Plasmapheresis
What is Plasmapheresis?
Plasmapheresis, also known as therapeutic plasma exchange (TPE), is a medical procedure that involves removing a patient’s plasma and replacing it with a substitute such as saline, albumin, or donor plasma. The process separates plasma from blood cells using a centrifuge or membrane filtration system, allowing the selective removal of pathogenic antibodies, immune complexes, and toxins. Plasmapheresis is widely used in hematology, neurology, and immunology to manage autoimmune and hematologic disorders including myasthenia gravis, Guillain-Barré syndrome, thrombotic thrombocytopenic purpura (TTP), and cryoglobulinemia. By rapidly reducing circulating autoantibodies and abnormal plasma proteins, plasmapheresis provides both diagnostic and therapeutic benefits in a range of immune-mediated diseases. Understanding the indications, mechanism, and complications of this procedure is essential for optimizing patient outcomes and integrating plasma exchange therapy into evidence-based clinical practice.
The word ‘plasmapheresis’ (meaning the withdrawal of plasma) was coined by Abel in 1914 to describe a process by which whole blood is withdrawn from the body and separated into its constitutive parts, the cells being returned to the body while the plasma is retained.
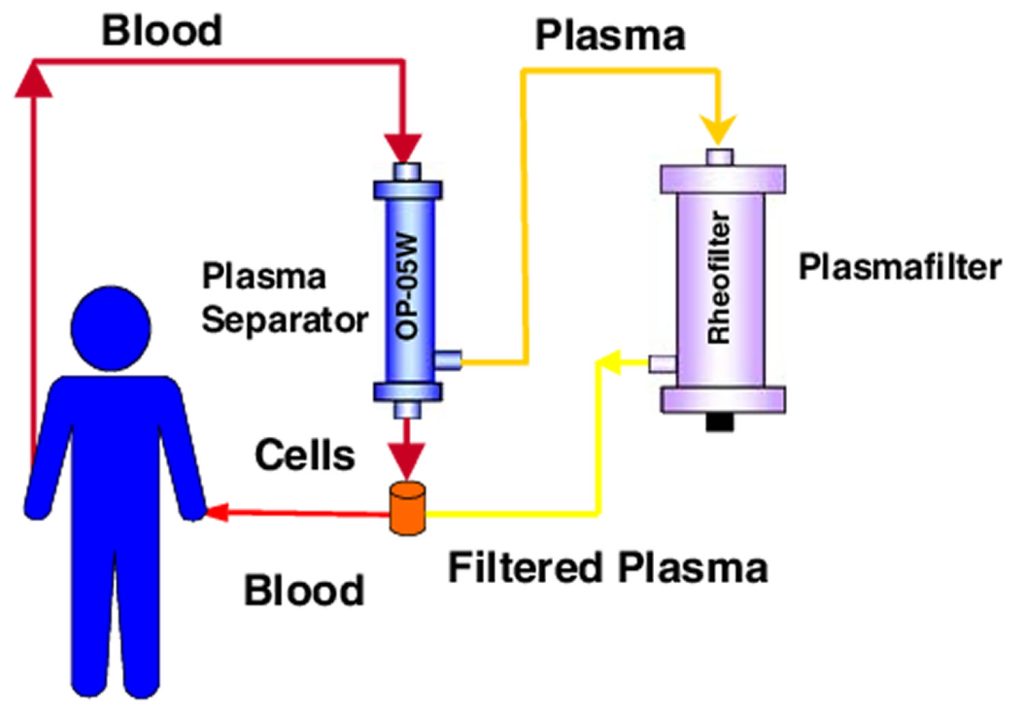
Plasmapheresis diagram illustrating plasma separation and plasma filtration using a plasma separator and rheofilter.
Plasmapheresis is used in the treatment of many different conditions, including autoimmune disorders. When the plasma is removed, it takes with it the antibodies that have been developed against self-tissue in an attempt to reduce the attack on the patient’s own body. Plasmapheresis carries with it the same risks as any intravenous procedure but is otherwise generally safe.
In other words, plasmapheresis is a process that filters the blood and removes harmful antibodies. It is a procedure done similarly to dialysis; however, it specifically removes antibodies from the plasma portion of the blood. Antibodies are part of the body’s natural defense system which help destroy things that are not a natural part of our own bodies, like germs or bacteria.
