Cold Agglutinin Disease
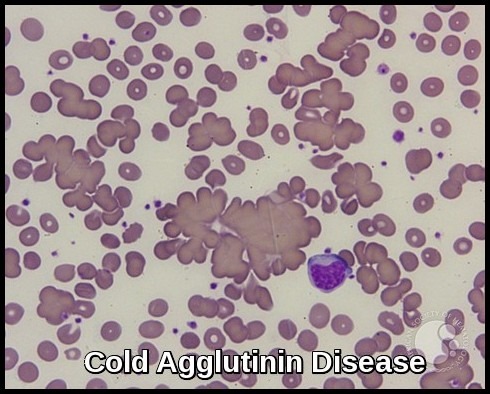
Cold Agglutinin Disease – Peripheral smear demonstrating prominent RBC agglutination due to IgM cold autoantibodies, a classic diagnostic feature of CAD.
Cold Agglutinin Disease (CAD) is a rare form of autoimmune hemolytic anemia (AIHA) caused by cold-reactive autoantibodies (cold agglutinins), usually immunoglobulin M (IgM), that bind to red blood cells at temperatures below 30°C (86°F). These antibodies induce red blood cell agglutination and activate the classical complement pathway, leading to predominantly complement-mediated hemolysis, particularly in peripheral body areas exposed to cold. CAD is classified under ICD-10 code D59.1 (Autoimmune hemolytic anemia) and accounts for approximately 15% of all AIHA cases, typically affecting older adults, most commonly those over 55 years of age.
The disease may occur as a primary condition, often associated with an underlying clonal B-cell lymphoproliferative disorder, or as a secondary syndrome linked to infections (such as Mycoplasma pneumoniae or Epstein–Barr virus), autoimmune diseases, or overt lymphoid malignancies. In addition to hemolytic anemia, CAD is increasingly recognized as a disorder with prominent cold-induced circulatory manifestations, including acrocyanosis and Raynaud-like symptoms, reflecting red blood cell agglutination in cooler peripheral circulation.
