Stem Cell Transplantation
Stem cell transplantation, also known as hematopoietic stem cell transplantation (HSCT), is a crucial therapeutic approach in hematology and oncology. It involves the infusion of healthy stem cells into a patient to replace damaged or malfunctioning cells.
Stem cells are a class of undifferentiated cells that are able to differentiate into specialised cell types. Commonly, stem cells come from two main sources:
Embryos formed during the blastocyst phase of embryological development (embryonic stem cells) and adult tissue (adult stem cells).
Both types are generally characterised by their potency, or potential to differentiate into different cell types (such as skin, muscle, bone, etc.).
The first successful bone marrow transplant between a related donor and recipient is performed in 1956 by Dr E Donnall Thomas in New York. The patient, who has leukaemia, is given radiotherapy and then treated with healthy bone marrow from an identical twin.
In 1958, the French oncologist and immunologist Dr Georges Mathé performs a human bone marrow transplant using non-related bone marrow on six Yugoslav engineers who were irradiated at different levels after a nuclear reactor incident. Following the transplant, Mathé defined the ‘graft versus host’ disease in his patients and was the first to study the debilitating and wasting condition that follows transplantation. He deduced that this must be due to an immune reaction of the cells in the donor marrow against the cells in the human patient.
Stem cells may be harvested from bone marrow, peripheral blood, umbilical cord blood.
Peripheral blood has largely replaced bone marrow as a source of stem cells, especially in autologous stem cell transplantation, because stem cell harvest is easier and neutrophil and platelet counts recover faster.
Umbilical cord stem cell transplantation has been restricted mainly to children because there are too few stem cells in umbilical cord blood for an adult.
A potential future source of stem cells is induced pluripotent stem cells (certain cells taken from adults and reprogrammed to act like stem cells).
Allogeneic Hematopoietic stem cell transplantation (HSCT) is limited mainly by lack of histocompatible donors. An HLA-identical sibling donor is ideal, followed by an HLA-matched sibling donor. Because only one fourth of patients have such a sibling donor, mismatched related or matched unrelated donors (identified through international registries) are often used. However, long-term disease-free survival rates may be lower than those with HLA-identical sibling donors.
The technique for umbilical cord HSCT is still being defined, but HLA-matching is probably unimportant.
The basis for stem cell transplantation is that blood cells (red cells, white cells and platelets) and immune cells (lymphocytes) arise from the stem cells, which are present in marrow, peripheral blood and cord blood. Intense chemotherapy or radiation therapy kills the patient’s stem cells. This stops the stem cells from making enough blood and immune cells.
Stem cell transplantation allows doctors to give large doses of chemotherapy or radiation therapy to increase the chance of eliminating blood cancer in the marrow and then restoring normal blood cell production.
The patient receives high-dose chemotherapy and/or radiation therapy, followed by the intravenous (IV) infusion of autologous stem cells (stem cells come from patient’s own body) or allogeneic stem cells (stem cells are from a healthy donor). The transplanted stem cells go from the patient’s blood to his or her marrow. Infusion of either bone marrow or peripheral blood progenitor cells is a relatively simple process that is performed at the bedside. Minimal toxicity is observed in most cases.
The donor is usually a brother or a sister if one is available and if he or she is a match for the patient. Otherwise, an unrelated person with stem cells that match the patient’s tissue type can be used. These matched unrelated donors (MUDs) can be found through stem cell donor banks or registries.
The new cells grow and provide a supply of red cells, white cells (including immune cells) and platelets. The donated stem cells make immune cells that are not totally matched with the patient’s cells. Patients and donors are matched to major tissue types but not minor tissue types. For this reason, the donor immune cells may recognise the patient’s cancer cells’ minor tissue types as foreign and kill the cancer cells. This is called “graft versus cancer effect.”
Transplantation-related mortality and morbidity rates have considerably decreased nowadays because of improved conditioning regimens, human leukocyte antigen (HLA) typing, supportive care, and prevention and treatment of serious infections.
Donor selection:
The following studies are routinely performed on hematopoietic stem cell donors:
> History and physical examination.
> Serum creatinine, electrolyte, and liver function studies.