Pheresis, or apheresis, describes any process that removes the blood, filters and retains elements of it, then returns the blood to the body. Platelets, red blood cells, white blood cells, or plasma may be separated.
The procedure is performed using a machine that removes small amounts of blood at a time.
Plasmapheresis machine used in therapeutic plasma exchange, showing plasma collection and filtration components.
There are two ways to separate the components of blood:
- Centrifugation. This process spins the blood, which divides it according to the density of the parts.
- Filtration. This involves passing the blood through a filter to separate plasma.
During plasma exchange, the machine will dispose of unhealthy plasma and replace it with healthy plasma from a donor. Unhealthy plasma can also be replaced with saline, albumin, or a combination of the two.
Technical Considerations:
Although the term plasmapheresis technically refers only to the removal of plasma, it is also widely used to encompass therapeutic plasma exchange in which a replacement product is transfused after removal of the plasma.
As distinct from plasmapheresis, cytapheresis is the selective removal of RBCs, WBCs, or platelets and can be accomplished by using identical centrifuge-based equipment. Applications include the following:
Erythrocytapheresis (selective removal of RBCs) is used in conditions such as sickle cell disease or malarial infection, in which RBCs are selectively removed and replaced with donor erythrocytes.
Leukapheresis (selective removal of WBCs) is used in conditions such as hyperleukocytosis, in which a pathologically high number of white cells are present (as, for example, in leukemia); it can also be used to collect peripherally circulating stem cells that can then be infused in an autologous or allogeneic stem cell transplant.
Platelet apheresis (selective removal of platelets) can be used in conditions of thrombocytosis (eg, essential thrombocythemia and polycythemia vera)
Indications of Plasmapheresis:
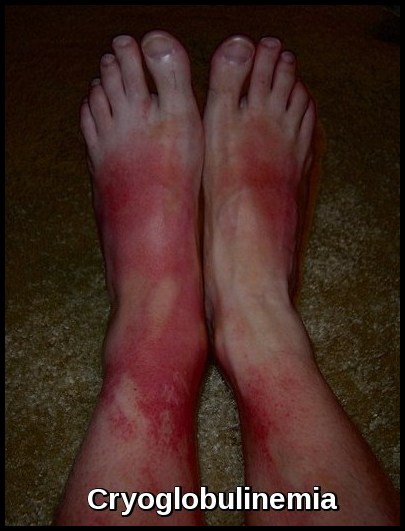
Plasmapheresis is currently used as a therapeutic modality in a wide array of conditions. Generally, it is used when a substance in the plasma, such as immunoglobulin, is acutely toxic and can be efficiently removed. Myriad conditions that fall into this category (including neurologic, hematologic, metabolic, dermatologic, rheumatologic, and renal diseases, as well as intoxications) can be treated with plasmapheresis.
Therapeutic plasmapheresis is a process by which constituents of plasma that are supposed to cause or aggravate disorders are selectively removed. The remaining components of blood are then mixed with a plasma substitute or an inert replacement and given back to the patient. Blood components that are removed may include immune complexes, lipids, antibodies, toxins, mediators of complement activation or inflammation. Molecules, that are supposed to be potentially harmful, are also removed.
This procedure is used for treating many autoimmune diseases with different success rates. Typically, the process is used to rapidly lessen immune complexes or circulating antibodies during autoimmune conditions. The method is frequently used along with other immunosuppressive therapies that help to enhance its beneficial effects or make them more long-lasting.
The Apheresis Applications Committee of the American Society for Apheresis (ASFA) periodically evaluates potential indications for apheresis and categorizes them from I to IV in the basis of the available medical literature. The following are some of the indications, and their categorization, from the society’s guidelines.
Category I (disorders for which apheresis is accepted as first-line therapy, either as a primary standalone treatment or in conjunction with other modes of treatment) are as follows:
- Guillain-Barre syndrome.
- Myasthenia gravis.
- Chronic inflammatory demyelinating polyneuropathy.
- Hyperviscosity in monoclonal gammopathies.
- Thrombotic thrombocytopenic purpura.
- Goodpasture syndrome (unless it is dialysis-dependent and there is no diffuse alveolar hemorrhage).
- Hemolytic uremic syndrome (atypical, due to autoantibody to factor H).
- Wilson disease, fulminant.

Guillain-Barré syndrome illustration highlighting clinical features and treatment options, including plasmapheresis and IVIG.

