Pyruvate Kinase Deficiency
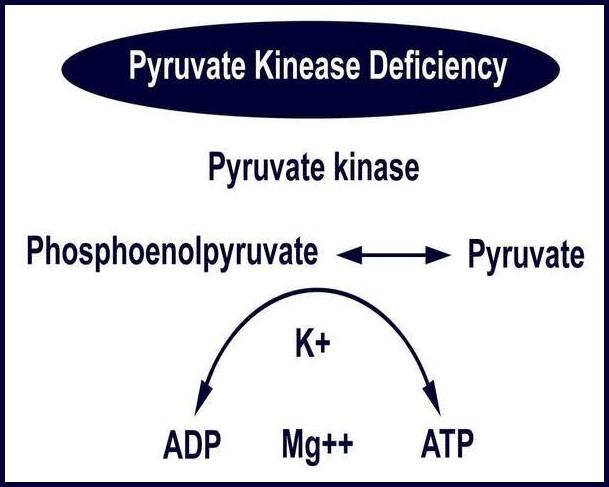
Metabolic mechanism of pyruvate kinase deficiency demonstrating impaired ATP generation in red blood cells leading to hemolytic anemia
Pyruvate kinase deficiency (PKD) is one of the most common inherited enzymatic disorders of red blood cells and a well-recognized cause of chronic non-spherocytic hemolytic anemia. The clinical severity of PKD varies widely, ranging from fully compensated mild anemia to severe, transfusion-dependent hemolysis. Among red cell enzyme deficiencies that cause hemolytic anemia, PKD is the second most frequent worldwide, following glucose-6-phosphate dehydrogenase (G6PD) deficiency.
PKD is inherited in an autosomal recessive manner and affects individuals across all ethnic groups. However, increased prevalence has been reported in certain genetically isolated populations, particularly the Amish community, due to founder effects. Both males and females are equally affected, and clinical expression depends on the specific genetic mutations present.
The disorder is characterized by reduced activity of the enzyme pyruvate kinase, which plays a critical role in maintaining adenosine triphosphate (ATP) production within red blood cells. In the absence of adequate pyruvate kinase activity, erythrocytes are unable to maintain membrane integrity and are prematurely destroyed in the circulation and spleen, leading to chronic hemolytic anemia.
In severe cases, the neonatal and early childhood history may include profound anemia, marked neonatal jaundice, kernicterus, and the need for exchange transfusion. These early life complications reflect the intensity of hemolysis that can occur in untreated or severe forms of pyruvate kinase deficiency.
Clinical features:
The clinical presentation of pyruvate kinase deficiency (PKD) is highly variable and depends on the degree of enzyme deficiency and ongoing hemolysis. Patients commonly present with jaundice, splenomegaly, and chronic hemolytic anemia of mild to severe intensity. The anemia is typically normocytic or macrocytic. In severe forms, manifestations may begin in utero, leading to hydrops fetalis. Newborns frequently present with prolonged neonatal jaundice and anemia, sometimes requiring exchange transfusion. In childhood, affected individuals may appear pale due to anemia and develop intermittent jaundice. Mild cases may remain undiagnosed until adulthood. Although anemia often stabilizes later in life, acute hemolytic episodes may be triggered by infection, physiological stress, or pregnancy. Pyruvate kinase deficiency results from pathogenic variants in the PKLR gene and follows an autosomal recessive pattern of inheritance.
- Reticulocytosis reflecting chronic compensatory erythropoiesis in pyruvate kinase deficiency
- Growth delay and failure to thrive due to longstanding hemolytic anemia
- Gallbladder (pigment) stones, typically after the first decade of life but may occur in childhood
- Frontal bossing secondary to chronic bone marrow hyperplasia
- Neonatal hyperbilirubinemia in affected newborns
- Persistent or recurrent jaundice due to ongoing hemolysis
- Raised serum lactate dehydrogenase (LDH) as a marker of red cell destruction
- Low or undetectable serum haptoglobin levels
- Mild to moderate splenomegaly from extravascular hemolysis
- Right upper-quadrant abdominal tenderness, often related to hepatobiliary involvement
- Chronic leg ulcers in adults with longstanding severe hemolysis
Diagnosis:
- Full blood count (FBC) showing chronic hemolytic anemia that is typically normocytic or macrocytic
- Differential blood count to assess associated leukocyte and platelet abnormalities
- Reticulocyte count, which is typically elevated due to compensatory marrow response
- Serum bilirubin (predominantly unconjugated) reflecting ongoing red cell destruction
- Direct Coombs (direct antiglobulin) test to exclude immune-mediated hemolytic anemia
- Peripheral blood film examination showing polychromasia, anisopoikilocytosis, and erythrocyte morphological changes related to hemolysis
- Bone marrow examination demonstrating marked normoblastic erythroid hyperplasia due to chronic hemolytic stress
- Evidence of extramedullary hematopoiesis on imaging in longstanding or severe disease
- Red cell pyruvate kinase enzyme activity assay, which is typically reduced to approximately 5–25% of normal in most patients with pyruvate kinase deficiency
- Molecular genetic testing of the PKLR gene for definitive confirmation, family screening, and prenatal diagnosis

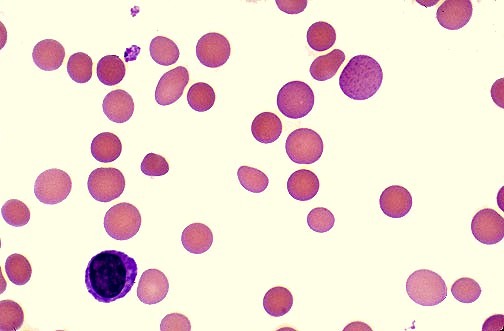
Peripheral blood film in pyruvate kinase deficiency demonstrating significant red cell shape variation following splenectomy.