> Serologic studies for cytomegalovirus (CMV), herpes viruses, HIV RNA, anti-HIV antibodies, hepatitis B and C viruses, human T-cell lymphotropic virus-1/2 (HTLV-I/II), and syphilis (VDRL); in autologous donations, CMV and VDRL testing are not required.
> ABO blood typing.
> Human leukocyte antigen (HLA) typing.
> Chest radiography.
> Electrocardiography (ECG).
Risks to donor:
The risks of a complication depend on patient characteristics, health care providers and the apheresis procedure, and the colony-stimulating factor used (G-CSF). G-CSF drugs include filgrastim (Neupogen, Neulasta), and lenograstim (Graslopin).
Drug risks:
Filgrastim is typically dosed in the 10 microgram/kg level for 4–5 days during the harvesting of stem cells. G-CSF can cause side effects, such as bone pain, muscle aches, headache, fatigue, nausea and vomiting. These usually disappear within a couple of days after the donor stops the injections. A pain reliever like acetaminophen is usually prescribed for the discomfort. If that doesn’t help, the doctor can prescribe another pain medicine e.g. Ibuprofen.Other documented adverse effects of filgrastim include splenic rupture (indicated by left upper abdominal or shoulder pain, risk 1 in 40000), Adult respiratory distress syndrome (ARDS), alveolar haemorrhage, and allergic reactions (usually expressed in first 30 minutes, risk 1 in 300). In addition, platelet and haemoglobin levels dip post-procedure, not returning to normal until one month.
The question of whether geriatrics (patients over 65) react the same as patients under 65 has not been sufficiently examined. Coagulation issues and inflammation of atherosclerotic plaques are known to occur as a result of G-CSF injection. G-CSF has also been described to induce genetic changes in mononuclear cells of normal donors. There is evidence that myelodysplasia (MDS) or acute myeloid leukaemia (AML) can be induced by GCSF in susceptible individuals.
Access risks:
Blood is drawn peripherally in a majority of patients, but a central line to jugular/subclavian/femoral veins may be used in 16 percent of women and 4 percent of men. Adverse reactions during apheresis were experienced in 20 percent of women and 8 percent of men, these adverse events primarily consisted of numbness/tingling, multiple line attempts, and nausea.
The most serious risk associated with donating bone marrow involves the use and effects of anaesthesia during surgery. After the surgery, the donor might feel tired or weak and has trouble walking for a few days. The area where the bone marrow was taken out might feel sore for a few days.
Types of Stem Cell Transplantation:
Hematopoietic stem cell transplantation (HSCT) is a rapidly evolving technique that offers a potential cure for hematologic cancers (leukemias, lymphomas, myeloma) and other hematologic disorders (eg, primary immunodeficiency, aplastic anemia, myelodysplasia). HSCT is also sometimes used for solid tumors (eg, some germ cell tumors) that respond to chemotherapy.
Stem cell transplantation contributes to a cure by restoring bone marrow after myeloablative cancer-eradicating treatments and by replacing abnormal bone marrow with normal bone marrow in nonmalignant hematologic disorders.
If you’re a candidate for a stem cell transplant, your doctor will usually recommend one of three types:
> Autologous: the stem cells come from your own body.
> Allogeneic: the stem cells are from a healthy person (the donor).
> Reduced-intensity stem cell transplantation: Like allogeneic transplant, the stem cells are from a healthy person (the donor), but the chemotherapy given is less intensive.
>Syngeneic transplantation is much less common than the other three. Syngeneic transplantation is rare for the simple reason that it’s only used on identical twins. In addition, the donor twin and the recipient twin must have identical genetic makeup and tissue type.
Indications for HSCT:
Autologous HSCT is used to treat the following conditions:
Multiple myeloma
Non-Hodgkin lymphoma
Hodgkin disease
Acute myeloid leukemia (AML)
Amyloidosis
Autologous stem cell transplant isn’t commonly used to treat acute lymphoblastic leukemia (ALL) because of the high relapse rate nor is it used frequently for myelodysplastic syndromes.
