Surgery and Hemostasis

Intraoperative surgical hemostasis demonstrating active bleeding control using electrocautery and precise surgical technique to minimize blood loss.
What is Hemostasis?
Hemostasis is the physiological process by which the body prevents and stops bleeding following vascular injury. It involves a tightly regulated interaction between platelets, coagulation factors, the endothelium, and fibrinolytic pathways to form a stable clot and restore vascular integrity.
When a blood vessel is damaged, platelets rapidly adhere to the injured endothelium and aggregate to form a primary platelet plug. Simultaneously, circulating clotting factors activate the coagulation cascade, generating fibrin strands that stabilize the platelet plug into a durable hemostatic clot. This coordinated response prevents excessive blood loss while allowing tissue repair and wound healing to proceed.
Under normal conditions, bleeding control occurs rapidly and efficiently. However, in major trauma or surgical procedures, physiological hemostasis may be insufficient, and clinicians must actively manage surgical bleeding to minimize blood loss, transfusion requirements, and perioperative complications.
Disorders of hemostasis may be congenital or acquired. Inherited bleeding disorders such as hemophilia impair clot formation and significantly increase surgical bleeding risk. Conversely, individuals without a prior bleeding history may develop acquired conditions, including acquired hemophilia, that disrupt normal coagulation.
Importantly, blood coagulation is a double-edged system. While it exists to prevent hemorrhage, surgery and trauma are also among the strongest risk factors for thrombosis. Surgical stress can increase baseline thrombotic risk dramatically, whereas patients with even mild coagulation defects may experience disproportionate bleeding following invasive procedures. This delicate balance between hemorrhage and thrombosis underpins the complexity of perioperative hemostatic management.
Surgery For Patients With Congenital Hemostatic Defects:
Currently, hemophiliacs can and do undergo any and all surgical procedures as dictated by their medical condition. Adequate levels of their genetically absent factor allow the operator to do whatever would be done in a similar procedure in a patient without hemophilia. A notable exception includes those hemophilia patients who have or are suspected to have an inhibitor. In this clinical scenario, it is generally not advised to perform elective surgery. Consultation with a center having experience and resources to treat these patients is in order.
Prophylaxis Against Thrombosis:
Prophylaxis against deep vein thrombosis (DVT) and pulmonary embolism in surgical patients is thoroughly reviewed in a 2012 consensus. In general, prophylaxis should be more aggressively used in hospitalized patients. Prophylaxis is safer and more effective than generally held while the toll of venous thromboembolism (VTE) without prophylaxis is usually significantly underestimated. The role of mechanical and similar adjuncts in prophylaxis against VTE verifies their use. Westrich demonstrated that intermittent pneumatic compression (IPC) devices in total knee replacement surgery were more effective than low molecular weight heparin (LMWH) and far more effective than aspirin alone in reducing both DVT and pulmonary embolism. However, they did notice only a 33% compliance rate with patients for whom IPC was prescribed. Others have demonstrated not only the effectiveness of IPC and similar devices but the additive effect of this mechanical methodology with pharmacologic prophylaxis.
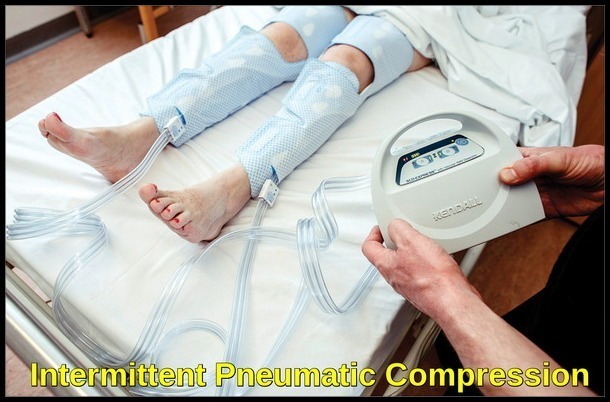
Intermittent pneumatic compression device used for postoperative venous thromboembolism (VTE) prophylaxis in surgical patients.
There remains a reticence on the part of some surgeons citing concern of possible additional risk for hemorrhage as a result of employing VTE prophylaxis. For many indications, IPC is probably effective. However, several analyses of VTE prophylaxis in neurosurgical patients have indicated that not only is chemical prophylaxis itself well-tolerated, but when combined with IPC, it adds to the efficacy of VTE prophylaxis either without any additional risk of hemorrhage or with such a small amount of increased risk that such risk of hemorrhage is easily absorbed by the decrease in overall morbidity and mortality from VTE. Additionally, successful employment of VTE prophylaxis avoids the concern for much higher and longer dosage of therapeutic anticoagulation should a VTE occur.