Treatment:
Management of pyruvate kinase deficiency (PKD) is individualized and depends on disease severity. In patients with mild to moderate PKD, treatment is predominantly supportive, with regular monitoring for anemia, hemolysis, and long-term complications.
Many clinicians recommend daily oral folic acid supplementation to support increased erythropoiesis, as folate is essential for effective bone marrow red blood cell production.
Moderate to severe anemia is often managed with red blood cell transfusions. Some patients require regular transfusions every 3–4 weeks, whereas others need transfusion support only intermittently during acute illness, physiological stress, surgery, or pregnancy. A proportion of patients remain transfusion-independent throughout life.
In neonates with severe hyperbilirubinemia, exchange transfusion may be lifesaving. This procedure rapidly removes bilirubin-rich blood and replaces it with donor blood, effectively preventing bilirubin-induced neurological damage (kernicterus).
Symptomatic pigment gallstones secondary to chronic hemolysis are managed surgically with cholecystectomy. Splenectomy may be considered in selected patients with severe hemolytic anemia and high transfusion requirements, as it can significantly reduce hemolysis and transfusion burden. However, response to splenectomy is variable, and the long-term risks of post-splenectomy sepsis and thrombosis must be carefully weighed.
In recent years, disease-modifying therapy with the oral pyruvate kinase activator mitapivat has emerged as an important treatment option in selected adult patients with PKD, leading to improved hemoglobin levels and reduced hemolysis in many cases.
Prognosis:
The prognosis of pyruvate kinase deficiency (PKD) is highly variable and largely depends on the degree of enzyme deficiency, severity of hemolysis, transfusion requirements, and development of long-term complications. Many patients with mild PKD maintain stable hemoglobin levels and have a near-normal life expectancy with supportive care alone. Individuals with moderate to severe disease may experience chronic anemia, transfusion dependence, and iron overload, which can affect long-term outcomes if not adequately managed.
Neonatal presentation with severe hyperbilirubinemia, kernicterus, or hydrops fetalis is associated with increased early morbidity and mortality. In childhood and adulthood, the prognosis improves significantly with appropriate transfusion support, folic acid supplementation, iron chelation when indicated, and timely surgical interventions such as splenectomy in selected cases. The introduction of disease-modifying therapy with mitapivat has further improved hemoglobin levels and quality of life in many adult patients with non–transfusion-dependent and transfusion-dependent PKD.
With modern supportive and targeted therapies, most patients with pyruvate kinase deficiency now survive into adulthood with good functional outcomes and improved quality of life.
Questions and Answers:
What is pyruvate kinase deficiency?
Pyruvate kinase deficiency is a rare inherited red blood cell enzyme disorder that causes chronic non-spherocytic hemolytic anemia due to impaired ATP production within erythrocytes, leading to premature red cell destruction.
Is pyruvate kinase deficiency inherited?
Yes, pyruvate kinase deficiency is inherited in an autosomal recessive manner, meaning both parents must carry a pathogenic PKLR gene mutation for the condition to appear in the child.
What are the main symptoms of pyruvate kinase deficiency?
The most common symptoms include chronic anemia, jaundice, splenomegaly, fatigue, gallstones, neonatal hyperbilirubinemia, and in severe cases hydrops fetalis.
Can pyruvate kinase deficiency cause severe jaundice in newborns?
Yes, severe neonatal jaundice is a well-recognized presentation of pyruvate kinase deficiency and may progress to kernicterus if not treated urgently with phototherapy or exchange transfusion.
How is pyruvate kinase deficiency diagnosed?
Diagnosis is based on laboratory evidence of hemolysis, reduced pyruvate kinase enzyme activity, exclusion of immune hemolysis, and confirmation with PKLR gene mutation testing.
What does the blood film show in pyruvate kinase deficiency?