Allogenic HSCT is used to treat the following disorders:
Acute myeloid leukemia
Acute lymphoblastic leukemia
Chronic myeloid leukemia
Chronic lymphocytic leukemia
Myeloproliferative disorders
Myelodysplastic syndromes
Multiple myeloma
Non-Hodgkin lymphoma
Hodgkin disease
Aplastic anemia
Pure red-cell aplasia
Paroxysmal nocturnal hemoglobinuria
Fanconi anemia
Thalassemia major
Sickle cell anemia
Hemophagocytic lymphohistiocytosis
Severe congenital neutropenia
Diamond-Blackfan anemia
Allogeneic stem cell transplant is more successful in younger patients than older patients. About three-quarters of the people who develop a blood cancer are older than 50.
In general, older individuals are more likely to:
> Have complicating medical problems.
> Develop graft versus host disease.
> Have a decreased tolerance for the cumulative effects of the intensive chemotherapy and for radiation treatments needed before the transplant.
Allogenic HSCT is the treatment of choice for all children with acute myeloid leukemia with a human leukocyte antigen (HLA) ̶ matched sibling in their first complete remission (CR1). In adults, this is reserved for those with high-risk features in their CR1. In adults with standard or good risk features, stem cell transplantation is reserved for their second complete remission (CR2). HSCT is the only curative option for patients with primary refractory or relapsed AML. Indications for stem cell transplantation in adults with acute lymphoblastic leukemia are similar to those for persons with AML.
Allogenic HSCT should be considered for patients with myelodysplastic syndrome who are younger than 60 years and who have an HLA-matched sibling donor.
In multiple myeloma, although autologous HSCT does not produce a cure, event-free survival rates and overall survival rates are prolonged approximately 1 year compared with survival rates achieved by chemotherapy. Autologous HSCT is associated with a much lower mortality rate than allogenic HSCT. Trials from France have shown the advantage of double autologous HSCT (tandem transplants) over single transplantation.
Procedure:
For bone marrow stem cell harvest, 700 to 1500 mL (maximum 15 mL/kg) of marrow is aspirated from the donor’s posterior iliac crests; a local or general anaesthetic is used.
For peripheral blood harvest, the donor is treated with recombinant growth factors (granulocyte colony-stimulating factor or granulocyte-macrophage colony-stimulating factor) to stimulate proliferation and mobilization of stem cells; standard apheresis is done 4 to 6 days afterwards. Fluorescence-activated cell sorting is used to identify and separate stem cells from other cells.
Stem cells are then infused over 1 to 2 hours through a large-bore central venous catheter.
After transplantation, recipients are given colony-stimulating factors to shorten the duration of posttransplantation leukopenia, prophylactic anti-infective drugs, and, after allogeneic HSCT, up to 6 months of prophylactic immunosuppressants (typically methotrexate and cyclosporine) to prevent donor T cells from reacting against recipient HLA molecules (graft-vs-host disease). Broad-spectrum antibiotics are usually withheld unless fever develops.
Engraftment typically occurs 10 to 20 days after HSCT (earlier with peripheral blood stem cells) and is defined by an absolute neutrophil count > 0.5 x109/L.
There are no contraindications to autologous HSCT.
Contraindications to allogeneic HSCT are relative and include age > 50, previous HSCT, and significant comorbidities.
Infection prophylaxis may include the following:
> Care in HEPA-filtered, positive-air-pressure–sealed rooms, with strict hand hygiene.
> Antibacterial prophylaxis with a fluoroquinolone (most patients).
> Antifungal prophylaxis with fluconazole or amphotericin B or voriconazole.
> Acyclovir prophylaxis (herpes simplex–positive patients).
> Ganciclovir, IV immunoglobulin (IVIg), and CMV-negative blood products (CMV-seronegative patients).
> Pneumocystis prophylaxis with trimethoprim-sulfamethoxazole or pentamidine.
> Prophylaxis of pneumococcal bacteremia with penicillin, erythromycin, or extended-spectrum fluoroquinolones (immunosuppressed GVHD patients).
> IVIg (patients with documented hypogammaglobulinemia).
> Gut decontamination with metronidazole or fluoroquinolones (acute GVHD).
> Hepatitis B virus (HBV) vaccine (all HBsAg-negative patients).