Myasthenia gravis presenting with ptosis, one of the classic clinical features and an indication for plasmapheresis in severe exacerbations.
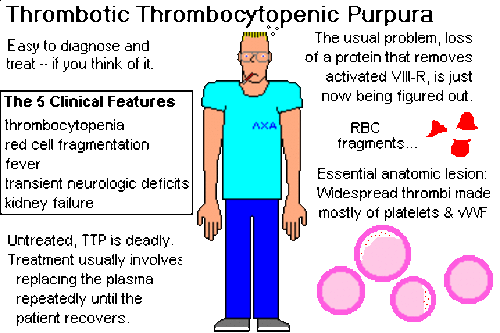
Thrombotic thrombocytopenic purpura (TTP) diagram summarizing clinical features, red cell fragmentation, and the importance of plasmapheresis in management.
Category II (disorders for which apheresis is accepted as second-line therapy, either as a standalone treatment or in conjunction with other modes of treatment) are as follows:
- Lambert-Eaton myasthenic syndrome.
- Multiple sclerosis (acute central nervous system demyelination disease unresponsive to steroids).
- RBC alloimmunization in pregnancy.
- Mushroom poisoning.
- Acute disseminated encephalomyelitis.
- Hemolytic uremic syndrome (atypical, due to complement factor mutations).
- Autoimmune hemolytic anemia (life-threatening cold agglutinin disease).
- Systemic lupus erythematosus (severe).
- Myeloma cast nephropathy.
Category III (disorders for which the optimal role of apheresis therapy is not established; decision-making should be individualized) are as follows:
- Post-transfusion purpura.
- Autoimmune hemolytic anemia (warm autoimmune hemolytic anemia).
- Hypertriglyceridemic pancreatitis.
- Thyroid storm.
Category IV (disorders in which published evidence demonstrates or suggests apheresis to be ineffective or harmful; institutional review board [IRB] approval is desirable if apheresis treatment is undertaken in these circumstances) are as follows:
- Stiff person syndrome.
- Hemolytic uremic syndrome (typical diarrhea-associated).
- Systemic lupus erythematosus (nephritis).
- Immune thrombocytopenia.
Benefits of Plasmapheresis:
Plasma exchange can help to alleviate symptoms of the conditions above by removing harmful substances from the blood.
If a person has an autoimmune condition, plasma exchange may also prevent the body from producing more harmful antibodies.
The procedure is usually one element of a treatment plan, which may include chemotherapy. Repeated plasma exchanges may be necessary.
Plasmapheresis Procedure:
A medical professional will perform plasmapheresis, usually in a hospital but sometimes in a private clinic.
Premedication with acetaminophen, diphenhydramine, and hydrocortisone are often given.
A local anesthetic will numb the affected area, and the procedure should not cause pain.
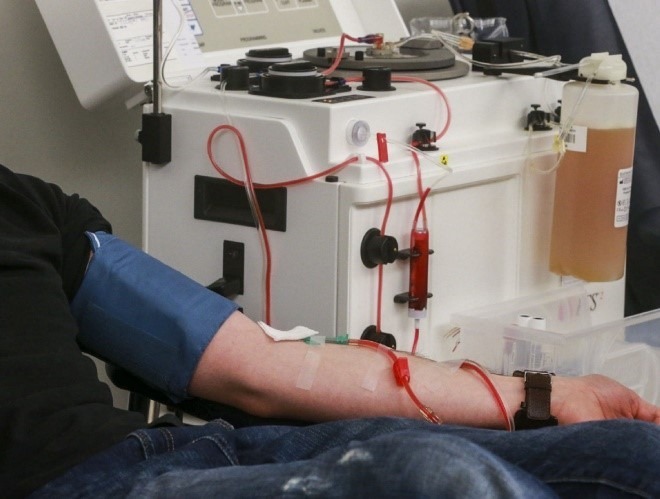
The doctor will then insert a small tube into a vein in the arm or the groin. The tube will bring blood to the machine, which will collect it, treat it, and return it to the body.
Plasma exchange takes between 2 and 4 hours. A person will need to remain as still as possible to help the blood to flow smoothly. It may help to watch television or read as a distraction.
Plasmapheresis Procedure Explained (Video)
This video provides a visual demonstration of therapeutic plasma exchange (plasmapheresis) in the context of autoimmune disorders such as myasthenia gravis. It covers the indication, machine setup, separation of plasma, removal of harmful antibodies and is aligned with the American Society for Apheresis (ASFA) Category I/II guidelines. Embedding it here enhances your understanding of the full procedure and its clinical application.