Early recognition of cold agglutinin disease, appropriate diagnostic evaluation with direct antiglobulin (Coombs) testing—typically showing complement (C3d) positivity, and a comprehensive hematologic assessment are essential for accurate diagnosis. Advances in understanding CAD as a complement-driven disorder have led to the emergence of targeted therapies, improving clinical outcomes and reducing the risk of cold-induced hemolytic exacerbations.
Clinical features:
Cold agglutinin disease (CAD) typically presents as a chronic, relapsing autoimmune hemolytic anemia, although acute exacerbations may occur. Patients commonly experience fatigue, pallor, and reduced exercise tolerance, reflecting the degree of anemia. Hemolysis in CAD is predominantly complement-mediated and often extravascular, though intravascular hemolysis may occur during severe episodes.
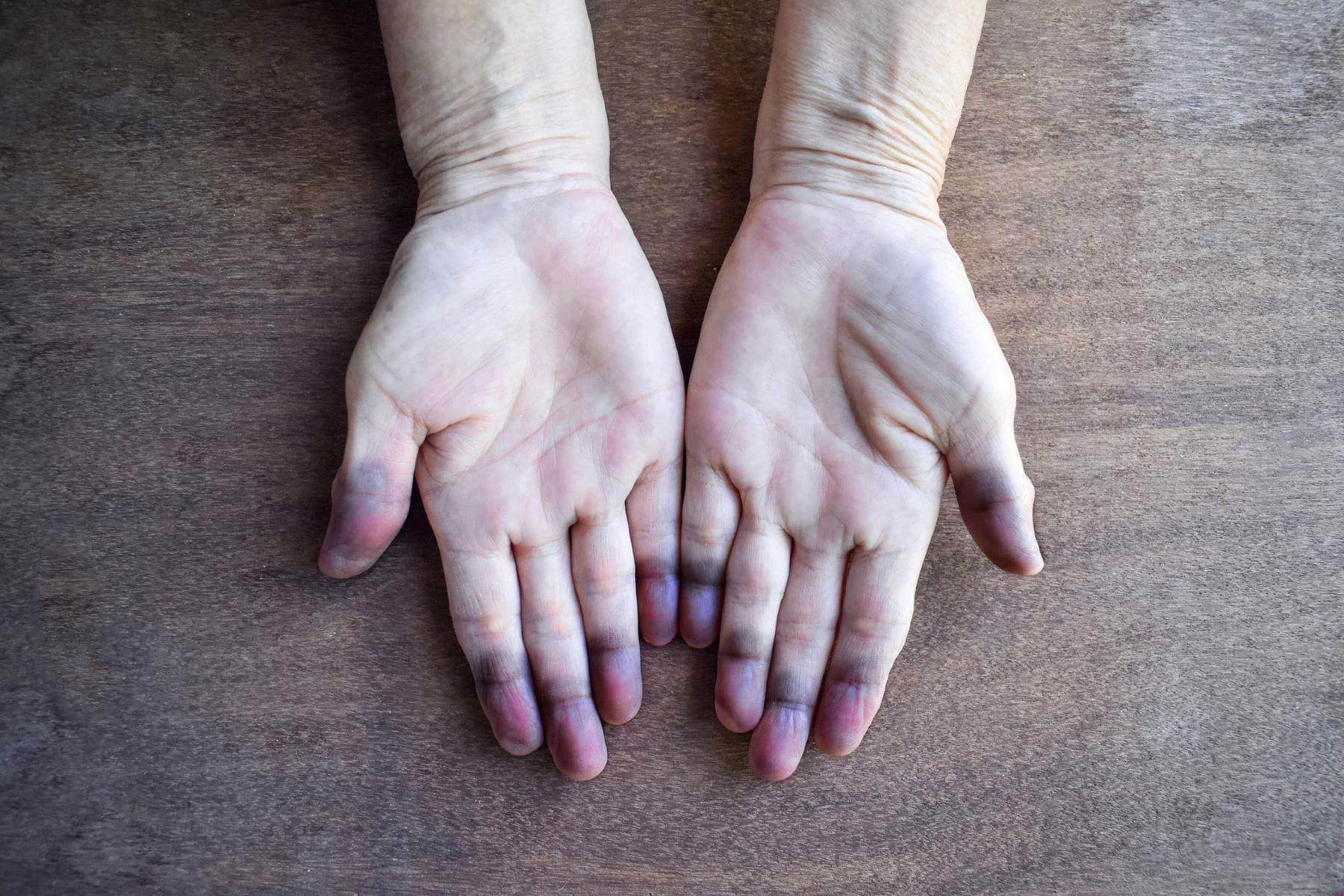
In addition to anemia-related symptoms, CAD is characterized by cold-induced circulatory manifestations due to red blood cell agglutination in peripheral tissues. These include acrocyanosis, livedo reticularis, and Raynaud-like symptoms affecting the fingers, toes, ears, and nose, which may be persistent or triggered by cold exposure.
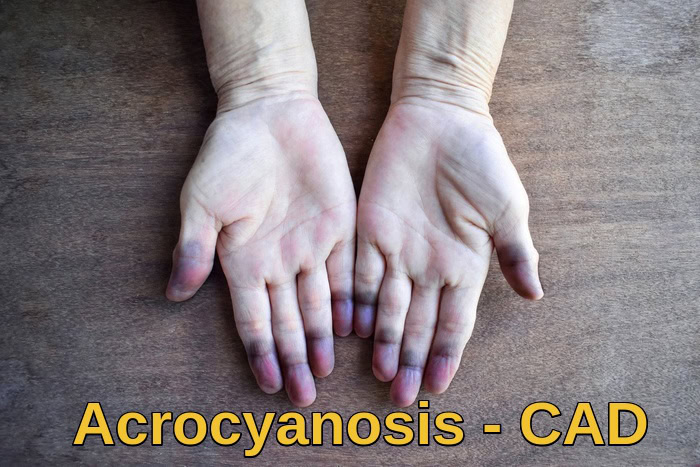
Acrocyanosis in Cold Agglutinin Disease demonstrating cold-induced peripheral cyanosis due to red blood cell agglutination and impaired microcirculation.
During hemolytic exacerbations, patients may develop dark urine (hemoglobinuria), jaundice, and, less commonly, systemic symptoms such as back or leg pain, headache, and gastrointestinal upset. Hepatosplenomegaly may be present, particularly in cases associated with underlying lymphoproliferative disorders.