Conditioning Regimens for Stem Cell Transplantation:
The conditioning regimen, also so-called preparative regimen, is a critical element in the hematopoietic stem cell transplant (HSCT) procedure. This is usually a combination of chemotherapy agents, with or without radiation therapy. The purpose of the preparative regimen is dual and varies according to the type of HSCT: i) to provide adequate immunosuppression to facilitate the patient’s body to accept the donor’s bone marrow cells and prevent
1) to provide adequate immunosuppression to facilitate the patient’s body to accept the donor’s bone marrow cells and prevent rejection; and
2) to eradicate the disease for which the transplant is being performed.
The former goal is needed only in patients undergoing allogeneic stem cell transplantation, but not in those receiving autologous transplants. Fortunately, today most preparative regimens achieve high efficiency in this regard, with graft rejection being an uncommon complication. Nevertheless, when this complication occurs it is associated with a dismal outcome. It is well known that the risk of graft rejection increases with the degree of HLA mismatch between donor and recipient.
Regarding the latter objective, conditioning regimens are usually designed to kill tumor cells resistant to conventional chemotherapy by giving maximally tolerated doses of multiple chemotherapeutic agents without causing fatal nonhematologic organ toxicity. Nevertheless, several novel approaches have been designed in an attempt to minimize toxicity. As an example, Reduced intensity regimens (RIC) and nonmyeloablative conditioning regimens have been developed to treat older patients or those with concurrent comorbidities.
The days the patient receives the preparative regimen are “minus” days (or – days). The number of days will vary, depending on the regimen. For example, the patient may receive treatment on day – 8 through day – 1. Someone else may receive treatment on day – 6 through day – 2. This part of the transplant process is the “countdown” to transplant day, or ‘Day Zero’.
Types of preparative regimens
Myeloablative regimens
A myeloablative regimen usually consists of a combination of agents given at maximally tolerated doses expected to eradicate the hematopoietic cells in the bone marrow and results in profound pancytopenia within one to three weeks from the time of administration. The resulting pancytopenia is long-lasting, usually irreversible, and in most instances fatal unless hematopoiesis is restored by infusion of hematopoietic stem cells.
Nonmyeloablative regimens
A nonmyeloablative regimen results in minimal cytopenia, significant lymphopenia, and does not require stem cell support. In this respect, fludarabine, cyclophosphamide, antithymocyte globulin and total body irradiation (TBI) ≤2 Gy are the most common agents used for this purpose.
Reduced intensity regimens
Reduced intensity regimens (RIC) are an intermediate category of regimens that do not fit the definition of myeloablative or nonmyeloablative. Such regimens cause cytopenias, which may be prolonged and result in significant morbidity and mortality, and require hematopoietic stem cell support.
Myeloablative preparative regimens
All myeloablative regimens are associated with a high degree of myelotoxicity. Cyclophosphamide plus TBI or busulfan are typical approaches for fully myeloablative regimens, but combining busulfan with fludarabine is being increasingly used. According to the use of radiation or not, conditioning regimens can be classified as follows:
Radiation-containing regimens
TBI has been the mainstay of preparative regimens since the beginning of the activity of HSCT. Initial preparative regimens included TBI administered as a single dose using opposing Cobalt-60 sources. At present, TBI-based regimens typically fractionate the radiation and administer the total dose over several days, typically four, which helps decrease toxicity and increase tolerability. Partial lung shielding is included in an effort to reduce the potential for irreversible lung injury.
The most common radiation-containing regimen used for patients receiving allogeneic HSCT has been TBI with cyclophosphamide. The maximally tolerated dose of TBI is approximately 15 Gy. Higher doses produce excessive nonhematologic toxicity, primarily to the lungs, but also to other organs including the heart. Cyclophosphamide is usually given at a dose of 60 mg/kg of adjusted ideal body weight on each of two successive days. Etoposide (VP16) or high dose cytarabine also have been given with TBI, with and without cyclophosphamide.
