Mycosis Fungoides
Mycosis fungoides (MF) and Sézary cell leukemia (SCL) represent closely related forms of cutaneous T-cell lymphoma, characterised by malignant CD4⁺ T-cell proliferation that primarily involves the skin and, in advanced stages, may progress to leukemic dissemination.
Clinical Features:
Mycosis Fungoides is an uncommon chronic T-cell lymphoma primarily affecting the skin and occasionally the internal organs. The disease is most common in middle-aged men.
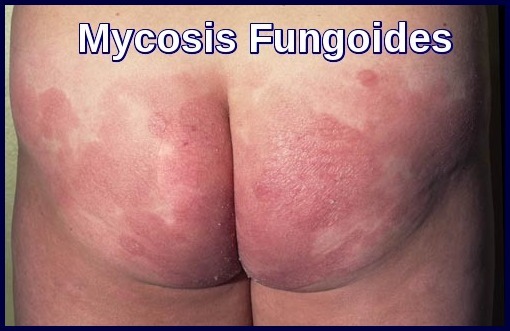
There are eczematous lesions, plaques, tumors, and ulcers of the skin without overt blood involvement. In the late stages, the whole body may appear red.
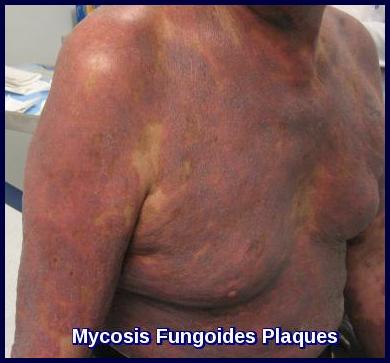
Advanced Mycosis Fungoides presenting with hyperpigmented, infiltrated plaques across the upper chest and arm.
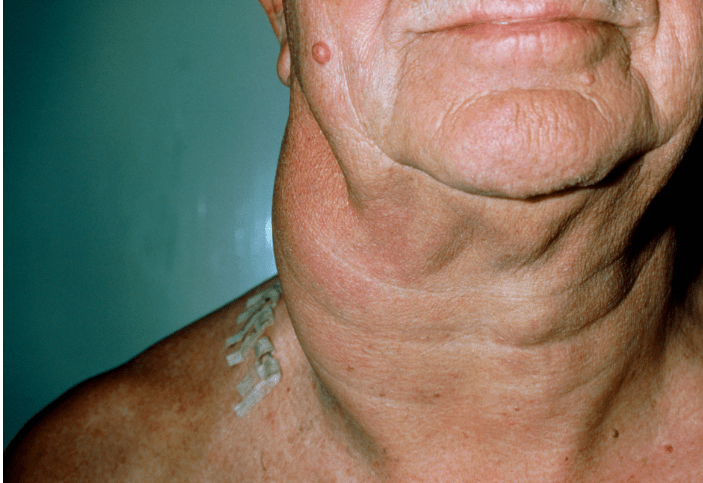
Lymphadenopathy is common and a leukemic element appears (Sezary Cell Leukemia); a diffuse pruritic skin rash and hepatosplenomegaly are then common. Systemic involvement of lymph nodes, liver, spleen, and lungs result in the advent of symptoms, which include fever, night sweats, and unintentional weight loss.

Sézary syndrome presenting with diffuse erythroderma (l’homme rouge), a severe leukemic variant related to Mycosis Fungoides.
Infections frequently occur.
It must be noted that some cases originate as Sezary Cell Leukemia.
Investigations:
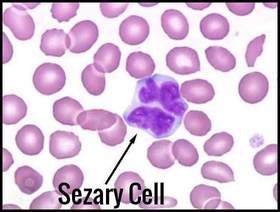
Typical Sézary cell with cerebriform nucleus seen in advanced Sézary syndrome, a leukemic variant associated with Mycosis Fungoides.
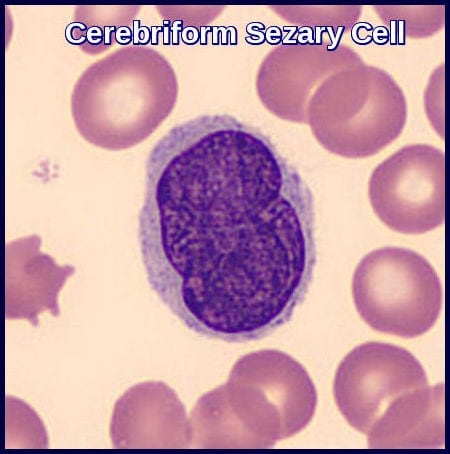
Cerebriform Sézary cell demonstrating the characteristic convoluted nucleus seen in Sézary syndrome linked to Mycosis Fungoides.
In Sezary Cell Leukemia there is a lymphocytosis which may be over 200X109/L. These cells are typically large mononuclear cells with bizarre folded grooved nuclei with little cytoplasm.
Moderate eosinophilia is common in both Mycosis Fungoides and Sezary Cell Leukemia.
In Sezary Cell Leukemia there is a moderate marrow infiltration with the malignant lymphocytes relative to the blood count.