Cold exposure and intercurrent infections are well-recognized triggers of disease activity, and symptoms often worsen during colder months. Episodes of acute hemolysis with hemoglobinemia and hemoglobinuria are more likely in winter and in secondary forms of the disease.
The clinical course may vary: some patients present with an abrupt onset of anemia and hemoglobinuria, while others experience a more gradual and insidious progression with chronic symptoms and intermittent exacerbations.
Classification:
Cold agglutinin disease (CAD) is broadly classified into primary (idiopathic or clonal) and secondary forms, based on the underlying etiology.
Primary CAD is now recognized as a distinct clonal B-cell lymphoproliferative disorder of the bone marrow, characterized by the production of monoclonal immunoglobulin M (IgM), most commonly of the kappa light chain subtype. Unlike overt lymphomas, primary CAD is typically associated with an indolent, low-grade lymphoproliferative process confined to the bone marrow and may not fulfill criteria for a defined lymphoma entity, although it shares features with conditions such as lymphoplasmacytic lymphoma and Waldenström’s macroglobulinemia.
Secondary cold agglutinin syndrome (CAS) occurs in association with identifiable underlying conditions, including infections (notably Mycoplasma pneumoniae, Epstein–Barr virus causing infectious mononucleosis, and cytomegalovirus), autoimmune diseases, and overt lymphoid malignancies such as non-Hodgkin lymphoma. In these cases, cold agglutinins may be monoclonal or polyclonal, depending on the underlying trigger.
From a clinical perspective, primary CAD and lymphoma-associated cases tend to follow a chronic, relapsing course, whereas infection-related cold agglutinin syndrome is usually acute and self-limited, often resolving with treatment of the underlying infection.
Diagnosis:
In some cases, the diagnosis of cold agglutinin disease (CAD) is initially suspected incidentally on a routine complete blood count (CBC), where abnormal red blood cell agglutination may lead to spurious results, such as elevated mean corpuscular volume (MCV) or falsely low red cell counts. These findings should prompt further evaluation.
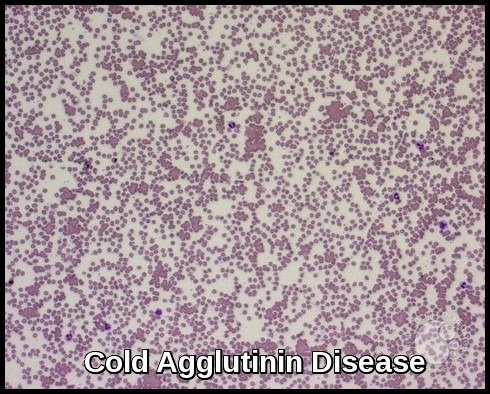
Cold Agglutinin Disease – Low-power view highlighting widespread RBC agglutination caused by IgM cold autoantibodies, a hallmark finding in CAD.
The diagnosis of CAD is established based on a combination of clinical evidence of hemolytic anemia and laboratory confirmation of complement-mediated autoimmunity. The hallmark diagnostic finding is a positive direct antiglobulin test (DAT) with a C3d-positive and IgG-negative pattern, reflecting complement deposition on red blood cells. In addition, circulating cold agglutinins, typically monoclonal IgM antibodies with a significant thermal amplitude, are detected in the serum.
Unlike CAD, warm autoimmune hemolytic anemia (AIHA) is predominantly mediated by IgG autoantibodies and usually demonstrates an IgG-positive DAT. Paroxysmal cold hemoglobinuria (Donath–Landsteiner hemolytic anemia) is caused by a biphasic hemolysin, typically a polyclonal IgG antibody directed against the P antigen, and is confirmed by the Donath–Landsteiner test, distinguishing it from CAD.
In secondary cases, particularly those associated with infections or lymphoproliferative disorders, peripheral blood findings may include lymphocytosis and the presence of atypical lymphocytes, depending on the underlying cause.
The differential diagnosis of CAD includes mixed autoimmune hemolytic anemia, in which both warm (IgG-mediated) and cold (complement-mediated) antibodies coexist, often resulting in a DAT positive for both IgG and C3d.