Chemotherapy-based conditioning regimens (without radiation)
A number of non-radiation-containing regimens in which TBI is replaced with additional chemotherapy have been developed. These approaches were initially developed for autologous transplantation, but they have also been used widely in the allogeneic setting. The most widely used chemotherapy-based regimen is the combination of busulfan and cyclophosphamide (BuCy) with their variants BuCy2 and BuCy4 (2 or 4 days of cyclophosphamide). A variety of other combinations that have been successful used include:
· Busulfan and etoposide (in AML)
· BCNU, cyclophosphamide, cytosine arabinoside and melphalan (BEAM)
· Cyclophosphamide, carmustine (BCNU), and etoposide (Hodgkin and non-Hodgkin lymphomas)
· Busulfan, melphalan, and thiotepa (aggressive or relapsed lymphoma).
Nonmyeloablative and reduced intensity preparative regimens
Less intense conditioning regimens are being used more frequently today to retain the desirable effects of standard high-dose conditioning regimens, but with significantly lower toxicity and, therefore, lower transplant-related mortality. The general principle in which the use of both nonmyeloablative and RIC preparative regimen is based is the so-called graft-versus-tumor (GVT) effect, which is mediated by donor immunocompetent cells in allogeneic HSCT. It is well known that the engraftment of donor type immunocompetent cells does not necessarily require a myeloablative preparative regimen, and can be achieved by using nonmyeloablative or RIC regimens.
RICy regimens are an intermediate category of regimens that do not fit the definition of myeloablative or nonmyeloablative. Such regimens cause cytopenia, which may be prolonged and result in significant morbidity and mortality, and require stem cell support. Regimens generally considered as RIC include ≤9 mg/kg of oral busulfan, or ≤140 mg/m2 of melphalan. The adoption of these less toxic conditioning regimens has expanded the number of patients eligible to receive transplants — patients who may have previously been excluded due to older age or existing co-morbidities. It should be noted, however, that not all diseases are equally susceptible to GVT effect. It is generally accepted that chronic myeloid leukemia, chronic lymphocytic leukemia, and low-grade indolent lymphomas (follicular lymphoma and mantle cell lymphoma) appear particularly responsive to GVT effect.
RIC regimens typically use combinations of chemotherapy drugs such as fludarabine, busulfan, and melphalan, with or without low-dose radiation.
Toxicity of preparative regimens:
Apart from the aforementioned myelotoxicity, the most common side effects that some of them can be life-threatening include:
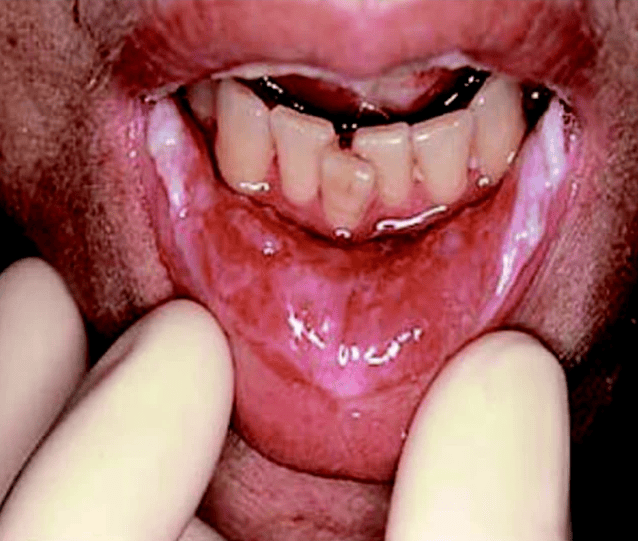
· Mucositis
· Nausea and vomiting
· Alopecia
· Diarrhea
· Rash
· Peripheral neuropathies
· Infertility. Sperm cryopreservation for male and oocyte cryopreservation in female patients can be attempted to overcome this complication, which is almost universal when using myeloablative regimens.
· Pulmonary toxicity: interstitial lung disease
· Hepatic toxicity: hepatic venoocclusive disease or sinusoidal obstructive syndrome.
Long-term complications following TBI used as part of a preparative regimen are relatively common:
· Asymptomatic alterations in pulmonary function (20%)
· Cataracts (15%)
· Sicca syndrome (13%)
· Hypothyroidism (6%)
· Thyroiditis (3%)
Complications of HSCT:
Complications of stem cell transplantation can occur early (< 100 days after transplantation) or later. After allogeneic HSCT, risk of infections is increased.