A medical professional will be present and check for side effects throughout the process.
After the plasma exchange is complete, the machine will be disconnected, and new blood tests will be performed.
Plasmapheresis procedure showing connection to a therapeutic plasma exchange machine used for antibody removal in autoimmune diseases.
Complications of Plasmapheresis:
Patients can experience symptoms of hypocalcemia and or hypomagnesemia during and after the procedure and can be treated with replacement calcium and magnesium, respectively.
Patients frequently become hypothermic during the procedure, in which case they should be warmed appropriately.
Patients can experience transfusion-related reactions, in particular with FFP, and should be treated with diphenhydramine, hydrocortisone, and/or epinephrine depending on the severity of the reaction. These reactions can occur during and after the transfusion.
Patients can experience hypotension as a result of rapid fluid shifts, and proper precautions should be taken to minimize complications such as unintended falls.
Patients can become thrombocytopenic and hypofibrinogenemic after plasmapheresis (especially if albumin is being used as a replacement product) and should be monitored for signs of bleeding.
Patients may also be at further risk for developing hypotension if they have a history of taking angiotensin-converting enzyme (ACE) inhibitors, in particular, while undergoing column-based plasmapheresis. The suspected mechanism is related to increased bradykinin levels caused by the use of ACE inhibitors. This accumulation of kinins leads to hypotension, flushing, and gastrointestinal symptoms. Patients are therefore advised to stop all ACE inhibitors at least 24 hours before starting plasmapheresis.
Contraindications of Plasmapheresis:
Plasmapheresis is contraindicated in the following patients:
- Patients who cannot tolerate central line placement.
- Patients who are actively septic or are hemodynamically unstable.
- Patients who have allergies to fresh frozen plasma or albumin, depending on the type of plasma exchange.
- Patients with heparin allergies should not receive heparin as an anticoagulant during plasmapheresis.
- Patients with hypocalcemia are at risk for worsening of their condition because citrate is commonly used to prevent clotting and can potentiate hypocalcemia.
- Patients taking angiotensin-converting enzyme (ACE) inhibitors are advised to stop taking the medication for at least 24 hours before starting plasmapheresis.
Plasmapheresis VS Plasma Exchange:
Definitions
- Apheresis is the general technique of extracorporeal blood purification whereby one constituent is removed and the remainder is returned to the patient.
- Removal may involve centrifugation or filtration.
- Cytapheresis is the removal of cellular components from the blood.
- Plasmapheresis is a subset of apheresis whereby plasma is removed; this is termed ‘plasma exchange’ when host plasma discarded and replaced by donor plasma or an alternative colloid.
Plasmapheresis (Mayo Clinic) Explained – Plasma Exchange Procedure
This video from the Mayo Clinic provides a clear, structured explanation of the plasmapheresis (therapeutic plasma exchange) procedure, demonstrating how plasma is separated, removed, and replaced to eliminate pathogenic antibodies in conditions such as myasthenia gravis, Guillain-Barré syndrome, and other ASFA Category I/II indications. It highlights the technical steps, clinical rationale, and safety considerations involved, helping patients and clinicians understand the role of plasmapheresis in managing autoimmune and neurologic disorders.
Plasmapheresis Cost:
The cost of plasmapheresis (therapeutic plasma exchange) can be significant, with each session in the United States typically ranging from USD 5,000 to 10,000 depending on the institution, indication, and replacement fluid used. Many patients require multiple sessions to achieve a clinical response, which increases overall treatment expenses. Comprehensive medical insurance coverage is strongly recommended, as it can substantially reduce out-of-pocket costs.
Summary:
Plasmapheresis, also known as plasma exchange, is a medical procedure that involves the removal, treatment, and return of blood plasma from a patient. This process is used to treat a variety of medical conditions, including autoimmune disorders, neurological disorders, and certain toxicities. Plasmapheresis is a valuable tool in the treatment of many medical conditions. By removing harmful substances from the bloodstream, this procedure can help alleviate symptoms and improve outcomes for patients. Medical professionals should be familiar with the indications for plasmapheresis and the potential risks and benefits of this procedure. As with any medical procedure, plasmapheresis carries some risks. These may include:
- Bleeding or infection at the site of catheter insertion
- Allergic reactions to replacement fluid or medication
- Low blood pressure or other cardiovascular complications
- Blood clots or other complications related to the use of catheters or other medical devices
Patients undergoing plasmapheresis should be closely monitored for any signs of adverse reactions or complications.