Skin biopsy in Mycosis Fungoides and Sezary Cell Leukemia shows infiltration of the dermis by the malignant cells with the formation of so-called Pautrier’s microabscesses. Histology may be equivocal early in the course because of insufficient quantities of lymphoma cells. The malignant cells are mature T cells (T4+, T11+, T12+).
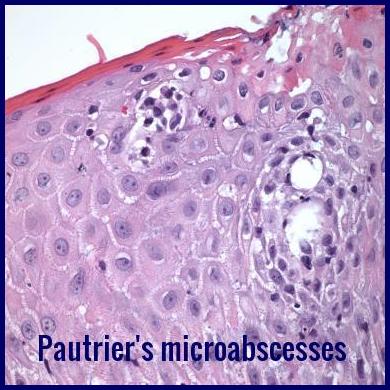
Pautrier microabscesses with intraepidermal atypical lymphocytes, a classic histologic feature of Mycosis Fungoides.
For staging, bone marrow biopsy and CT of chest, abdomen, and pelvis.
PET scan may also be used for suspected visceral involvement.
Treatment:
Mycosis Fungoides is treated with topical steroids, ultraviolet light and psoralens, electron beam radiation therapy or topical nitrogen mustard. Electron beam radiation therapy, in which most of the energy is absorbed in the first 5 to 10 mm of tissue, and topical nitrogen mustard have proved highly effective. Systemic treatment is primarily used when other therapies have failed, after relapse, or in patients with documented extranodal or extracutaneous disease. Systemic therapy is also given for Sezary Cell Leukemia. Various lymphoma-type regimens are used.
The lack of consistent efficacy from previously used therapies may arise in part from the heterogeneity of cutaneous T-cell lymphoma (CTCL), which encompasses a wide range of lymphomas such as mycosis fungoides (MF), primary cutaneous CD30+ lymphoproliferative disorders, and Sézary syndrome (SS).
ALCANZA was the first reported Phase 3 trial in CD30+ CTCL investigating the use of a systemic agent (brentuximab vedotin) against physician’s choice, after one prior systemic therapy. Significantly more patients achieved durable response with brentuximab vedotin as measured by ORR4 (objective response rate lasting at least 4 months), vs physician’s choice of either methotrexate or bexarotene. Brentuximab vedotin improved outcomes across all key secondary endpoints and significantly reduced patient symptom burden compared with physician’s choice. Brentuximab vedotin improved patient-reported quality of life outcomes and was associated with a manageable tolerability profile.
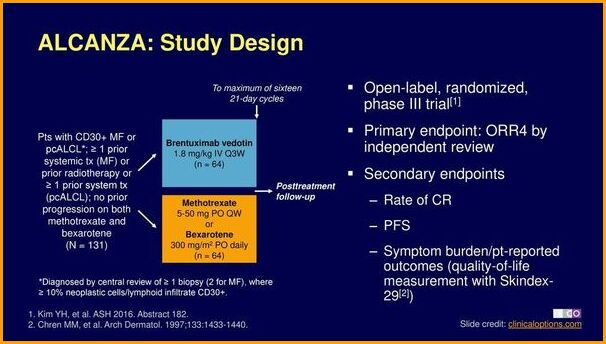
ALCANZA phase III trial evaluating brentuximab vedotin versus standard therapies in CD30-positive Mycosis Fungoides and primary cutaneous ALCL.

Brentuximab vedotin (Adcetris), an anti-CD30 antibody–drug conjugate approved for relapsed or refractory Mycosis Fungoides.
Brentuximab targets a protein called CD 30 that is found also on Hodgkin lymphoma and anaplastic large cell lymphoma cells. Brentuximab sticks to the CD30 protein and delivers a drug to the cell. The drug then kills the cell. Brentuximab Vedotin may cost over $100,000 for a course of treatment, or $4,500 per vial.
Prognosis:
Median survival in Mycosis Fungoides is 7 to 10 years after diagnosis. However, survival rates vary markedly depending on the stage at diagnosis.
The prognosis is worse in Sezary Cell Leukemia.
Summary:
Mycosis Fungoides is the most common form of cutaneous T-cell lymphoma, presenting with slowly progressive patches, plaques, and tumours that may evolve into Sézary syndrome, the leukemic variant characterised by erythroderma and circulating cerebriform lymphocytes. Diagnosis relies on clinicopathologic correlation, including skin biopsy, immunophenotyping, and detection of Pautrier microabscesses and malignant CD4⁺ T-cell clones. Staging incorporates skin involvement, lymph nodes, blood tumour burden, and systemic spread. Management is stage-adapted, ranging from topical steroids, phototherapy, and retinoids in early disease to radiotherapy, systemic therapies, brentuximab vedotin, and extracorporeal photopheresis in advanced or refractory cases. Early recognition and multidisciplinary evaluation are essential to improve outcomes and quality of life.
Questions and Answers:
What are the early skin signs of Mycosis Fungoides?
Early Mycosis Fungoides typically presents with slowly evolving patches of erythema, scaling, and pruritus on sun-protected areas such as the buttocks and flanks, often mimicking eczema or psoriasis.
