Miscellaneous Red Cell Abnormalities

Red blood cell morphology showing size variation, hemoglobin distribution, shape abnormalities, and RBC inclusions commonly seen in miscellaneous red cell abnormalities.
Miscellaneous Red Cell Abnormalities: Causes and Overview
This section explores the common miscellaneous red blood cell (RBC) abnormalities and their clinical relevance. Hemoglobin production, a vital function of the bone marrow, is an intricate biochemical process involving the coordinated synthesis of heme and globin chains. Adult human bone marrow synthesizes 4 X 1014 molecules of hemoglobin every second. Heme and globin chains (alpha and beta) in adults are manufactured in separate cell compartments—mitochondria and cytoplasm, respectively—and then combined in the cytoplasm in an amazingly accurate manner.
Four major problems can manifest during this delicate process:
- Qualitative defects of globin chain synthesis result in hemoglobinopathies such as sickle cell disease.
- Quantitative defects of globin chain synthesis result in hemoglobinopathies such as thalassemia.
- Defects in the synthesis of the heme portion result in porphyrias.
- Defects involving incorporation of iron into the heme molecule result in sideroblastic anemias.
Howell-Jolly bodies
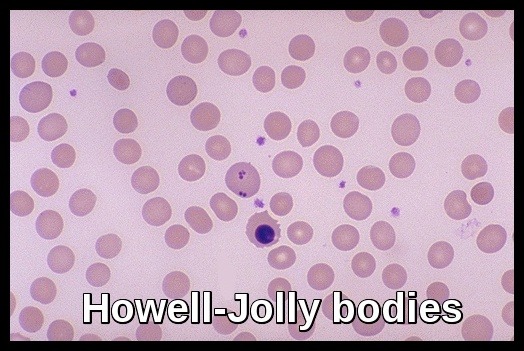
Peripheral blood smear showing Howell-Jolly bodies as small, dark-purple nuclear remnants within erythrocytes, typically associated with hyposplenism.
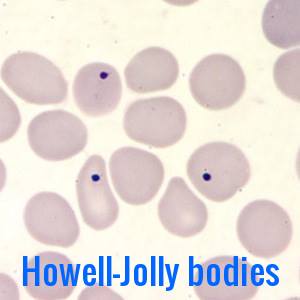
Peripheral smear showing Howell-Jolly bodies, representing retained nuclear fragments due to hyposplenism or splenectomy.
Howell-Jolly bodies are nuclear remnants found in red blood cells (erythrocytes) under various pathological states. They most commonly present in patients with absent or impaired function of the spleen; this is because one of the spleen’s functions is to filter deranged blood cells and remove the intracellular inclusions left by the erythrocyte precursors. William Howell and Justin Jolly in the early 1900s first discovered them in the early 1900s.
Howell-Jolly bodies occur predominantly following splenectomy and in hyposplenic conditions such as coeliac disease. Post-splenectomy blood films show also the following features: target cells, acanthocytes, spherocytes, nucleated red cells, Pappenheimer bodies, and Heinz bodies. The spleen is also responsible for the surface remodeling of red cells.
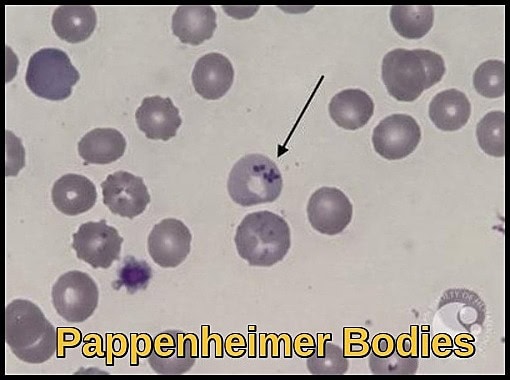
Peripheral blood smear demonstrating Pappenheimer bodies, which represent iron-containing granules seen in sideroblastic anaemia, post-splenectomy states and abnormal iron utilisation disorders.