Investigations:
- Complete blood count (CBC) and blood film
May show anemia with red blood cell agglutination; spurious results such as elevated MCV and reduced RBC count can occur due to in vitro agglutination. Blood film may demonstrate red cell clumping and features of hemolysis. - Hemolysis screen
Includes reticulocyte count (usually elevated), total and indirect bilirubin (elevated), lactate dehydrogenase (LDH) (elevated), reduced serum haptoglobin, and urinalysis for hemoglobinuria.
Direct antiglobulin test (DAT) typically shows a C3d-positive and IgG-negative pattern, consistent with complement-mediated hemolysis. - Cold agglutinin testing
Measurement of cold agglutinin titre and thermal amplitude. Blood samples must be collected and transported warm (37°C) to avoid false results due to in vitro agglutination. - Serum immunoglobulins and serum protein electrophoresis (SPEP)
Useful to detect a monoclonal IgM paraprotein, commonly seen in primary CAD or associated lymphoproliferative disorders. Immunofixation may be required for further characterization. - Serum folate
Assessed due to increased consumption in chronic hemolysis; deficiency may exacerbate anemia. - Infection screen
Serology for Mycoplasma pneumoniae, Epstein–Barr virus (EBV), cytomegalovirus (CMV), HIV, and hepatitis C virus, particularly in suspected secondary cold agglutinin syndrome. - Cryoglobulin testing
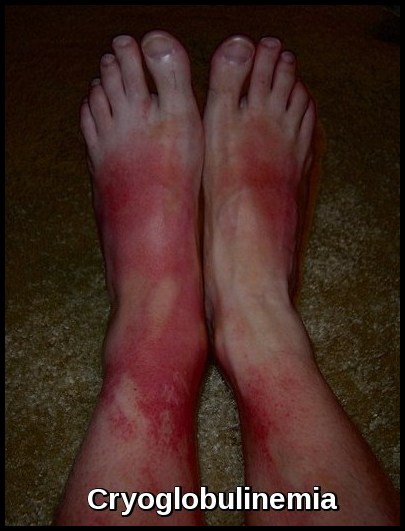
Should be performed selectively when there are atypical features such as vascular purpura, hyperviscosity, or markedly elevated IgM levels, or in the presence of hepatitis virus infection.

Qualitative test for cryoglobulin demonstrating visible cryoglobulin deposits in the patient’s serum at 4°C and their disappearance after warming to 37°C, confirming temperature-dependent precipitation.
- Imaging studies
- Chest radiograph: indicated if respiratory symptoms are present; may reveal pulmonary infiltrates in Mycoplasma pneumoniae infection or suggest lymphadenopathy.
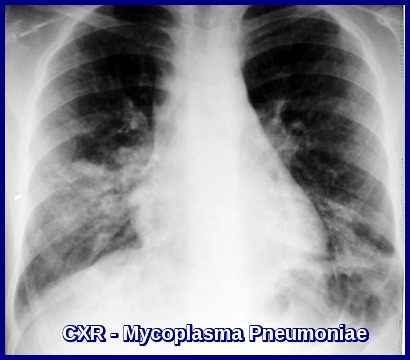
Chest X-ray in Mycoplasma pneumoniae infection demonstrating bilateral patchy interstitial infiltrates typical of atypical pneumonia, commonly associated with cold agglutinin hemolysis.
-
- CT scan of chest, abdomen, and pelvis: performed when an underlying lymphoproliferative disorder is suspected, to assess for lymphadenopathy, hepatosplenomegaly, or occult malignancy.
- Bone marrow examination (when indicated)
Recommended in suspected primary CAD or when a lymphoproliferative disorder is likely, to identify a clonal B-cell population and guide further management.
Treatment:
Patients with mild anemia and minimal clinical symptoms may not require active pharmacologic treatment, and strict avoidance of cold exposure remains the cornerstone of management. Keeping the patient warm, including during medical procedures and transfusions, is often sufficient in mild cases. In secondary cold agglutinin syndrome, treatment should be directed at the underlying cause; for example, Mycoplasma pneumoniae infection may be treated with macrolide antibiotics such as azithromycin or clarithromycin.
Active treatment is indicated in patients with symptomatic anemia, transfusion dependence, or clinically significant cold-induced circulatory symptoms such as severe acrocyanosis or Raynaud-like phenomena that impair quality of life or threaten tissue viability. Folic acid supplementation should be considered in all patients with ongoing hemolysis to prevent or correct folate deficiency.