Summary of key concepts in surgical hemostasis, including perioperative hemorrhage risk, antithrombotic prophylaxis, and VTE prevention strategies.
Perioperative Guidelines on Antiplatelet and Anticoagulant Agents:
Direct oral anticoagulants (DOACs) have become increasingly popular for the prevention and treatment of thromboembolic disorders. However, their use can complicate perioperative management, particularly in patients undergoing surgery. The optimal timing of DOAC cessation before surgery remains unclear, and there is a lack of consensus among guidelines.
A literature search was performed using PubMed and Cochrane Library databases to identify relevant studies and guidelines published from 2010 to 2023. The search terms used were “DOACs,” “direct oral anticoagulants,” “NOACs,” “novel oral anticoagulants,” “surgery,” “perioperative management,” and “duration of holding.” Only English-language articles were included.
Results:
Several studies have investigated the optimal duration of DOAC cessation before surgery. A systematic review and meta-analysis of 11 studies involving 4,596 patients found that a DOAC interruption period of 2-4 days was associated with a lower risk of bleeding compared to shorter or longer interruption periods. Another systematic review and meta-analysis of 23 studies involving 16,834 patients found that a DOAC interruption period of 3-5 days was associated with a lower risk of bleeding compared to shorter or longer interruption periods.
Guidelines from various organizations have also provided recommendations on the duration of holding DOACs before surgery. The American College of Chest Physicians (ACCP) recommends holding DOACs for at least 48 hours before surgery for low-risk procedures and up to 7 days for high-risk procedures. The European Society of Cardiology (ESC) recommends holding DOACs for at least 24 hours before low-risk procedures and up to 4 days for high-risk procedures. The International Society on Thrombosis and Haemostasis (ISTH) recommends holding DOACs for at least 24 hours before low-risk procedures and up to 5 days for high-risk procedures.
Conclusion:
The optimal duration of holding DOACs before surgery remains a topic of debate, with varying recommendations from different guidelines. However, the available evidence suggests that a DOAC interruption period of 2-5 days is associated with a lower risk of bleeding compared to shorter or longer interruption periods. Clinicians should consider the individual patient’s bleeding and thrombotic risk, as well as the type and urgency of the planned procedure when deciding on the appropriate duration of DOAC cessation.
Vitamin K antagonists should be stopped 3 to 5 days before surgery. Preoperative laboratory testing is recommended. Bridging therapy does not decrease the perioperative thromboembolic risk and might increase the perioperative bleeding risk. In patients on direct-acting oral anticoagulants (DOAC), a discontinuation interval of 24 and 48 h in those scheduled for surgery with low and high bleeding risk, respectively, has been shown to be saved. Several guidelines for regional anesthesia recommend a conservative interruption interval of 72 h for DOACs before neuraxial anesthesia. Finally, aspirin is commonly continued in the perioperative period, whereas potent P2Y12 receptor inhibitors (clopidogrel, ticlopidine, ticagrelor, prasugrel, and cangrelor) should be stopped, drug-specifically, 3 to 7 days before surgery.
Bridging Anticoagulation:
Patients who are on long-term anticoagulant therapy and require temporary interruption of their medication for a surgical procedure are at risk of thromboembolic events, such as stroke or deep vein thrombosis. Anticoagulation bridging is a strategy used to manage this risk while minimizing the risk of bleeding complications.
What is Anticoagulation Bridging?
Anticoagulation bridging involves temporarily replacing a patient’s long-term anticoagulant medication with a shorter-acting medication, such as heparin or low molecular weight heparin (LMWH), before and after a surgical procedure. The goal of bridging therapy is to reduce the risk of thromboembolic events while minimizing the risk of bleeding complications.
The decision to use bridging therapy depends on several factors, including the patient’s individual risk of thromboembolism and bleeding, the type of surgery being performed, and the specific anticoagulant medication being used. It is important to carefully weigh the risks and benefits of bridging therapy for each patient on an individual basis.
What Does the Research Say?