Questions and Answers:
What is plasmapheresis used for?
Plasmapheresis, or therapeutic plasma exchange, is used to remove harmful antibodies, immune complexes, and pathogenic proteins from the bloodstream. It is commonly indicated in conditions such as TTP, myasthenia gravis, Guillain-Barré syndrome, hyperviscosity syndromes, and other ASFA Category I and II autoimmune or neurologic disorders.
How does plasmapheresis work?
Plasmapheresis separates plasma from whole blood using an apheresis machine. The patient’s plasma is removed and replaced with albumin or donor plasma, allowing rapid removal of circulating antibodies and inflammatory mediators that drive disease activity.
Is plasmapheresis painful or risky?
Plasmapheresis is generally well-tolerated, though mild side effects such as hypotension, tingling, fatigue, or temporary catheter discomfort can occur. Serious complications are uncommon but may include allergic reactions, electrolyte imbalances, or infection related to central venous access.
How many plasmapheresis sessions are usually required?
The number of sessions depends on the underlying disease. For example, TTP may require daily exchanges until platelet recovery, while myasthenia gravis or Guillain-Barré syndrome typically require 4–6 sessions over 1–2 weeks. Treatment frequency is determined by clinical response and laboratory markers.
Is plasmapheresis more effective than IVIG?
Plasmapheresis and IVIG are both effective for immune-mediated neurological disorders such as myasthenia gravis and Guillain-Barré syndrome. Choice of therapy depends on clinical severity, comorbidities, availability, and patient-specific factors. In some situations, plasmapheresis offers faster antibody removal.
How long does each plasmapheresis session take?
A typical plasmapheresis session lasts 2 to 3 hours. This includes catheter setup, plasma separation, filtration, replacement fluid infusion, and monitoring by the apheresis team.
Does plasmapheresis provide permanent results?
Plasmapheresis offers rapid improvement by removing circulating antibodies, but it does not stop the body from producing new ones. For long-term disease control, it is usually combined with immunosuppressive therapy such as steroids, rituximab, azathioprine, or mycophenolate.
What conditions are ASFA Category I indications for plasmapheresis?
ASFA Category I indications — where plasmapheresis is accepted as first-line therapy — include thrombotic thrombocytopenic purpura (TTP), Guillain-Barré syndrome, myasthenia gravis crisis, hyperviscosity syndrome due to monoclonal proteins, and certain cases of antibody-mediated transplant rejection.
How much does plasmapheresis cost?
In the United States, each plasmapheresis session typically costs USD 5,000–10,000 depending on hospital setting, replacement fluids, and the number of cycles required. Patients needing multiple sessions benefit significantly from comprehensive medical insurance coverage.
What is the difference between plasmapheresis and plasma exchange?
Although often used interchangeably, “plasmapheresis” usually refers to plasma removal without significant volume replacement, whereas “therapeutic plasma exchange (TPE)” involves removing plasma and replacing it with albumin or donor plasma. In clinical practice, TPE is the standard method for immune-mediated diseases.