Cold agglutinin disease is typically refractory to corticosteroids, and their routine use is not recommended. Rituximab, an anti-CD20 monoclonal antibody, is a well-established first-line therapy for symptomatic patients, either as monotherapy or in combination with agents such as bendamustine, which may provide more durable responses in selected cases.
Recent advances have established CAD as a complement-mediated disorder, leading to the development of targeted therapies. Sutimlimab, a monoclonal antibody that inhibits C1s in the classical complement pathway, has demonstrated rapid and sustained control of hemolysis, improvement in hemoglobin levels, and reduced transfusion requirements, and is increasingly used in appropriate patients.
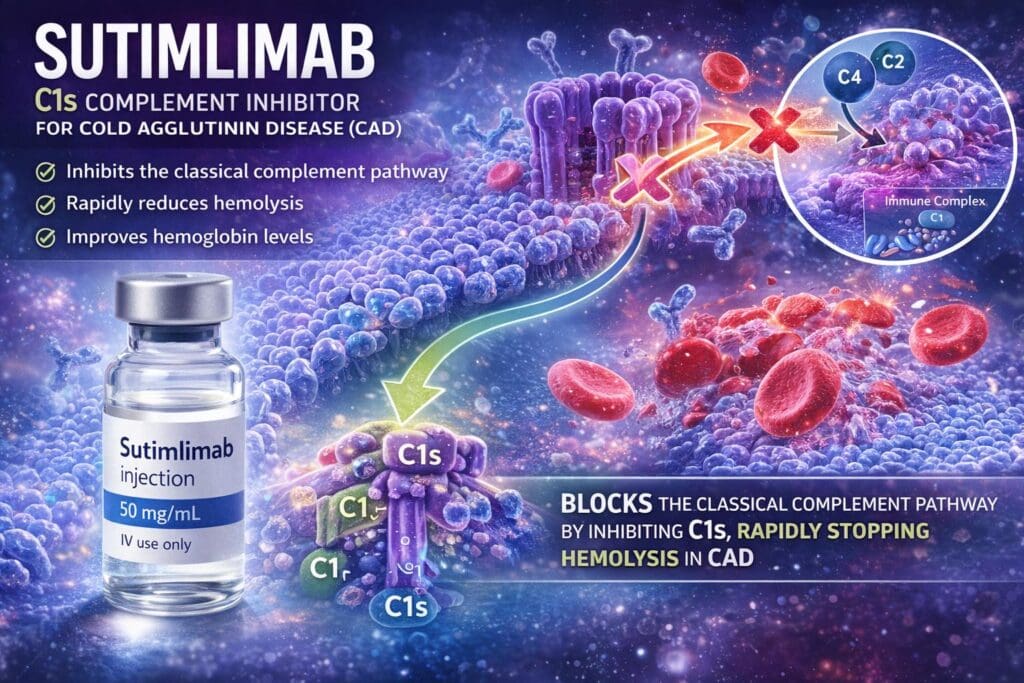
Sutimlimab in Cold Agglutinin Disease illustrating targeted inhibition of the classical complement pathway (C1s), leading to rapid reduction in hemolysis and improvement in hemoglobin levels.
In cases associated with an underlying lymphoproliferative disorder, treatment should be directed at the clonal disease, with modern chemoimmunotherapy regimens preferred over older agents such as chlorambucil or cyclophosphamide, which are now less commonly used.
Red blood cell transfusion may be required in severe anemia, and blood should be warmed prior to administration to prevent exacerbation of hemolysis. Plasmapheresis can provide temporary reduction of circulating IgM antibodies and is useful in severe or life-threatening hemolysis, or as a bridging measure while other therapies take effect. It may also be considered prior to hypothermic surgical procedures.
Splenectomy is generally ineffective in CAD because hemolysis occurs predominantly in the liver rather than the spleen. It is not routinely recommended, although it may have a limited role in selected cases, particularly when splenic involvement or diagnostic uncertainty exists.
Prognosis:
Cold agglutinin disease (CAD) typically follows a chronic, relapsing course, and in many patients the overall prognosis is relatively favorable with appropriate supportive care and avoidance of cold exposure. However, the disease can be associated with significant morbidity due to persistent hemolysis, fatigue, and cold-induced circulatory symptoms that may impair quality of life.