Early complications:
Major early complications include:
Failure to engraft
Rejection
Acute graft-vs-host disease (GVHD)
Failure to engraft and rejection affect < 5% of patients and manifest as persistent pancytopenia or irreversible decline in blood counts. Treatment is corticosteroids for several weeks.
Acute GVHD occurs in recipients of allogeneic HSCT (in 40% of HLA-matched sibling graft recipients and 80% of unrelated donor graft recipients). It causes fever, rash, hepatitis with hyperbilirubinemia, vomiting, diarrhoea, abdominal pain (which may progress to ileus), and weight loss.
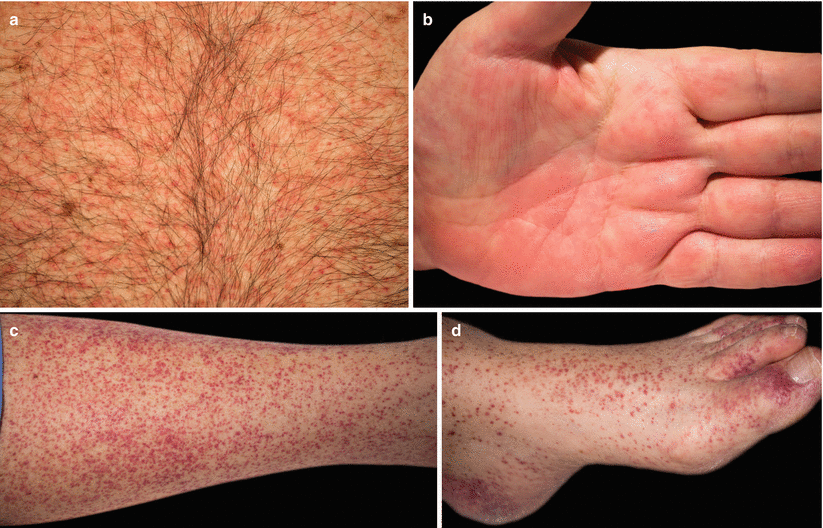
Manifestations of stage II acute cutaneous GVHD. A morbilliform and folliculocentric eruption on the trunk (a), palmar involvement (b), folliculocentric involvement of the lower leg (c) and foot (d) (Courtesy of Dr. Ryan Trowbridge)
Risk factors for acute GVHD include:
HLA and sex mismatching
Unrelated donor
Older age of recipient, donor, or both
Donor presensitization
Inadequate GVHD prophylaxis
Diagnosis of acute GVHD is obvious based on history, physical examination, and liver function test results. Treatment is methylprednisolone 2 mg/kg IV once/day, increased to 10 mg/kg if there is no response within 5 days. Other therapies include antithymocyte globulin (ATG), cyclosporine alone and mycophenolate mofetil (MMF).
Later complications:
Major later complications include:
Chronic GVHD
Disease relapse
Chronic GVHD may occur by itself, develop from acute GVHD, or occur after resolution of acute GVHD. It typically occurs 4 to 7 months after HSCT (range 2 months to 2 years). Chronic GVHD occurs in recipients of allogeneic HSCT (in about 35 to 50% of HLA-matched sibling graft recipients and 60 to 70% of unrelated donor graft recipients).
Chronic GVHD affects primarily the skin (eg, lichenoid rash, scleroderma) and mucous membranes (eg, keratoconjunctivitis sicca, periodontitis, orogenital lichenoid reactions), but it also affects the GI tract and liver. Immunodeficiency is a primary feature; bronchiolitis obliterans similar to that after lung transplantation can also develop. Ultimately, GVHD causes death in 20 to 40% of patients who have it.