How is Mycosis Fungoides diagnosed?
Diagnosis requires clinicopathologic correlation, including skin biopsies showing atypical epidermotropic T cells, Pautrier microabscesses, immunophenotyping, and T-cell receptor clonality testing.
What is the difference between Mycosis Fungoides and Sézary syndrome?
Mycosis Fungoides primarily affects the skin, whereas Sézary syndrome is its leukemic variant characterised by erythroderma, lymphadenopathy, and circulating cerebriform Sézary cells.
What do Sézary cells look like under the microscope?
Sézary cells show a distinctive cerebriform nucleus with deep convolutions on peripheral blood smear and characteristically display an aberrant CD4 positive immunophenotype with loss of pan–T-cell markers such as CD7 and CD26.
What are Pautrier microabscesses?
Pautrier microabscesses are clusters of atypical lymphocytes within the epidermis and represent a classic histopathological feature of Mycosis Fungoides.
How is Mycosis Fungoides staged?
Staging is based on the TNMB system, assessing skin surface involvement, lymph node disease, visceral spread, and blood tumour burden, including presence of Sézary cells.
Is Mycosis Fungoides curable?
Mycosis Fungoides is generally chronic and incurable but highly treatable, with many patients achieving long-term control through skin-directed and systemic therapies.
What are the treatment options for early-stage Mycosis Fungoides?
Early-stage disease is commonly treated with topical corticosteroids, phototherapy (NB-UVB or PUVA), topical retinoids, and localized radiotherapy.
How is advanced or refractory Mycosis Fungoides treated?
Advanced disease may require systemic therapies such as interferon-α, bexarotene, methotrexate, histone deacetylase inhibitors, extracorporeal photopheresis, or brentuximab vedotin for CD30 positive cases.
What is brentuximab vedotin used for in Mycosis Fungoides?
Brentuximab vedotin (Adcetris) is an anti-CD30 antibody-drug conjugate approved for CD30 positive Mycosis Fungoides and CTCL, supported by the phase III ALCANZA trial.
Does Mycosis Fungoides spread to internal organs?
In later stages, MF may involve lymph nodes, blood, liver, spleen, or lungs, particularly when progressing to tumour-stage disease or Sézary syndrome.
Can Mycosis Fungoides cause erythroderma?
Erythroderma is uncommon in early MF but is a hallmark of Sézary syndrome, presenting with diffuse redness, scaling, and severe pruritus.
Are blood tests useful for Mycosis Fungoides?
Yes. Flow cytometry, CD4:CD8 ratio assessment, Sézary cell counts, and TCR clonality studies help evaluate blood involvement, especially in suspected Sézary syndrome.
What triggers Mycosis Fungoides to progress?
Progression is usually related to biological behaviour rather than lifestyle factors, influenced by malignant T-cell clonality, host immune response, and tumour microenvironment.
Does Mycosis Fungoides shorten life expectancy?
Early-stage MF often has a near-normal life expectancy, whereas tumour-stage disease or Sézary syndrome carries a higher risk and requires more intensive monitoring and therapy.
References:
Willemze R, Jaffe ES, Burg G, et al. WHO-EORTC classification for cutaneous lymphomas. Blood. 2005 May 15;105(10):3768–85.
Vonderheid EC, Bernengo MG, Burg G, et al. Update on erythrodermic cutaneous T-cell lymphoma: report of the International Society for Cutaneous Lymphomas. J Am Acad Dermatol. 2002 Jan;46(1):95–106.
Olsen E, Vonderheid E, Pimpinelli N, et al. Revisions to the staging and classification of Mycosis Fungoides and Sézary syndrome: ISCL/EORTC proposal. Blood. 2007 Sep 15;110(6):1713–22.
Prince HM, Kim YH, Horwitz SM, et al. Brentuximab vedotin vs physician’s choice in CD30-positive cutaneous T-cell lymphoma (ALCANZA trial). Lancet. 2017;390:555–66.
Hughes CF, Khot A, McCormack C, et al. Lack of durable disease control with chemotherapy for Mycosis Fungoides and Sézary syndrome: comparative outcome analysis. Blood. 2015;125:71–81.
Cutaneous T-Cell Lymphoma – ALCANZA Trial Overview. Medscape.
Available at: https://www.medscape.com/infosites/259045.1/isarticle-3
ADCETRIS (Brentuximab Vedotin) – Cost, side effects, and prescribing information.
Available at: http://cancerworld.info/adcetris-brentuximab-vedotin-cost-side-effects-package-insert/
Keywords:
mycosis fungoides, mycosis fungoides rash, mycosis fungoides symptoms, mycosis fungoides treatment, mycosis fungoides stages, mycosis fungoides pictures, sezary syndrome, mycosis fungoides vs sezary syndrome, cutaneous t-cell lymphoma, pautrier microabscesses, sezary cells, erythroderma, brentuximab vedotin, adcetris, alcanza trial, cutaneous lymphoma diagnosis, ctcl management, ctcl prognosis, t-cell lymphoma skin


Request Online Consultation With Dr M Abdou
Fee: US$100
Secure payment via PayPal (credit and debit cards accepted)
Pay Now