The peripheral blood film typically shows anisopoikilocytosis, polychromasia, reticulocytosis, and post-splenectomy changes if splenectomy has been performed.
Can pyruvate kinase deficiency be mild?
Yes, some patients have mild disease with near-normal hemoglobin levels and may remain undiagnosed until adulthood, especially if hemolysis is well compensated.
Does pyruvate kinase deficiency require regular blood transfusions?
Not all patients require transfusions, but moderate to severe cases often need regular or intermittent transfusion support, especially during infections, surgery, or pregnancy.
What is the role of splenectomy in pyruvate kinase deficiency?
Splenectomy can significantly reduce hemolysis and transfusion requirements in selected patients, but it carries long-term risks such as infection and thrombosis.
Can pyruvate kinase deficiency cause gallstones?
Yes, chronic hemolysis leads to excess bilirubin production, predisposing patients to pigment gallstones, often requiring cholecystectomy.
What is the prognosis of pyruvate kinase deficiency?
The prognosis is highly variable, but with modern supportive care, iron chelation, and targeted therapy, most patients now reach adulthood with good quality of life.
Is there a targeted drug treatment for pyruvate kinase deficiency?
Yes, mitapivat is an oral pyruvate kinase activator that has been shown to improve hemoglobin levels and reduce hemolysis in many adult patients.
Can pyruvate kinase deficiency affect pregnancy?
Yes, pregnancy may worsen anemia and increase transfusion requirements, and careful multidisciplinary monitoring is essential.
Does pyruvate kinase deficiency cause iron overload?
Iron overload can occur due to repeated blood transfusions or increased intestinal iron absorption and may require chelation therapy.
Is pyruvate kinase deficiency the same as G6PD deficiency?
No, both are red cell enzyme disorders causing hemolytic anemia, but they involve different enzymes, inheritance patterns, triggers, and clinical behavior.
References:
Yaish HM. Pyruvate Kinase Deficiency: Practice Essentials, Background, Pathophysiology. Medscape. Updated regularly. Accessed April 2014.
Abdel Fattah M, Abdel Ghany E, Adel A, Mosallam D, Kamal S. Glucose-6-phosphate dehydrogenase and red cell pyruvate kinase deficiency in neonatal jaundice cases in Egypt. Pediatric Hematology and Oncology. 2010;27(4):262–271.
Haldeman-Englert C. Pyruvate kinase deficiency. MedlinePlus Medical Encyclopedia. U.S. National Library of Medicine. Updated February 3, 2014.
National Organization for Rare Disorders (NORD). Pyruvate Kinase Deficiency. Rare Disease Database. 2005. Accessed July 18, 2011.
Grace RF, Zanella A, Neufeld EJ, Morton DH, Eber S, Yaish H, Glader B. Erythrocyte pyruvate kinase deficiency: 2015 status report. American Journal of Hematology. 2015;90(9):825–830.
Zanella A, Fermo E, Bianchi P, Chiarelli LR, Valentini G. Pyruvate kinase deficiency: genotype–phenotype association. Blood Reviews. 2007.
Yates A. Pyruvate Kinase Deficiency Overview: A Rare Cause of Anemia. Verywell Health.
Agios Pharmaceuticals. FDA approval of Pyrukynd (mitapivat) for pyruvate kinase deficiency. U.S. Food and Drug Administration. February 2022.
Grace RF, et al. Mitapivat versus placebo for pyruvate kinase deficiency. A randomized, double-blind Phase III trial. New England Journal of Medicine. 2022.
Keywords:
pyruvate kinase deficiency, PKD anemia, pyruvate kinase deficiency symptoms, pyruvate kinase deficiency diagnosis, pyruvate kinase deficiency treatment, PKLR gene mutation, autosomal recessive hemolytic anemia, non spherocytic hemolytic anemia, neonatal jaundice PKD, kernicterus pyruvate kinase deficiency, hydrops fetalis hemolytic anemia, splenectomy in pyruvate kinase deficiency, mitapivat Pyrukynd, mitapivat treatment PKD, targeted therapy pyruvate kinase deficiency, chronic hemolytic anemia causes, reticulocytosis hemolytic anemia, gallstones in hemolytic anemia, iron overload transfusion anemia, pyruvate kinase deficiency blood film, enzyme deficiency anemia, rare inherited anemia, pediatric hemolytic anemia, adult pyruvate kinase deficiency, exchange transfusion neonatal jaundice


Request Online Consultation With Dr M Abdou
Fee: US$100
Secure payment via PayPal (credit and debit cards accepted)
Pay Now



What does NAS mean in a blood test? What is NAS?
Hi Clo,
The abbreviation is not familiar to me but it could be for Sodium (Na) serum level or if for an infant it could stand for Neonatal Abstinence Syndrome.
BW,