References:
Elliot Stieglitz, MD; Emmanuel C. Besa, MD. Plasmapheresis: Background, Indications, Contraindications. Medscape. Available at: https://emedicine.medscape.com/article/1895577-overview
Schwartz J, Padmanabhan A, Aqui N, Balogun RA, Connelly-Smith L, Delaney M, et al. Guidelines on the Use of Therapeutic Apheresis in Clinical Practice—Evidence-Based Approach from the American Society for Apheresis (ASFA): The Seventh Special Issue. J Clin Apher. 2016;31(3):149–162. doi:10.1002/jca.21470
Claire Sissons. Plasmapheresis: Side Effects and How It Works. Medical News Today. Updated April 10, 2018. https://www.medicalnewstoday.com/articles/321451.php
McLeod BC, Sniecinski I, Ciavarella D, Owen H, Price TH, Randels MJ, et al. Frequency of Immediate Adverse Effects Associated with Therapeutic Apheresis. Transfusion. 1999;39(3):282–288. doi:10.1046/j.1537-2995.1999.39399283085.x
Mokrzycki MH, Kaplan AA. Therapeutic Plasma Exchange: Complications and Management. Am J Kidney Dis. 1994;23(6):817–827. doi:10.1016/S0272-6386(12)70133-8
Brian G. Weinshenker, MD. Plasma Exchange in the Treatment of Multiple Sclerosis and Neuromyelitis Optica. Mayo Clinic. https://www.mayoclinic.org/
Chris Nickson. Apheresis, Plasmapheresis and Plasma Exchange. Life in the Fast Lane (LITFL). https://litfl.com/apheresis-plasmapheresis-and-plasma-exchange/
HealthJade Editorial Team. Plasmapheresis Overview. https://healthjade.net/plasmapheresis/
Kapoor R, Mehta R, Sharma S. Plasmapheresis in Neurology: An Overview. Ann Indian Acad Neurol. 2011;14(3):148–153. doi:10.4103/0972-2327.85922
Keegan M, Pineda A. Plasma Exchange in Neurologic Disorders. Neurology. 2002;58(8 Suppl 5):S21–S26. doi:10.1212/WNL.58.suppl_5.S21
Clark WF, Rock GA, Buskard NA, et al. Therapeutic Plasma Exchange: An Update from the Canadian Apheresis Group. Ann Intern Med. 1999;131(7):453–462. doi:10.7326/0003-4819-131-7-199910050-00008
Klassen J, Smith W, Tarr T, Emery D, Gray D. Plasmapheresis in the Treatment of Autoimmune Disease: Review of Evidence and Clinical Practice. J Clin Apher. 2019;34(4):362–375. doi:10.1002/jca.21779
StatPearls Publishing. Therapeutic Plasma Exchange. StatPearls [Internet]. Treasure Island (FL). https://www.ncbi.nlm.nih.gov/books/NBK441956/
National Institutes of Health (NIH). Apheresis and Plasma Exchange Information for Clinicians. https://www.nih.gov/
Keywords:
plasmapheresis, therapeutic plasma exchange, plasma exchange, TPE procedure, TPE indications, plasmapheresis indications, ASFA guidelines, thrombotic thrombocytopenic purpura, TTP treatment, myasthenia gravis crisis, MG exacerbation, Guillain-Barré syndrome treatment, GBS plasmapheresis, CIDP plasmapheresis, autoimmune neuropathy treatment, hyperviscosity syndrome, cryoglobulinemia plasma exchange, Goodpasture syndrome, apheresis therapy, antibody removal therapy, plasma filtration, rheopheresis, plasma exchange complications, plasmapheresis side effects, plasmapheresis cost, therapeutic apheresis, hematology procedures, neurology autoimmune therapy, Dr Moustafa Abdou hematology, Ask Hematologist plasmapheresis

Request Online Consultation With Dr M Abdou
Fee: US$100
Secure payment via PayPal (credit and debit cards accepted)
Pay Now



I didn’t know that plasmapheresis might include therapeutic plasma exchange processes like leukapheresis and platelet apheresis. I wonder if leukapheresis is the type of plasmapheresis that my brother needs. My mom says they think he might have leukemia, so she’s taking him to a doctor soon.
Hi Sarah,
Thank you for your comment.
Although the term plasmapheresis technically refers only to the removal of plasma, it is also widely used to encompass therapeutic plasma exchange in which a replacement product is transfused after removal of the plasma. As distinct from plasmapheresis, cytapheresis is the selective removal of RBCs, WBCs, or platelets and can be accomplished by using identical centrifuge-based equipment.
Regarding your Mother’s blood problem, you can send me her most recent FBC image by email to have a look and advice.
Kind regards,
I read that plasmapheresis is a treatment protocol for patients with PGNMID condition. How good it is?
Hi Nedhal,
Thank you for your comment.
It would be better to discuss with a Nephrologist if plasmapheresis has an effective role here or not? However, since the pathogenesis of PGNMID involves a clonal proliferation of B-cells, treating patients with rituximab or bortezomib along with steroids can lead to the improvement of proteinuria and renal function.
BW,
Thank u very much
Hello,
My friend is suffering very terribly from akathisia, following 12 months of 37 prescriptions of many different psychoactive drugs. I think that she is literally poisoned by them due to polymorphisms in her CYP450 genes, meaning that her body can’t fully metabolise and excrete them.