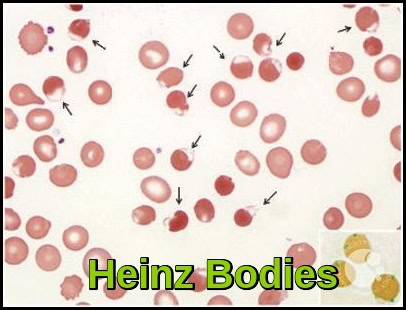
Peripheral smear demonstrating Heinz bodies in rasburicase-induced oxidative hemolytic anaemia in a patient with chronic lymphocytic leukemia.
Following splenectomy, other blood changes including transient leucocytosis and thrombocytosis are commonly seen.
Basophilic Stippling
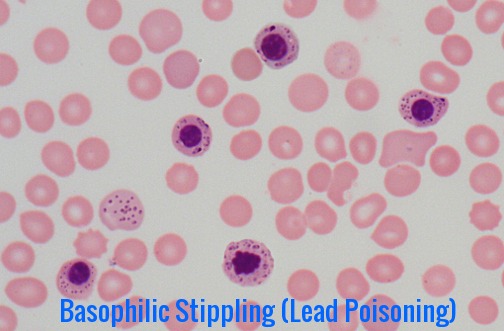
Peripheral blood smear showing coarse basophilic stippling, a characteristic red cell abnormality associated with lead poisoning and disordered erythropoiesis.
Basophilic stippling is one example of several clinically significant miscellaneous red cell abnormalities in which erythrocyte inclusions are identified on peripheral blood smears. The presence of basophilic stippling is attributed to aggregates of ribosomes or fragments of ribosomal RNA precipitated throughout the cytoplasm of circulating erythrocytes. This finding is associated with acquired and heritable hematologic disorders affecting erythropoiesis and erythrocyte maturation.
Basophilic stippling is a frequent manifestation of hematologic disease in the peripheral blood, and it is also observable in bone marrow aspirates. It is implicated in cases of lead poisoning but can be an indicator of various heavy metal toxicities. Alternative causes of basophilic stippling such as hemoglobinopathies, nutritional deficiencies, severe infections, sideroblastic anemia, TTP, and myelodysplasia warrant consideration as well in the context of appropriate clinical history.
Congenital Dyserythropoietic Anemias (CDA)

Bone marrow aspirate demonstrating multinuclear erythroblasts and abnormal erythroid maturation, features typical of congenital dyserythropoietic anaemias.
This is a group of rare miscellaneous red cell abnormalities. Congenital dyserythropoietic anemias belong to a group of inherited conditions characterized by a maturation arrest during erythropoiesis with a reduced reticulocyte production.
CDA is characterized by ineffective erythropoiesis with erythroid cell death within the bone marrow.
Megaloblastoid changes may occur and erythroid multinuclearity is typical.
There are at least three varieties of CDA one of which (CDA II) is associated with lysis of the RBCs by acidified group matched allogeneic sera (Ham’s test).
Sideroblastic Anemias
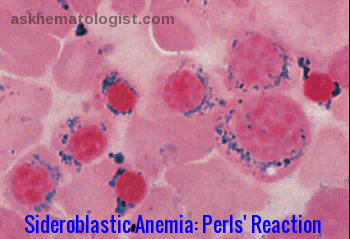
Bone marrow smear using Perls’ Prussian blue stain showing ring sideroblasts, a diagnostic feature of sideroblastic anaemia.
- These are a group of dyserythropoietic disorders in which iron-containing granules (demonstrated by Prussian blue stain – Perls’ reaction) surround the nuclei of some erythroblasts.
- The disorder may be hereditary or acquired.