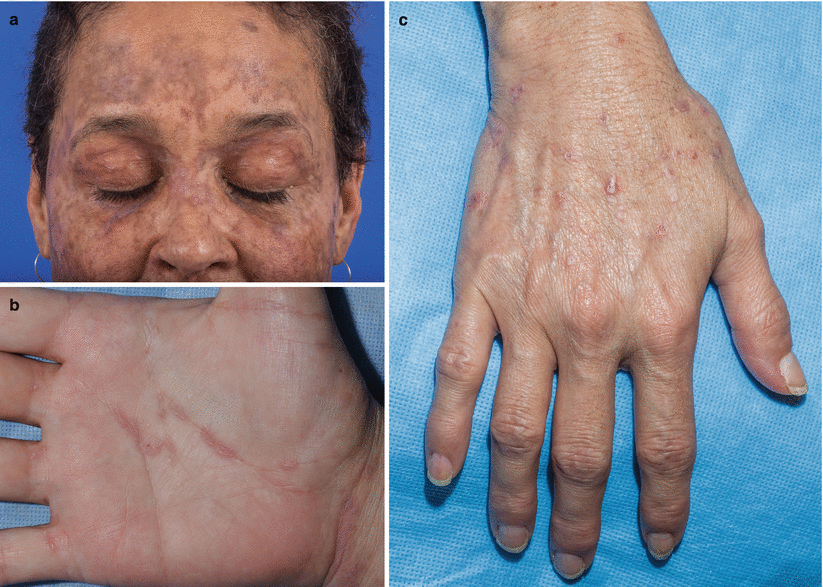
Lichen planus–like chronic graft-versus-host disease (cGVHD). (a) Woman with lichen planus–like cGVHD on the face. (b) Man with lichen planus–like cGVHD on the palm and wrist. (c) Woman with lichen planus–like papules on the dorsal hand and wrist
Treatment may not be necessary for GVHD that affects the skin and mucous membrane; treatment of more extensive disease includes steroids, cyclosporine, thalidomide, MMF, azathioprine, clofazimine, PUVA therapy, extracorporeal photopheresis (a modification of PUVA treatment), rituximab and imatinib. T-cell depletion of allogeneic donor grafts using monoclonal antibodies or mechanical separation reduces incidence and severity of GVHD but also eliminates the graft-vs-tumor effect that may enhance stem cell proliferation and engraftment and reduce disease relapse rates. Relapse rates with autologous HSCT are higher because there is no graft-vs-tumor effect and because circulating tumor cells may be transplanted. Ex vivo tumor cell purging before autologous transplantation is under study.
In patients without chronic GVHD, all immunosuppression can be stopped 6 months after HSCT; thus, late complications are rare in these patients.
Prognosis:
Prognosis after HSCT varies by indication and procedure.
Overall, disease relapse occurs in:
40 to 75% of recipients of autologous HSC transplants.
10 to 40% of recipients of allogeneic HSC transplants.
Success (cancer-free bone marrow) rates are:
30 to 40% for patients with relapsed, chemotherapy-sensitive lymphoma.
20 to 50% for patients with acute leukemia in remission.
Compared with chemotherapy alone, HSCT improves survival of patients with multiple myeloma. Success rates are low for patients with more advanced disease or with responsive solid cancers (eg, germ cell tumors). Relapse rates are reduced in patients with graft-vs-host disease (GVHD), but overall mortality rates are increased if GVHD is severe.
Intensive conditioning regimens, effective GVHD prophylaxis, cyclosporine-based regimens, and improved supportive care (eg, antibiotics as needed, herpesvirus and cytomegalovirus prophylaxis) have increased long-term disease-free survival after HSCT.
References:
Leukemia & Lymphoma Society. Stem Cell Transplantation – Treatment Overview. https://www.lls.org/treatment/types-of-treatment/stem-cell-transplantation
Science Learning Hub. Bone Marrow Transplants – Timeline. https://www.sciencelearn.org.nz/resources/1971-bone-marrow-transplants-timeline
Perumbeti A., MD, FAAP. Hematopoietic Stem Cell Transplantation. Medscape Reference. https://emedicine.medscape.com/article/208954-overview (Accessed April 23, 2016)
Hertl M., MD, PhD; Russell P.S., MD. Hematopoietic Stem Cell Transplantation – Immunology and Allergic Disorders. MSD Manual Professional Edition. https://www.msdmanuals.com/professional/immunology-allergic-disorders/transplantation/hematopoietic-stem-cell-transplantation (Accessed September 2016)
Rzepecki P., Pielichowski W., Oborska S., Barzal J., Mlot B. Palonosetron in Prevention of Nausea and Vomiting After Highly Emetogenic Chemotherapy Before Hematopoietic Stem Cell Transplantation – Single Center Experience. Transplant Proc. 2009; 41(8): 3247–3249.
