;36(1):19-23. doi: 10.1016/j.mpmed.2007.10.003. Available from: https://www.amedicinejournal.com/article/S1357-3039(07)00397-2/fulltext
Siddik ZH. Mechanisms of Action of Cancer Chemotherapeutic Agents: DNA-Interactive Alkylating Agents and Antitumour Platinum-Based Drugs. In: John Wiley & Sons, Ltd, editors. Encyclopedia of Life Sciences. Chichester: John Wiley & Sons, Ltd; 2005. doi: 10.1038/npg.els.0003923. Available from: https://onlinelibrary.wiley.com/doi/10.1038/npg.els.0003923
Pinkel D. The use of body surface area as a criterion of drug dosage in cancer chemotherapy. Cancer Res. 1958 Aug;18(7):853-6. PMID: 13561330. Available from: https://cancerres.aacrjournals.org/content/18/7/853
Parker WB. Enzymology of purine and pyrimidine antimetabolites used in the treatment of cancer. Chem Rev. 2009 Jul 8;109(7):2880-93. doi: 10.1021/cr900028p. PMID: 19476376. Available from: https://pubs.acs.org/doi/10.1021/cr900028p
Adjei AA. Pemetrexed (ALIMTA), a novel multitargeted antineoplastic agent. Clin Cancer Res. 2004 Jun 15;10(12 Pt 2):4276s-80s. doi: 10.1158/1078-0432.CCR-040005. PMID: 15217971. Available from: https://aacrjournals.org/clincancerres/article/10/12/4276s/197170/Pemetrexed-ALIMTA-A-Novel-Multitargeted
Rowinsky EK, Donehower RC. The clinical pharmacology and use of antimicrotubule agents in cancer chemotherapeutics. Pharmacol Ther. 1991 Oct;52(1):35-84. doi: 10.1016/0163-7258(91)90086-2. PMID: 1686950. Available from: https://www.sciencedirect.com/science/article/abs/pii/0163725891900862
Yue QX, Liu X, Guo DA. Microtubule-binding natural products for cancer therapy. Planta Med. 2010 Aug;76(11):1037-43. doi: 10.1055/s-0030-1250118. PMID: 20623512. Available from: https://www.thieme-connect.com/products/ejournals/abstract/10.1055/s-0030-1250118
Lodish H, Berk A, Zipursky SL, Matsudaira P, Baltimore D, Darnell J. Molecular Cell Biology. 4th ed. Section 12.3, The Role of Topoisomerases in DNA Replication. New York: W. H. Freeman; 2000. Available from: https://www.ncbi.nlm.nih.gov/books/NBK21490/
Elad S, Zadik Y, Hewson I, Baddour LM, Boucek RJ, Aitken SL, et al. A systematic review of viral infections associated with oral involvement in cancer patients: a spotlight on Herpesviridae. Support Care Cancer. 2010 Aug;18(8):993-1006. doi: 10.1007/s00520-010-0901-7. PMID: 20464421. Available from: https://link.springer.com/article/10.1007/s00520-010-0901-7
Brydøy M, Fosså SD, Dahl O, Bjøro T. Gonadal dysfunction and fertility problems in cancer survivors. Acta Oncol. 2007 Jan 1;46(4):480-9. doi: 10.1080/02841860601166958. PMID: 17497316. Available from: https://www.tandfonline.com/doi/full/10.1080/02841860601166958
del Pino BM. Chemotherapy-induced Peripheral Neuropathy. NCI Cancer Bull. 2010 Feb 23;7(4):6.
Rüther U, Nunnensiek C, Schmoll HJ, editors. Secondary Neoplasias Following Chemotherapy, Radiotherapy, and Immunosuppression. Basel: Karger; 2000. (Contributions to Oncology; vol. 55). doi: 10.1159/isbn.978-3-318-00470-9. Available from: https://www.karger.com/Book/Home/224671
Arnon J, Meirow D, Lewis-Roness H, Ornoy A. Genetic and teratogenic effects of cancer treatments on gametes and embryos. Hum Reprod Update. 2001 Jul-Aug;7(4):394-403. doi: 10.1093/humupd/7.4.394. PMID: 11476352. Available from: https://academic.oup.com/humupd/article/7/4/394/636881
Deeken JF, Löscher W. The blood-brain barrier and cancer: transporters, treatment, and Trojan horses. Clin Cancer Res. 2007 Mar 15;13(6):1663-74. doi: 10.1158/1078-0432.CCR-06-1574. PMID: 17363513. Available from: https://aacrjournals.org/clincancerres/article/13/6/1663/194488/The-Blood-Brain-Barrier-and-Cancer-Transporters
Makin G, Hickman JA. Apoptosis and cancer chemotherapy. Cell Tissue Res. 2000 Jul;301(1):143-52. doi: 10.1007/s004410000160. PMID: 10924265. Available from: https://link.springer.com/article/10.1007/s004410000160
Gerber DE. Targeted therapies: a new generation of cancer treatments. Am Fam Physician. 2008 Feb 1;77(3):311-9. PMID: 18297954. Available from: https://www.aafp.org/pubs/afp/issues/2008/0201/p311.html
Krumbhaar EB. Role of the blood and the bone marrow in certain forms of gas poisoning. JAMA. 1919 Jan 4;72(1):39-41. doi: 10.1001/jama.1919.02610010045012. Available from: https://jamanetwork.com/journals/jama/article-abstract/437033
Fenn JE, Udelsman R. First use of intravenous chemotherapy cancer treatment: rectifying the record. J Am Coll Surg. 2011 Mar;212(3):413-7. doi: 10.1016/j.jamcollsurg.2010.10.018. PMID: 21147008. Available from: [suspicious link removed]
Goodman LS, Wintrobe MM, Dameshek W, Goodman MJ, Gilman A, McLennan MT. Nitrogen mustard therapy for Hodgkin’s disease, lymphosarcoma, leukemia and related disorders. JAMA. 1946 Sep 21;132(3):126-32. doi: 10.1001/jama.1946.02870380008004. PMID: 20997190. Available from: https://jamanetwork.com/journals/jama/article-abstract/266211
Joensuu H. Systemic chemotherapy for cancer: from weapon to treatment. Lancet Oncol. 2008 Mar;9(3):304-12. doi: 10.1016/S1470-2045(08)70075-X. PMID: 18308253. Available from: https://www.thelancet.com/journals/lanonc/article/PIIS1470-2045(08)70075-X/fulltext
Keywords:
chemotherapy principles, cytotoxic agents, alkylating agents, antimetabolites, chemotherapy mechanism of action, chemotherapy side effects, chemotherapy regimens, induction chemotherapy, adjuvant chemotherapy, neoadjuvant chemotherapy, combination chemotherapy, myelosuppression, alopecia, cancer treatment, chemotherapy drugs, systemic therapy, targeted molecular therapy, cellular mechanisms of action, pharmacokinetics, body surface area dosing, bsa limitations, nitrogen mustards history, cell cycle independent drugs, mitotic inhibitors, topoisomerase inhibition, oral mucositis clinical reference, chemotherapy induced peripheral neuropathy, fractional kill hypothesis, oncology training guides, hematology website articles, ask hematologist


Request Online Consultation With Dr M Abdou
Fee: US$100
Secure payment via PayPal (credit and debit cards accepted)
Pay Now