She has asked me to be her advocate and together we have asked her doctor to arrange for a test of her CYP450 genes and a test for the presence of psychoactive metabolites of the pharmaceuticals prescribed to her, in her blood. If the doctor agrees to do the tests, and if metabolites are found, showing that she is literally poisoned, could she then justifiably be treated by plasmapharesis to remove the metabolites, to give her relief from torturous suffering and save her life.
She lives in Oregon, USA.
I would be grateful for your thoughts,
Thank you,
Hi Elisabeth,
Contrary to the literature about drug removal during hemodialysis, data regarding drug removal during plasmapheresis are sparse. Over the last 40 years, approximately 70 publications—mostly case reports of overdoses—have described the effects of plasmapheresis on pharmaceutical agents.
BW,
Hi. My name is Mansi. My father is a chronic liver cirrhosis patient since 7-8 years and has developed alot of jaundice in past 20 days. No medicine was giving good results to control rising bilurubin so To control his bilirubin level doctors suggested plasma exchange process as his T- cells were very low too. I wanted to ask if this process is safe and beneficial or not. Thankyou.
Hi Mansi,
I don’t think plasma exchange could offer a long-term benefit in such a case.
BW,
There are a lot of new gene therapies on the horizon for diseases like Duchenne muscular dystrophy that use a viral vector for delivery. One of the most common vectors used is AAV (adeno-associated virus) that is harmless to people, however many people have been exposed to it and have antibodies to it. As of right now the pharmaceutical companies are saying that AAV based gene therapy can be given only once and only to people who don’t have antibodies to the vector already from previous natural exposure. Isn’t plasmapheresis the solution for this? What am I missing?
Thank you!
Hi Ana,
Thank you for your comment.
Plasmapheresis is a procedure that filters and purifies the blood by removing harmful antibodies located in the plasma, thereby preventing them from attacking the body e.g. in autoimmune diseases. Theoretically, your assumption is logic regarding the removal of AAV antibodies from the body via plasmapheresis prior to gene therapy but it is very unlikely that the procedure would remove 100% of antibodies in the plasma. In some autoimmune diseases, one round of plasmapheresis is estimated to remove 60-70% of the disease-causing autoantibodies in the plasma.
BW,
Can you do Plasmapheresis and pulse Steroid together? Will it remove steroids too? Should you wait to do plasmapheresis after 3 days of pulse in very sick CNS lupus/Sjogren patient?
Hi Ferha,
Thank you for your comment.
In general, avoid administration of your medications including steroids right before plasmapheresis if possible.
In this study, the amount of steroids removed by plasma exchange was not significant.
Removal of prednisone and prednisolone by plasma exchange
BW,
My daughter has autoimmune encephalitis seronegative . We have tried rituximab and Actemra. the progress has been slow do you think plasmapheresis could be beneficial?
Hi Chelsea,
Thanks for your comment.
I’ll refer you to this article hoping it could help to answer your query:
Therapeutic plasma exchange as a life-saving therapy in a suspected case of autoimmune encephalitis: A case report from a tertiary health-care center
Abstract:
In half of the suspected autoimmune encephalitis (AIE) patients, no antibodies are identified despite extensive investigation. Therapeutic plasma exchange (TPE) is a potential first-line therapy for various subtypes of AIE. Here, we present a case of autoantibody-negative-suspected AIE, managed successfully with TPE after patient showed no response to steroids. A total of 5 sessions of TPE was done. One standard TPE procedure session was 1.2–1.5 plasma volume exchanges using 5% albumin as a replacement fluid. After five sessions, patient’s clinical condition improved significantly, and a repeated magnetic resonance imaging after 5th cycle of TPE revealed a reduction in the areas of signal alteration. This was suggestive of regression of disease. Patient was discharged on 10th day of hospital admission. With early suspicion even in the absence of detectable autoantibodies, TPE plays an important role in the management of encephalitis.
BW,
My sister had optic neuritis , how often does she have to have plasmapheresis done after the first treatment? We dont have insurance , any medication she can take in replacement to doing the treament? And if does what kind of medication?
Hi Mae,
Thank you for your comment.
Generally speaking, optic neuritis could lead to severe visual impairment despite corticosteroids.
Plasma exchange as second-line therapy can lead to a functionally important visual recovery in more than half of patients with severe optic neuritis.
However, in the particular case of your sister you have to consult her ophthalmologist for what further treatment she would need.
BW,
Hello Dr.