- The acquired form may be secondary to certain drugs (e.g. antituberculous agents like isoniazid), lead poisoning, other toxic conditions, nutritional deficiencies (copper, vitamin B6), alcohol, as a clonal disorder (myelodysplasia) which may progress to acute leukemia or idiopathic.
- Treatment of sideroblastic anemia may include removal of toxic agents; administration of pyridoxine, thiamine, or folic acid; transfusion (along with antidotes if iron overload develops from transfusion); other medical measures; or bone marrow or liver transplantation.
Alcohol-induced bone marrow damage

Illustration of acute alcohol intoxication as part of alcohol-related disorders, which can contribute to macrocytosis and other red cell abnormalities.
- Excessive alcohol intake can result in dyserythropoiesis as well as morphological changes due to vitamin deficiencies.
- Bone marrow findings took the form of ineffective erythropoiesis associated with impaired iron utilization, vacuolated proerythroblasts, multinuclear erythroblasts, megaloblasts, and iron-containing plasma cells as well as vacuolated precursor cells of the granulocytopoietic series.
- In the differential diagnosis, alcohol-induced bone marrow damage is to be distinguished from the myelodysplastic syndrome of the RA and RARS form.
- Alcohol-induced bone marrow damage is reversible. The toxic defect probably does not reside in the stem cell but is more peripheral.
Questions and Answers:
What are miscellaneous red cell abnormalities?
These are non-specific but clinically useful red blood cell changes seen on a peripheral smear, including inclusions, shape abnormalities, size variation and marrow-related defects. They often reflect underlying disorders such as hyposplenism, dyserythropoiesis, iron metabolism defects or toxic exposures.
What do Howell-Jolly bodies indicate?
Howell-Jolly bodies are nuclear remnants caused by impaired splenic function. They are typically seen after splenectomy or in functional hyposplenism such as sickle cell disease, celiac disease and immune deficiency states.
What causes basophilic stippling?
Basophilic stippling results from abnormal ribosomal aggregation and is commonly associated with lead poisoning, sideroblastic anaemia, thalassemias, alcoholic liver disease and defective haem synthesis.
What are ring sideroblasts?
Ring sideroblasts are erythroblasts with iron-loaded mitochondria forming a perinuclear ring on Perls’ Prussian blue stain. They are characteristic of sideroblastic anaemia, myelodysplastic syndromes and alcohol-related marrow toxicity.
How does alcohol misuse affect red blood cells?
Excessive alcohol intake can cause macrocytosis, sideroblastic changes, vacuolated erythroid precursors, and impaired haem synthesis. Alcohol suppresses bone marrow and interferes with pyridoxine metabolism.
What hematologic abnormalities are seen in congenital dyserythropoietic anaemias (CDA)?
CDA is characterised by ineffective erythropoiesis with multinuclear erythroblasts, abnormal nuclear morphology and dysplastic erythroid maturation. Depending on the subtype, macrocytosis, haemolysis and iron overload may also be present.
What conditions cause red cell inclusions on a blood film?
Red cell inclusions may result from hyposplenism (Howell-Jolly bodies), lead toxicity (basophilic stippling), unstable hemoglobins (Heinz bodies), iron-loading disorders (Pappenheimer bodies) and megaloblastic or dyserythropoietic processes.
Why is red cell morphology important in diagnosis?
Peripheral smear examination can reveal early indicators of systemic disease, including marrow failure, nutritional deficiencies, toxins, haemolytic processes, and inherited RBC disorders. Morphology often guides further laboratory investigation.
What is the significance of target cells, ovalocytes, and teardrop cells?
Target cells suggest liver disease, thalassemias and post-splenectomy states; ovalocytes may indicate iron deficiency or hereditary elliptocytosis; teardrop cells raise concern for marrow infiltration or myelofibrosis.
When should abnormal RBC morphology prompt further testing?
Persistent abnormalities, unusual inclusions, macrocytosis without B12/folate deficiency, suspected lead exposure, or dysplastic features should trigger bone marrow evaluation, iron studies, haematinics, and possibly genetic testing depending on the pattern.