The prognosis is influenced by disease severity and underlying etiology. Patients with mild anemia and stable disease often have a benign course, whereas those with recurrent or severe hemolysis may develop transfusion dependence and complications related to chronic anemia. In addition, patients with CAD associated with an underlying lymphoproliferative disorder may have outcomes determined largely by the behavior of the underlying clonal disease.
Recent advances in therapy, particularly the introduction of complement inhibitors such as sutimlimab, have significantly improved disease control and quality of life, reducing hemolysis and transfusion requirements in many patients.
Overall, while CAD is rarely life-threatening, careful long-term monitoring is required to detect disease progression, treatment complications, and the emergence or evolution of associated lymphoid malignancies.
Summary:
Cold agglutinin disease (CAD) is a rare, complement-mediated autoimmune hemolytic anemia caused by IgM autoantibodies that bind red blood cells at low temperatures, leading to agglutination and hemolysis. It accounts for approximately 15% of autoimmune hemolytic anemia cases and mainly affects older adults. CAD may be primary, related to a clonal B-cell disorder, or secondary to infections, autoimmune diseases, or lymphoproliferative malignancies.
Clinically, CAD presents with a combination of chronic hemolytic anemia and cold-induced circulatory symptoms such as acrocyanosis and Raynaud-like changes. Diagnosis is based on evidence of hemolysis and a characteristic direct antiglobulin test (DAT) showing C3d positivity with IgG negativity, along with detection of cold agglutinins.
Management depends on disease severity. Mild cases may require only cold avoidance, while symptomatic patients benefit from therapies such as rituximab or complement inhibitors like sutimlimab, which reduce hemolysis and improve hemoglobin levels. Supportive care includes folate supplementation and warmed transfusions when needed.
Prognosis is generally favorable, although the disease is often chronic, and outcomes depend on severity and any underlying lymphoproliferative disorder.
Questions and Answers:
What are the key symptoms of Cold Agglutinin Disease?
Patients typically present with fatigue, pallor, jaundice, and dark urine due to chronic hemolytic anemia, along with cold-induced circulatory symptoms such as acrocyanosis, livedo reticularis, and Raynaud-like changes affecting the extremities.
What triggers flares in Cold Agglutinin Disease?
Cold exposure and intercurrent infections are the main triggers. Low temperatures enhance IgM binding to red blood cells and activate the classical complement pathway, leading to increased hemolysis and worsening symptoms.
How is Cold Agglutinin Disease diagnosed?
Diagnosis is based on evidence of hemolytic anemia and a characteristic direct antiglobulin test showing C3d positivity with IgG negativity, along with detection of cold agglutinins, elevated LDH and bilirubin, low haptoglobin, and red cell agglutination on blood film.
How is Cold Agglutinin Disease different from warm AIHA and paroxysmal cold hemoglobinuria?
CAD is mediated by IgM and complement activation with a C3d-positive DAT, whereas warm AIHA is IgG-mediated with IgG positivity on DAT. Paroxysmal cold hemoglobinuria is caused by a biphasic IgG antibody and confirmed by the Donath–Landsteiner test.
Is Cold Agglutinin Disease associated with infections?
Yes. Secondary cold agglutinin syndrome may occur with infections such as Mycoplasma pneumoniae, Epstein–Barr virus, and cytomegalovirus, and is usually acute and self-limited, resolving with treatment of the underlying infection.
What is the current treatment approach for Cold Agglutinin Disease?
Management depends on severity. Mild cases require strict cold avoidance, while symptomatic patients may benefit from rituximab-based therapy or complement inhibitors such as sutimlimab, which target the underlying complement-mediated hemolysis.
Is Cold Agglutinin Disease associated with lymphoma?
Primary CAD is now considered a clonal B-cell disorder of the bone marrow and is often associated with low-grade lymphoproliferative conditions, including lymphoplasmacytic lymphoma and Waldenström macroglobulinemia.
When is transfusion required in CAD and how is it given safely?
Transfusion is indicated in symptomatic or severe anemia. Red blood cells should be administered using in-line blood warmers to prevent cold-induced agglutination and exacerbation of hemolysis.