I left the hospital in June last year. Had 3 weeks in ICU plus 30 days more with tracheostomy. This was because of Covid19. Since I left the hospital have gain significant improvement with phisical therapy and rounds of IVIG last year. Plasmapheresis has been offered to me now because still have severe neuropatic pain in feet and calf, with numbness and one foot drop that I have been dealing with since I left the hospital. My diferential diagnosis is Critical Patient Neuropathy/Miopathy versus possible AIDP.
My questions is. ¿Am I too late for Plasmapheresis? ¿How many sessions are needed in the treatment? I have heard 4-5 sessions. Finaly How many hours per session? I am 46 years old.
Hi Ivan,
Thank you for your comment and I wish you a speedy recovery.
Evidence suggests that patients with severe Guillain-Barre’ syndrome may benefit from plasma exchange after immunoglobulin treatment in refractory cases.
Plasma exchange should be considered early in Guillain-Barre’ syndrome cases with axonal involvement, and in the recurrent or familial Guillain-Barré syndrome forms.
I would suggest reading this article for further information.
In mild Guillain-Barré syndrome two sessions of plasma exchange are superior to none.
In moderate Guillain-Barré syndrome four sessions are superior to two.
In severe Guillain-Barré syndrome six sessions are no better than four.
The Plasmapheresis procedure can take anywhere from 2 to 4 hours, and you will be required to stay still as much as possible during this procedure.
BW,
Thank you for a helpful article. I hope you won’t mind if I ask a couple of questions. (Forgive me if these are answered in the article – I’m not very well at the moment and my reading comprehension isn’t the best.)
Is the whole blood volume treated?
Does the process remove all antibodies, or just harmful antibodies?
If it removes all antibodies (good and bad), does that leave the patient compromised in any way?
Can removing harmful antibodies have a positive long-term effect of any kind, or will the patient need to continue the treatments indefinitely?
Hi Sarah,
Thank you for your comment.
Plasmapheresis removes plasma containing pathogenic substances.
Blood components are separated by centrifugation or ultrafiltration.
Discarded plasma must be replaced, usually with a protein-electrolyte solution or donor plasma.
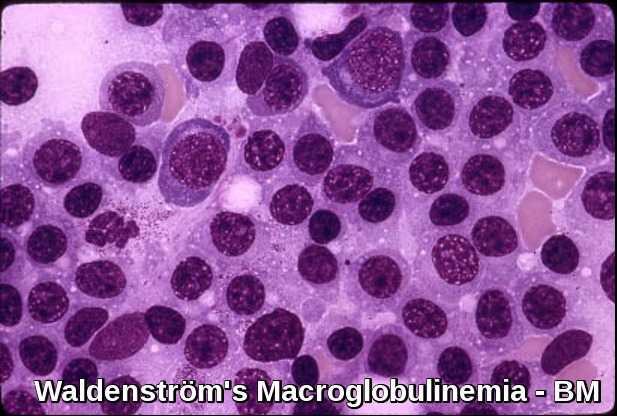
About 80% of plasmaphereses are performed for the following five core indications: thrombotic thrombocytopenic purpura, Guillain-Barré syndrome, chronic inflammatory demyelinating polyneuropathy, myasthenia gravis, and Waldenström’s macroglobulinemia. New indications for plasmapheresis, such as sepsis and multiple-organ failure, merit investigation with large randomized controlled trials.
For further reading please visit this link.
BW,
Hi Doctor,
My mother in law diagnosed with GBS in January and treatment was given through IV, ( dont know the name of the medication), now she cant walk again and doctor advised to admit her at hospital again for plasma exchange, my question is can she become normal after one plasma exchange?or do we have to do again after sometime,
thank you
Hi Nimesha,
Thank you for your comment.
There’s no cure for Guillain-Barre syndrome, but two types of treatments can speed recovery and reduce the severity of the illness:
1- Plasma exchange (plasmapheresis).
2- Immunoglobulin therapy (IVIg).
BW,
Plasmaphoresis will help for auto immune disease before a open heart surgery or to treat conditions that block arteries for autoimmune disease
Hi Erika,
Thank you for reaching out.
Plasmapheresis can be useful in managing severe autoimmune conditions before open-heart surgery, especially if there is a risk of acute exacerbation.
However, it is generally part of a broader strategy that includes stabilizing the underlying autoimmune condition and managing cardiovascular risk factors.
The treatment plan should be tailored to the patient’s specific needs and involve a multidisciplinary team.
Best wishes,
Dr. M. Abdou