References:
Jones, K. W. (2010). Evaluation of Cell Morphology and Introduction to Platelet and White Blood Cell Morphology. Semantic Scholar.
Scafidi, J. M., & Gupta, V. (2020). Histology, Howell–Jolly Bodies. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing. Updated May 11, 2020. Available from: NCBI Bookshelf.
Sears, M. D., & Udden, M. D. (2012). Howell–Jolly Bodies: A Brief Historical Review. The American Journal of the Medical Sciences, 343(5), 407–409.
Cheson, B. D., Rom, W. N., & Webber, R. C. (1984). Basophilic Stippling of Red Blood Cells: A Nonspecific Finding of Multiple Etiology. American Journal of Industrial Medicine, 5(4), 327–334.
Sanchez, J. R., & Lynch, D. T. (2020). Histology, Basophilic Stippling. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing. Updated July 3, 2020. Available from: NCBI Bookshelf.
Denecke, J., & Marquardt, T. (2009). Congenital Dyserythropoietic Anemia Type II (CDAII/HEMPAS): Where Are We Now? Biochimica et Biophysica Acta – Molecular Basis of Disease, 1792(9), 915–920.
Iolascon, A., Esposito, M. R., & Russo, R. (2012). Clinical Aspects and Pathogenesis of Congenital Dyserythropoietic Anemias: From Morphology to Molecular Approach. Haematologica, 97(12), 1786–1794.
McLintock, L. A., & Fitzsimons, E. J. (2002). Erythroblast Iron Metabolism in Sideroblastic and Sideropenic States. Hematology, 7, 189–195.
Girard, D. E., Kumar, K. L., & McAfee, J. H. (1987). Hematologic Effects of Acute and Chronic Alcohol Abuse. Hematology/Oncology Clinics of North America, 1, 321–334.
Venkataraman, G. (2017, April 20). Pappenheimer Bodies [Image #00061019]. American Society of Hematology Image Bank. Retrieved from https://imagebank.hematology.org/image/61019/pappenheimer-bodies
Hrisinko, M. A., & Chen, Y.-H. (2016, May 2). Rasburicase-Induced Heinz Body Hemolytic Anemia in a Patient with Chronic Lymphocytic Leukemia [Image #00060725]. ASH Image Bank. Retrieved from https://imagebank.hematology.org/image/60725/rasburicaseinduced-heinz-body-hemolytic-anemia-in-a-patient-with-chronic-lymphocytic-leukemia
Keywords:
red cell abnormalities, abnormal red blood cells, red blood cell morphology, RBC morphology, abnormal erythrocytes, RBC inclusions, red cell inclusions, RBC inclusion bodies, blood smear abnormalities, peripheral blood smear findings, red cell shape abnormalities, poikilocytosis, anisocytosis, microcytes, macrocytes, spherocytes, schistocytes, target cells, elliptocytes, ovalocytes, stomatocytes, teardrop cells, sickle cells, acanthocytes, burr cells, echinocytes, Howell-Jolly bodies, Heinz bodies, basophilic stippling, coarse basophilic stippling, fine basophilic stippling, Cabot rings, Pappenheimer bodies, sideroblasts, ring sideroblasts, sideroblastic anemia, Perls stain, dyserythropoiesis, congenital dyserythropoietic anemias, CDA, alcohol-related macrocytosis, alcohol-induced marrow changes, RBC agglutination, rouleaux formation, hemolytic anemia, oxidative hemolysis, rasburicase-induced hemolysis, lead poisoning smear findings, iron metabolism disorders, ineffective erythropoiesis, abnormal RBC maturation, red cell morphology interpretation


Request Online Consultation With Dr M Abdou
Fee: US$100
Secure payment via PayPal (credit and debit cards accepted)
Pay Now



Amazing pictures & information. Thank you for sharing.