Several studies have investigated the use of anticoagulation bridging before surgical procedures. A systematic review and meta-analysis published in the Annals of Internal Medicine in 2013 found that bridging therapy was associated with a higher risk of major bleeding complications, but no significant reduction in thromboembolic events. However, the authors noted that the quality of evidence was low and that further research is needed to determine the optimal use of bridging therapy.
A randomized controlled trial published in the New England Journal of Medicine in 2015 compared bridging therapy with a placebo in patients undergoing elective surgery who were at high risk of thromboembolism. The study found no significant difference in the rate of thromboembolic events between the two groups, but a higher rate of major bleeding complications in the bridging therapy group. The authors concluded that routine bridging therapy may not be necessary in patients at high risk of thromboembolism.
Clinical Guidelines:
Clinical guidelines from professional medical associations provide recommendations for the use of anticoagulation bridging before surgical procedures. The American College of Chest Physicians (ACCP) published guidelines in 2012 that recommend against routine bridging therapy in patients at low risk of thromboembolism and suggest individualized assessment of the risks and benefits of bridging therapy in patients at high risk of thromboembolism.
The American College of Cardiology (ACC) and the American Heart Association (AHA) published guidelines in 2014 that recommend against routine bridging therapy in patients with atrial fibrillation who are undergoing elective surgery.
Conclusion:
Anticoagulation bridging before surgical procedures is a strategy used to manage the risk of thromboembolic events in patients who are on long-term anticoagulant therapy. The decision to use bridging therapy should be based on an individualized assessment of the risks and benefits for each patient. While some studies have suggested that routine bridging therapy may not be necessary for all patients, clinical guidelines provide recommendations for the use of bridging therapy in specific patient populations. Further research is needed to determine the optimal use of anticoagulation bridging before surgical procedures.
There are many patients who are receiving long-term treatment with the blood thinner warfarin, whether because of atrial fibrillation or a mechanical heart valve. Such patients frequently require warfarin to be stopped because of an upcoming surgery/procedure. There is uncertainty about whether such patients should receive bridging anticoagulation before and after the surgery/ procedure.
Bridging anticoagulation refers to giving short-acting blood thinner, usually, low-molecular-weight heparin (LMWH) given by subcutaneous injection for 10 to 12 days around the time of the surgery/procedure when warfarin is interrupted and its anticoagulant effect is outside a therapeutic range. Bridging anticoagulation aims to reduce patients’ risk of developing blood clots, such as stroke, but may also increase patients’ risk of developing potentially serious bleeding complications after surgery.
How Is Bridging Anticoagulation Given?
After warfarin is stopped, 5 to 6 days before surgery (to allow sufficient time for its anticoagulant effect to wane), bridging anticoagulation is started 3 days before surgery, with the last dose given 24 hours before surgery. After surgery, bridging is resumed no earlier than 24 hours after surgery; at the same time, warfarin is restarted. Bridging is continued, typically for 4 to 6 days, until the anticoagulant effect of warfarin has resumed and the blood is sufficiently thinned again.
What Low-Molecular-Weight Heparin and What Dose Should Be Used for Bridging?
There is no standardized bridging drug or dose. A therapeutic-dose regimen, for example, enoxaparin (Lovenox) 1 mg/kg twice daily, is often used in North America, although some physicians in other countries use lower doses.
What Happens for Patients Who Are Having a Minor Procedure, Such as a Tooth Extraction or Skin Cancer Removal?
For patients having minor dental work, such as a tooth extraction or root canal, it may not be necessary to stop warfarin. Some dentists allow patients to continue warfarin (especially if there are concerns about stopping it), so long as they take a special mouthwash called tranexamic acid (Amicar) just before and 3 times daily for 1 to 2 days after the dental procedure, to help prevent bleeding. For patients having minor skin procedures or cataract surgery, interruption of warfarin is often not required because there is minimal bleeding.
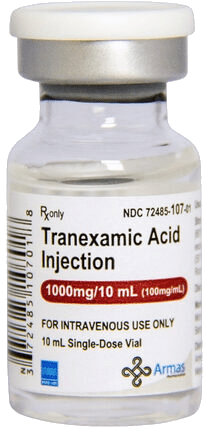
Tranexamic acid injection (1000 mg/10 mL) used intravenously to reduce surgical bleeding and support perioperative hemostasis.
What Happens for Patients Who Are Having a More Extensive Surgery/Procedure in Which the Bleeding Risk Is High?