References:
Röth A, Berentsen S, Barcellini W, et al. Diagnosis and treatment of autoimmune hemolytic anemia in adults: Recommendations from the First International Consensus Meeting. Blood Reviews. 2021;47:100789. doi:10.1016/j.blre.2020.100789
Berentsen S, Barcellini W. Current challenges and future prospects in cold-antibody hemolytic anemia. Journal of Blood Medicine. 2020;11:13–25. doi:10.2147/JBM.S189231
Berentsen S, Randen U, Tjønnfjord GE. Cold agglutinin–mediated autoimmune hemolysis. Hematology/Oncology Clinics of North America. 2015;29(3):455–471
Swiecicki PL, Hegerova LT, Gertz MA. Clinical review of IgM-mediated cold agglutinin hemolysis. Blood. 2013;122(7):1114–1121
Sundar P, Shanmugasundaram S, Manickam S. Cryoglobulinemia diagnosed by peripheral smear examination: Value of a basic underestimated test. Journal of Laboratory Physicians. 2022;15. doi:10.1055/s-0042-1757420
Siegfried J. Acrocyanosis as an initial symptom of cold agglutinin disease: a case report. Rare Disease Advisor. Published May 21, 2025. Available at: https://www.rarediseaseadvisor.com/news/acrocyanosis-initial-symptom-cold-agglutinin-disease-case-report/
Agarwal SK, Ghosh PK, Gupta D. Cardiac surgery considerations in patients with cold-reactive proteins. Annals of Thoracic Surgery. 1995;60(4):1143–1150
Dacie JV. The autoimmune haemolytic anaemias. In: The Haemolytic Anaemias. 3rd ed. Edinburgh: Churchill Livingstone; 1992. Vol 3:210–362
Michel M. Overview of cold-antibody hemolytic anemia. Orphanet Encyclopedia. Available at: https://www.orpha.net/consor/cgi-bin/OC_Exp.php?lng=en&Expert=56425
Aljubran SA, Kaliner MA. Overview of cold-antibody autoimmune hemolysis: Background, pathophysiology, and etiology. Medscape. Available at: https://emedicine.medscape.com/article/135327-overview
American Society of Hematology (ASH). Cold agglutinin disease image bank. Available at: https://imagebank.hematology.org/imageset/909/cold-agglutininin-disease
National Organization for Rare Disorders (NORD). Cold agglutinin disease overview. Available at: https://rarediseases.org/rare-diseases/cold-agglutinin-disease
National Institutes of Health (NIH). Cold-induced hemolytic anemia. MedlinePlus. Available at: https://medlineplus.gov/ency/article/000578.htm
Centers for Disease Control and Prevention (CDC). Autoimmune hemolytic anemia overview. Available at: https://www.cdc.gov/blooddisorders
Keywords:
cold agglutinin disease, CAD, cold agglutinin hemolytic anemia, cold autoimmune hemolysis, cold antibody hemolysis, cold agglutinin syndrome, complement mediated hemolysis, IgM cold agglutinins, C3d positive DAT, autoimmune hemolytic anemia CAD, cold induced hemolysis, acrocyanosis cold agglutinin disease, Raynaud phenomenon CAD, cold agglutinin disease symptoms, cold agglutinin disease diagnosis, DAT C3 positive anemia, cold agglutinin titers, thermal amplitude cold agglutinins, red cell agglutination blood film, hemolytic anemia cold exposure, cold agglutinin disease in adults, primary cold agglutinin disease, secondary cold agglutinin syndrome, Mycoplasma pneumoniae cold agglutinins, EBV cold agglutinin syndrome, lymphoproliferative disorder CAD, Waldenstrom macroglobulinemia cold agglutinin, cold agglutinin disease treatment, rituximab cold agglutinin disease, sutimlimab CAD, complement inhibitor C1s CAD, cold agglutinin disease management, warmed blood transfusion CAD, plasmapheresis cold agglutinin disease, cold agglutinin disease prognosis, cryoglobulinemia vs CAD, hematology cold agglutinin disease, Ask Hematologist CAD, Dr Moustafa Abdou cold agglutinin disease

Request Online Consultation With Dr M Abdou
Fee: US$100
Secure payment via PayPal (credit and debit cards accepted)
Pay Now