Nakasone H., Kanda Y., Ueda T., Matsumoto K., Shimizu N., Minami J., et al. Retrospective Comparison of Mobilization Methods for Autologous Stem Cell Transplantation in Multiple Myeloma. Am J Hematol. 2009; 84(12): 809–814.
Sanz J. Conditioning Regimens for Stem Cell Transplantation. University Hospital La Fe, Valencia, Spain. http://www.bmtcare.com/index.php?option=com_content&view=article&id=68&itemid=59&lang=en
Atkinson K. Chronic Graft-Versus-Host Disease. Bone Marrow Transplant. 1990; 5(2): 69–82.
Couriel D.R., Hosing C., Saliba R., et al. Extracorporeal Photochemotherapy for the Treatment of Steroid-Resistant Chronic GVHD. Blood. 2006; 107(8): 3074–3080.
Cutler C., Miklos D., Kim H.T., et al. Rituximab for Steroid-Refractory Chronic Graft-Versus-Host Disease. Blood. 2006; 108(2): 756–762.
Olivieri A., Locatelli F., Zecca M., Sanna A., Cimminiello M., Raimondi R., et al. Imatinib for Refractory Chronic Graft-Versus-Host Disease With Fibrotic Features. Blood. 2009; 114(3): 709–718.
Pamphilon D., Siddiq S., Brunskill S., Dorée C., Hyde C., Horowitz M., Stanworth S. Stem Cell Donation – What Advice Can Be Given to the Donor? Br J Haematol. 2009; 147(1): 71–76.
NHS Blood & Transplant. Donating Stem Cells. https://www.blood.co.uk/stem-cell-donor-registry/donating-stem-cells/
DKMS. Peripheral Blood Stem Cell Collection – PBSC Procedure. https://www.dkms.org/donor-info/stem-cell-donation/peripheral-blood-stem-cell-collection
Kobyzeva D., Shelikhova L., Loginova A., Kanestri F., Tovmasyan D., Maschan M., Khismatullina R., Ilushina M., Baidildina D., Myakova N., Nechesnyuk A. Optimized Conformal Total Body Irradiation Among Recipients of TCRαβ/CD19-Depleted Grafts in Pediatric Patients With Hematologic Malignancies: Single-Center Experience. Front Oncol. 2021; 11: 785916. doi:10.3389/fonc.2021.785916
Fall-Dickson R., Ramsay. Oral Mucositis-Related Oropharyngeal Pain and Tumor. Semanticscholar. https://www.semanticscholar.org/paper/Oral-mucositis-related-oropharyngeal-pain-and-tumour-Fall%E2%80%90Dickson-Ramsay/e985be396bf7467a7f2a473f9df86e4b6e12c5fc
Mikailov D., Plovanich M. Grading and Treatment of Acute Graft-Versus-Host Disease. PlasticsurgeryKey. 2017. https://www.plasticsurgerykey.com/grading-and-treatment-of-acute-graft-versus-host-disease/
PlasticsurgeryKey. Diagnosis, Staging, and Treatment of Chronic Graft-Versus-Host Disease. https://www.plasticsurgerykey.com/diagnosis-staging-and-treatment-of-chronic-graft-versus-host-disease/
National Cancer Institute (NCI). Stem Cell Transplant for Cancer. https://www.cancer.gov/about-cancer/treatment/types/stem-cell-transplant
World Health Organization (WHO). Guidelines on Hematopoietic Stem Cell Transplantation. WHO Technical Report Series. Geneva: World Health Organization; 2018. https://www.who.int/
Keywords:


Request Online Consultation With Dr M Abdou
Fee: US$100
Secure payment via PayPal (credit and debit cards accepted)
Pay Now






I am very happy to see this post because it really a nice post. Thanks