In such patients (eg, those having hip or knee replacement or cancer surgery), bridging anticoagulation should be given carefully, especially after surgery. Some physicians may choose to delay the resumption of therapeutic-dose low-molecular-weight heparin bridging for 2 to 3 days after surgery, and others may substitute a lower dose of low-molecular-weight heparin after surgery. There is no single approach, but the intent is to prevent bleeding, typically at the surgical/procedure site. If bleeding occurs, it will further delay the resumption of anticoagulation and will expose patients to an increased risk of blood clots. In other words, if bleeding occurs (perhaps because low-molecular-weight heparin bridging was given too close to surgery), this will have the opposite effect of what bridging was meant to do and can harm patients.
Who Should Receive Bridging Anticoagulation?
This is an unanswered question because there are no completed high-quality clinical studies (referred to as randomized, controlled trials) that tell us who should be bridged. In the meantime, clinical experts have suggested a risk classification scheme to help identify which patients may or may not need bridging (Table), but much work needs to be done.
Summary:
Perioperative hemorrhage is an inherent risk of any surgical procedure and requires careful preoperative assessment, intraoperative vigilance, and evidence-based bleeding management. Effective surgical hemostasis depends on coordinated platelet function, activation of the coagulation cascade, fibrin stabilization, and appropriate use of local and systemic hemostatic agents such as tranexamic acid.
At the same time, surgery and trauma significantly increase the risk of venous thromboembolism (VTE), including deep vein thrombosis (DVT) and pulmonary embolism (PE). Balancing bleeding risk with thrombosis prevention is central to modern perioperative care. Evidence from randomized trials demonstrates that appropriate VTE prophylaxis substantially reduces thromboembolic events and mortality without causing a clinically significant increase in major bleeding in most surgical patients.
Thromboprophylaxis should be individualized based on surgical risk, patient comorbidities, thrombophilia status, history of VTE, cancer, immobility, and overall bleeding risk. Options include mechanical methods such as intermittent pneumatic compression devices, pharmacological agents such as low-molecular-weight heparin or direct oral anticoagulants, and combination strategies in selected high-risk patients.
Optimal perioperative management integrates bleeding control, anticoagulation planning, and structured risk stratification to achieve safe and effective surgical outcomes.
Questions and Answers:
What is surgical hemostasis?
Surgical hemostasis is the process of controlling bleeding during and after an operation through platelet activation, coagulation cascade stabilization, fibrin clot formation, and use of mechanical or pharmacological hemostatic techniques.
Why is perioperative bleeding a concern?
Perioperative bleeding increases the risk of transfusion, infection, prolonged hospitalization, reoperation, and mortality. Careful preoperative assessment and intraoperative bleeding control are essential to minimize complications.
What increases the risk of bleeding during surgery?
Bleeding risk may be increased by inherited bleeding disorders such as hemophilia, acquired coagulopathies, liver disease, thrombocytopenia, antithrombotic medications, major trauma, or extensive surgical procedures.
Why are surgical patients at high risk of thrombosis?
Surgery promotes thrombosis through tissue injury, inflammation, endothelial activation, immobilization, and hypercoagulability, significantly increasing the risk of deep vein thrombosis (DVT) and pulmonary embolism (PE).
What is VTE prophylaxis in surgery?
VTE prophylaxis refers to preventive strategies used to reduce the risk of venous thromboembolism in surgical patients, including mechanical methods such as intermittent pneumatic compression and pharmacological agents such as low-molecular-weight heparin or direct oral anticoagulants.
Does thromboprophylaxis increase surgical bleeding?
In most general surgical patients, evidence shows that guideline-directed thromboprophylaxis significantly reduces VTE and mortality without substantially increasing major bleeding risk when appropriately selected.
What role does tranexamic acid play in surgery?
Tranexamic acid is an antifibrinolytic agent that reduces surgical bleeding by inhibiting plasminogen activation and stabilizing fibrin clots, and it is widely used in trauma, orthopedic, cardiac, and obstetric surgery.
How is thromboprophylaxis chosen for a surgical patient?
The choice depends on the type of surgery, patient-specific risk factors, prior VTE, thrombophilia, malignancy, mobility status, and bleeding risk, ensuring an individualized balance between hemorrhage prevention and thrombosis reduction.
References:
Lawson JW, Kitchens CS. Surgery and hemostasis. Curr Opin Hematol. 2015;22(5):420-427. doi:10.1097/MOH.0000000000000172
Kearon C, Hirsh J. Management of anticoagulation before and after elective surgery. N Engl J Med. 1997;336(21):1506-1511.
Kitchens CS. Occult hemophilia. Johns Hopkins Med J. 1980;146(6):255-259.
Kitchens CS, Lawson JW. Surgery and hemostasis. In: Kitchens CS, Kessler CM, Konkle BA, editors. Consultative Hemostasis and Thrombosis. 3rd ed. Philadelphia: Elsevier; 2013. p.651-672.
Bates SM, Greer IA, Middeldorp S, et al. Antithrombotic therapy and prevention of thrombosis: ACCP guidelines. Chest. 2012;141(2 Suppl):691S-736S.
Douketis JD, Spyropoulos AC, Spencer FA, et al. Perioperative management of antithrombotic therapy: ACCP Evidence-Based Clinical Practice Guidelines (9th ed). Chest. 2012;141(2 Suppl):e326S-e350S.
Douketis JD, Spyropoulos AC, Kaatz S, et al. Perioperative bridging anticoagulation in patients with atrial fibrillation. N Engl J Med. 2015;373(9):823-833.
Spyropoulos AC, Al-Badri A, Sherwood MW, Douketis JD. Periprocedural management of patients receiving vitamin K antagonists or direct oral anticoagulants. J Thromb Haemost. 2016;14(5):875-885.
Steffel J, Verhamme P, Potpara TS, et al. 2018 EHRA practical guide on non-vitamin K antagonist oral anticoagulants in atrial fibrillation. Eur Heart J. 2018;39(16):1330-1393.
Kearon C, Akl EA, Ornelas J, et al. Antithrombotic therapy for VTE disease: CHEST guideline and expert panel report. Chest. 2016;149(2):315-352.
Moster M, Bolliger D. Perioperative guidelines on antiplatelet and anticoagulant agents: 2022 update. Curr Anesthesiol Rep. 2022;12:286-296. doi:10.1007/s40140-021-00511-z
Fu JH, McDonald K, Divino CM, Yang J. Optimal timing for resumption of direct oral anticoagulants after elective surgery: systematic review and meta-analysis. JAMA Surg. 2021;156(5):e210682.
Dentali F, Douketis JD, Gianni M, Lim W, Crowther MA. Anticoagulant prophylaxis to prevent symptomatic VTE in hospitalized medical patients: meta-analysis. Ann Intern Med. 2007;146(4):278-288.
Turpie AGG, Bauer KA, Caprini JA, et al. Fondaparinux plus intermittent pneumatic compression vs IPC alone for prevention of VTE after abdominal surgery: randomized comparison. J Thromb Haemost. 2007;5(9):1854-1861.
Hamilton MG, Yee WH, Hull RD, et al. Venous thromboembolism prophylaxis in cranial neurosurgery: systematic review and meta-analysis. Neurosurgery. 2011;68(3):571-581.
Keywords:
surgery and hemostasis, surgical hemostasis, perioperative bleeding management, intraoperative bleeding control, coagulation in surgery, perioperative anticoagulation management, VTE prophylaxis in surgery, thromboprophylaxis surgical patients, venous thromboembolism prevention surgery, deep vein thrombosis prevention after surgery, pulmonary embolism prevention perioperative, intermittent pneumatic compression device surgery, mechanical thromboprophylaxis, pharmacological thromboprophylaxis, low molecular weight heparin surgery, DOAC perioperative management, warfarin bridging anticoagulation, perioperative bridging therapy, antiplatelet management before surgery, direct oral anticoagulants elective surgery, surgical bleeding risk assessment, hemostatic agents in surgery, topical hemostats surgery, antifibrinolytics in surgery, tranexamic acid perioperative use, patient blood management surgery, transfusion avoidance strategies surgery, surgical coagulopathy management, inherited bleeding disorders surgery, hemophilia surgery management, acquired hemophilia perioperative, thrombophilia surgery risk, hypercoagulability after surgery, surgical thrombosis risk factors, balancing bleeding and thrombosis surgery, evidence based VTE guidelines surgery, perioperative hemostasis strategies, surgical complication prevention hemostasis


Request Online Consultation With Dr M Abdou
Fee: US$100
Secure payment via PayPal (credit and debit cards accepted)
Pay Now




Cool blog!
amazing…
I want to be a mbbs
Best wishes!
This article has helped me a lot with some good solutions to my queries.
Thank you!